|
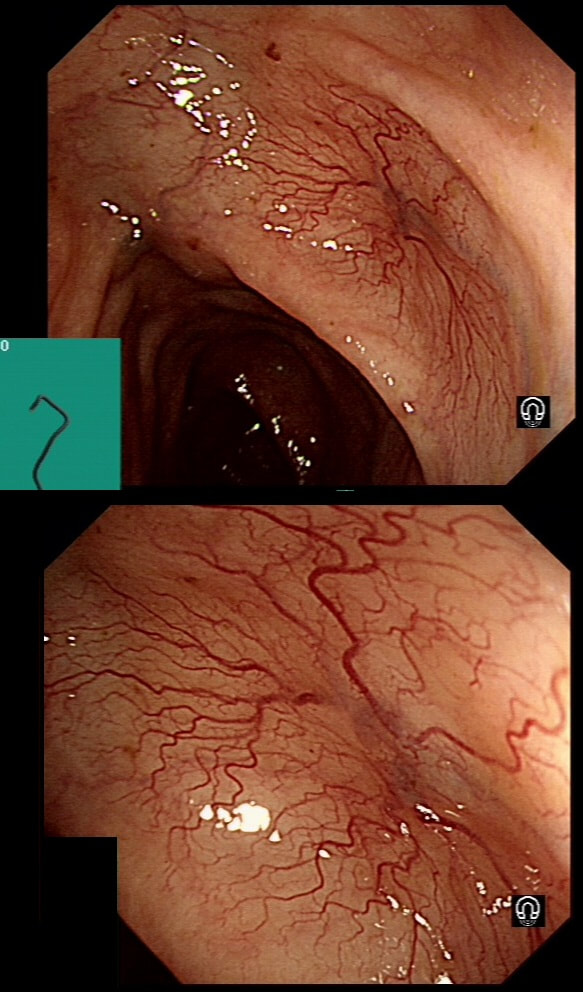
This lesion was found in the transverse colon of a patient with iron deficiency anaemia (IDA).
HOW WILL YOU APPROACH IT WITH APC?
■ Zap the centre first
Absolutely, ablate the central feeding vessel!
■ Start in periphery and work towards centre
Would work but take longer than neccessary
■ In a spoke an wheel pattern starting in the periphery
Would also work but would take longer
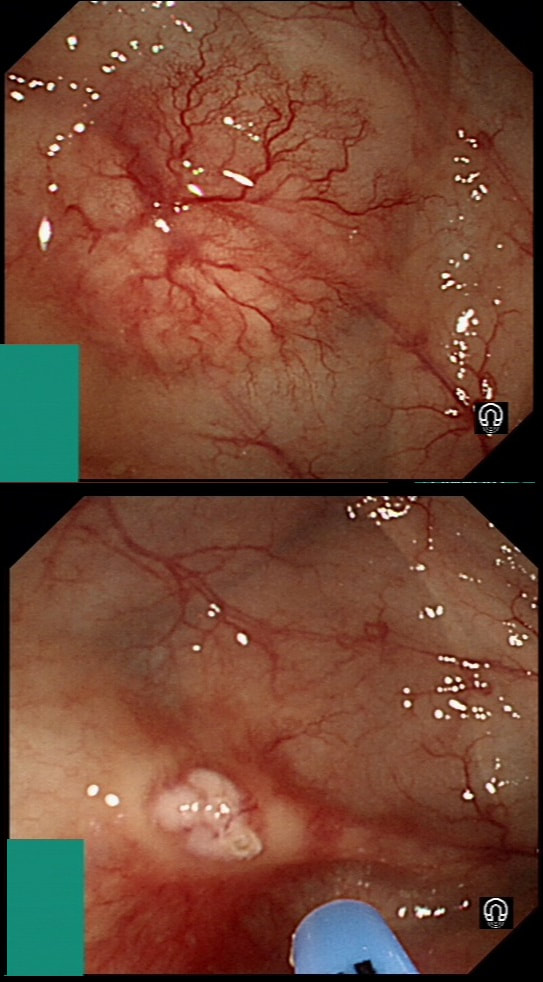
Previous biopsies have confirmed that this rectal polyp harbours TVA+HGD. It's removed by piecemeal EMR and at the end of the 2min video clip you see the end result. WHAT WOULD YOU DO NEXT?
