|
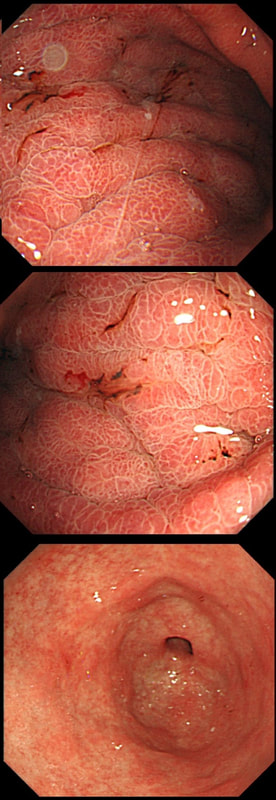
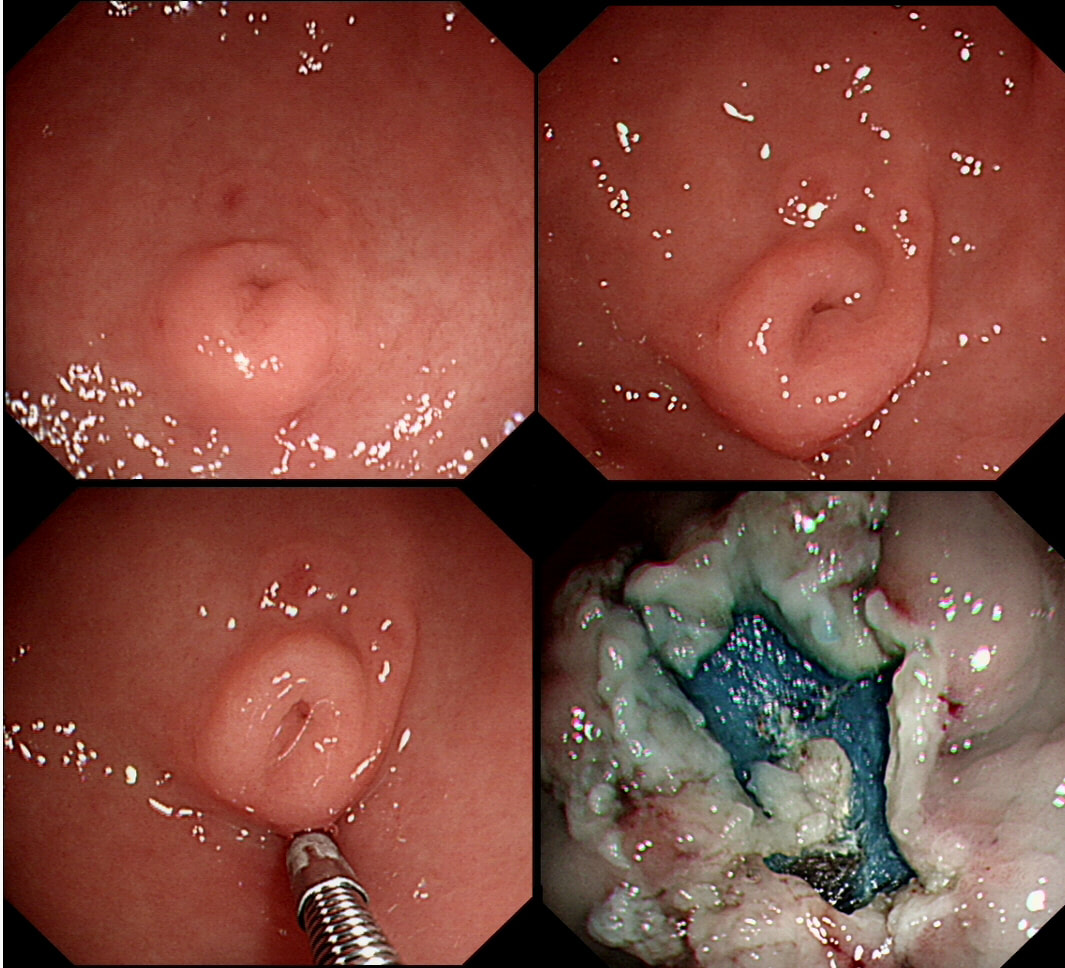
This 60 year old patient is undergoing a gastroscopy because of early satiety and weight loss. A CLO test was negative
WHAT IS THE LIKELY DIAGNOSIS?
■ Amyloidosis
Could indeed look like this BUT you'd expect the patient to be on haemodialysis!