■ Place clips
Always clips !
■ Wait for histology
And of course, you have requested an 'urgent report'!
■ Request EUS
Life is too short for EUS!
■ Organise an MRI
For local staging and confirmed T2,N1 cancer
■ Request staging CT
Looking for mets in the Chest+Abdomen+Pelvis - proved negative!
explanation
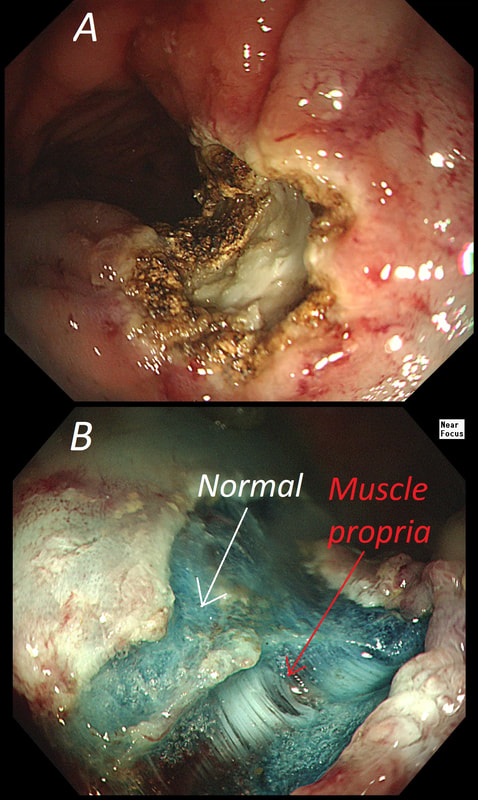
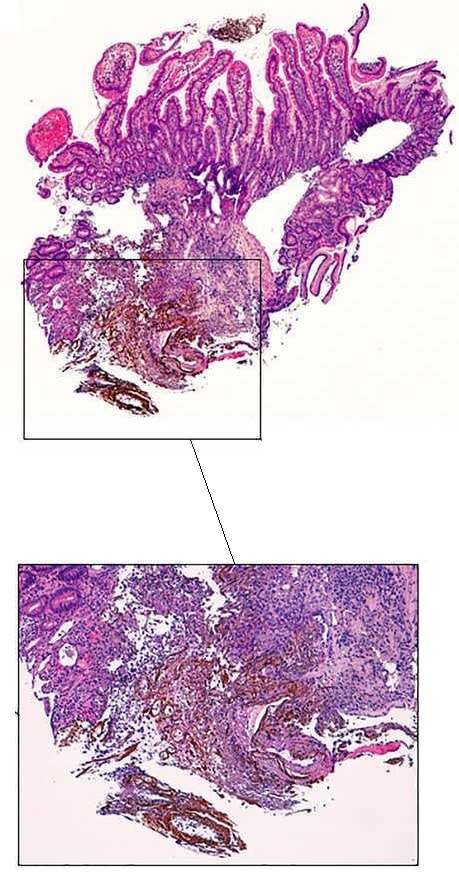
The learning point of this video clip is the EMR defect; it's white (image A) !!! Perhaps the picture below explains it better. Normally my EMR defects are blue (because I mix indigo carmine dye into the submucosal mix). In 'image B' below, the white arrow in the second image shows you what a 'healthy EMR defect should look like. IF you cut too deep, you can see the white, linear fibres of the muscle propria layer (red arrow). Of course this is a warning sign that you MUST carefully close the defect with lots of clips. Actually, next to the tip of the red arrow you can see a black 'micro-perforation' where the full thickness of the muscle layer has been breeched. Naturally, this is were your first clips goes!
Anyway, the mucosal defect in the video clip is just - white, without any linear muscle fibres. This is fibrosis! I have seen fibrosis like this below large sigmoid polyps which have been yanked about with the forceful sigmoid peristalsis. However, the more usual reason for this appearance is that you are looking at the fibrous tissue below a cancer, called 'desmoplasia'. Consequently, if I see a fibrous tissue in the base of the lesion I would do the following: Place clips (because I always do) Fast-track the histology Organise an MRI Request staging CT (chest+abdomen+pelvis) The ultimate diagnosis? Histology confirmed that the polyp was malignant and the imaging (of course requested at the time of the resection), confirmed a T2, N1 carcinoma. A week later, we had a full diagnosis. Unfortunately, the patient turns out not to be a surgical candidate. There is rarely unbridled joy after the endoscopic removal of a CRC ...
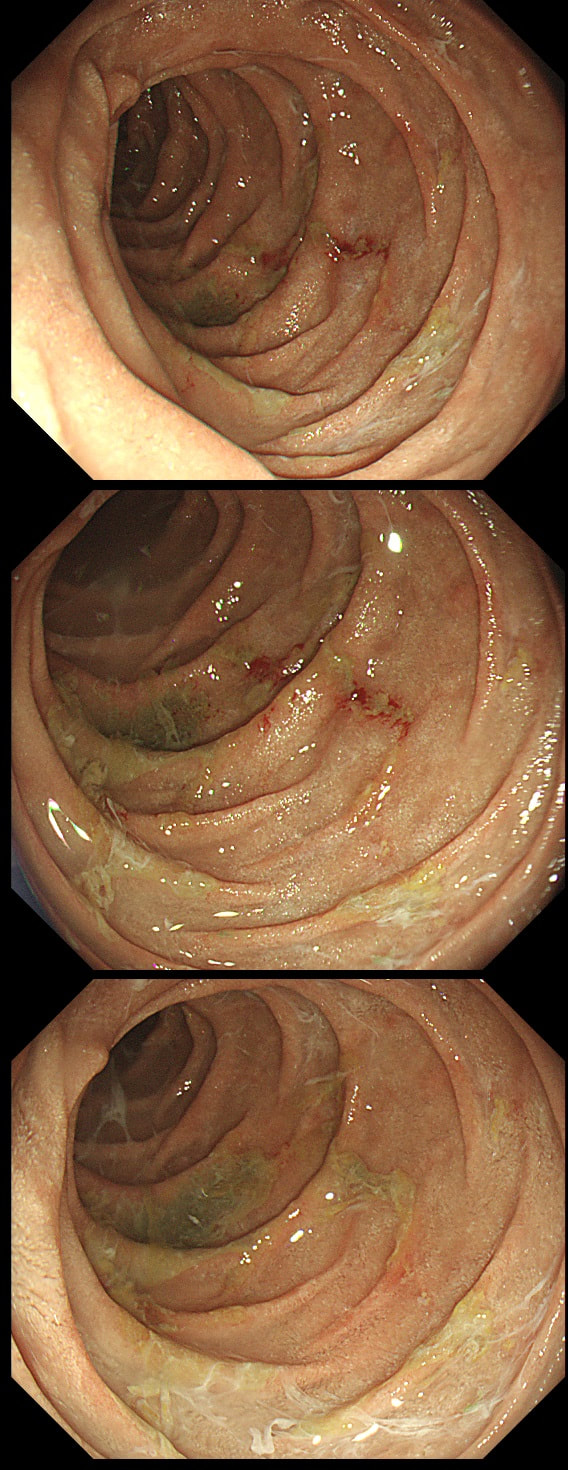
This patient was undergoing a gastroscopy for iron deficiency anaemia. A small ulcer is found in D2.
WHAT IS THE LIKELY AETIOLOGY?
■ Peptic ulcer
Statistically probably most likely diagnosis
■ Pill ulcer
Yes, but what pill?
■ Adenoma
Often do appear depressed here (IIc lesions) but not in this case
■ Early cancer
Early duodenal cancer usually look like sessile adenomas or, less commonly like indurated ulcerated lesions
explanation
If it wasn't for the somewhat odd, elongated appearance of the ulcer, it wouldn't seem odd. However, the only clue to the aetiology is that yellowish appearance. You may be familiar with 'iron pill gastritis' but can you get iron pill duodenitis? Yes you can! It has even been reported in the hypopharynx and the oesophagus. Those dark spots on the histology slides are crystalline iron deposits are easy to spot for the pathologists.
Of course, ferrous sulphate (Fe2+) tablets is the non-toxic form of iron (naturally). However, the tablets can produce mucosal inflammation if the iron becomes oxidised to Ferric iron (Fe3+). You may remember from medical school that most dietary nonheme iron is in the toxic, oxidized form of ferric iron (Fe3+). To absorb iron it first needs to be reduced, in the presence of acid, to ferrous iron (Fe2+). After this, the iron is easily transported across the enterocytes via a divalent metal-ion transporter 1 (DMT1) protein. Here is a link to an open access article if you want to brush up on iron absorption. Naturally, the treatment is to change to the liquid form of iron supplement which is much less toxic to the mucosa than solid iron tablets. These three polyps were removed en-bloc from the proximal colon. In an earlier examination 2 SSL's had been removed. A subsequent colonoscopy finds no further polyps. WHAT WOULD YOU ADVISE AS REGARDS SURVEILLANCE?
■ There is no need for surveillance
You may be right if the patient is old and with multiple comorbidities!
■ Surveillance every 2 years
Yes, this is what guidelines usually recommend for 'SPS'
■ Surveillance every 5 years
Too long if pt fulfills WHO criteria for SPS
explanation
If course, this patient has 'Serrated Polyposis Syndrome'. It's important to recognise that the serrated polyp count is cumulative over multiple colonoscopies. The recently updated 2019 WHO criteria for Serrated Polyposis Syndrome recognize two types of the syndrome: a 'proximal phenotype' with serrated polyps proximal to the rectum, all being ≥5 mm in size, with at least two being ≥10 mm in size (criterion I 2019), and a more 'distal phenotype' with more than 20 serrated polyps of any size throughout the large bowel (criterion II 2019) [Gastroenterology 2020;158:1520–23]. Personally, I believe that there are more than two subtypes of the Serrated Polyposis Syndrome. There is accumulating evidence that the syndrome includes multiple conditions with variable phenotypes and with different risks of progression to CRC [Gut 2010;59:1094–1100]. This would explain the huge range of cancer risk (25%-70%) in published studies [GIE 2016;83:563–65]. Of course, the likely mixture of several 'syndromes', makes writing guidelines difficult. A recent consensus update by the US Multi-Society Task Force recommend offering a follow-up colonoscopy to average risk patients based on number and size of the SSL's found [GIE 2020;91:463–85]. Interestingly, the US guidelines make a distinction between 'hyperplastic polyps' and 'sessile serrated polyps' although pathologists can't reliably make that distinction. Furthermore, the guidelines excludes patients with an increased life-time risk of cancer which of course excludes patients with Serrated Polyposis Syndrome. I find it all somewhat confusing! James East's BSG guideline 2017 [ Gut 2017;0:1–16 ] recommend surveillance every other year whilst the more recent BSG/ACPGBI guideline of 2020 [ Gut 2020;69:201– 23 ] would seem to suggest 3 yr for all 'high risk cases. But these guidelines expressively don't cover pts with hereditary cancers. Hereditary cancers are instead covered by the BSG/ACPGBI 2019 guideline [ Gut 2019;0:1–34 ] which recommends annual surveillance until all polyps are cleared and then every 2 years. Finally, I admit that I also take the age of the patient into consideration as well as the presence of both serrated and adenomatous polyps. A 40 year old person is surely more likely to benefit from surveillance than a 75 year old person with multiple comorbidities? In particular, I would worry about a young patient, perhaps 35 year old, with 1-2 large serrated polyps and perhaps only a single adenoma. Current guidelines don't flag these individuals up but personally, I would organise another surveillance colonoscopy in a few years time. Clearly, more research is needed to unpick the different serrated sub-pathways ! These polyps are found in a rectal pouch WHAT IS THE UNDERLYING DIAGNOSIS?
■ Serrated polyposis syndrome
Polyps is SPS are flat (usually)
■ FAP
Looks like a classic example of rectal surveillance in FAP but polyps do look odd don't they?
■ Juvenile polyposis
Yes! Pt had already developed a CRC and after a subtotal colectomy offered rectal surveillance
explanation
These polyps have a funny crypt pattern, but THERE IS A PATTERN! Accordingly, they are likely to be 'hamartomatous' (a non-sensical histological term which basically means overgrowth of normal tissue). Of course, this patient has Juvenile Polyposis syndrome. Although the polyps are 'hamartomatous', as in Peutz-Jeghers syndrome, there is a stark difference. The polyps in JP often turn dysplastic and are presumably the origin of this patients adenocarcinomas. In contrast, the polyps in PJS are extremely rarely reported to harvest dysplasia (I've never seen a dysplastic polyp in PJS). By the way, the WHO criteria for diagnosis of juvenile polyposis syndrome are one of either:
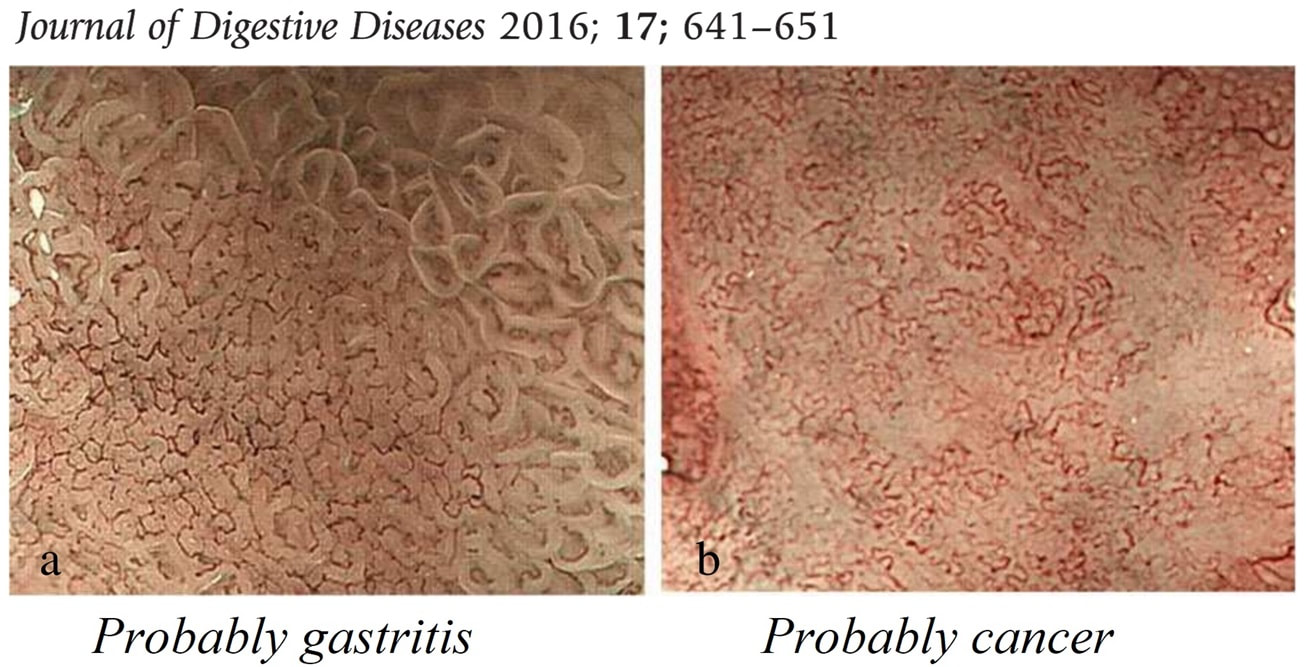
This patient is under surveillance after having had two previous EGC's. The stomach has been sprayed with indigo carmine dye when this lesion caught my eyes. WHAT IS THE LIKELY DIAGNOSIS?
■ Likely to be gastritis only
The non-staining is suspicious and rarely lie...
■ Likely to be a patch of intestinal metaplasia
There appears to be a crypt pattern in the centre but it can still be neoplastic!
■ Likely to be neoplastic
Non-staining, slightly raised and in an 'unstable stomach' - surely!
explanation
Clearly this is an unstable stomach having given rise to two previous EGC's. In patients attending for surveillance, I always spray indigo-carmine dye throughout the stomach using a spray catheter. I then look carefully at any spots where the dye seems to have slid off. This is such a 'lesion' which was confirmed as a subtle EGC. After my samples had confirmed my suspicion it was removed by ESD and was confirmed as HGD/IMca. I consider the both as the same histology as one pathologists HGD is another pathologists IMca. There was no LVI. However, I was surprised to find that one mucosal resection edge was involved by cancer. Looking back at the clip, I think that it's the 4 O'clock border which has an irregular extension which I failed to spot.
Most EGC's look like this, a shallow depression with a subtle elevated margin. I find the crypt pattern more difficult to interpret. However, as you know, the more destroyed it is, the more likely the lesion is to be poorly differentiated. |
Categories
All
|