■ Autoimmune gastritis
Indistinguishable from Hp gastritis but doesn't explain the symptoms
■ CMV gastritis
Possible but more common in children
■ Gastric lymphoma
Could be but you would normally expect ulceration
■ Diffuse type gastric cancer
Yes! The mucosa appears 'swollen' as it's infiltrated by malignant cells spreading between and below the crypts
explanation
As you know, the so called "Lauren’s classification" divides into two types of gastric cancer; the more common 'intestinal type' and the less common 'diffuse type'.
Actually the molecular workup done by “The Cancer Genome Atlas” Research Network" has actually identified four subtypes:
Intestinal type gastric cancer is becoming less common, probably because of the reduced prevalence of H. pylori, whilst the incidence of diffuse type cancers is not falling. There are other recognised differences between diffuse and intestinal type gastric cancer. The 'diffuse type' affect younger patients, the sex ratio is identical whilst the 'intestinal type' is more common in men. Presumably this means that known mutagens such as smoking and alcohol does not predispose to diffuse type gastric cancer? 'Diffuse type' cancers are more common in the proximal stomach and can be inherited as part of B-cadherin mutations. Finally, 'diffuse type' cancer develop BELOW the surface epithelium making them very difficult to spot. There is no irregularity of the overlying crypt or vessel pattern in these lesions. However, late detection is probably not the only reason why diffuse type cancer have a worse prognosis.
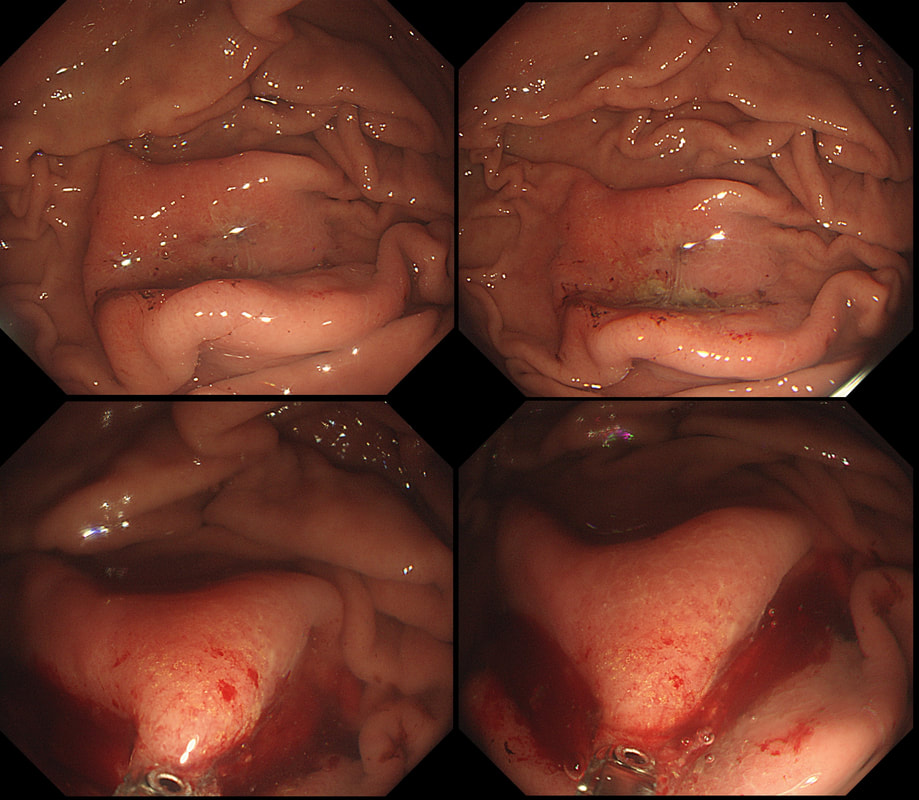
This lesion, on the greater gastric curve, was found in a patient undergoing gastroscopy because of iron deficiency anaemia
WHAT IS THE LIKELY DIAGNOSIS?
■ Helicobacter associated gastritis
Can look odd but the yellow pigment doesn't fit
■ CMV gastritis
Nope, CMV would give a more widespread gastritis with erosions
■ Gastric lymphoma
Could look similar but how about that yellow stain?
■ Gastric cancer
Should always be suspected in IDA but wrong this time
■ Something else
Yes! Those yellow intramucosal crystals is iron!
explanation
This is the typical yellow/brown stain of "Iron tablet gastropathy". The intramucosal deposits of extracellular crystalline iron are best seen with the "Perl stain". The iron is an irritant, inducing inflammation and there is usually erythema of the surrounding mucosa (see another example below). Mucosal erosions and ulceration have also been described.
A liquid form of iron supplement is thought to be less toxic to the mucosa and when you see this, you should suggest changing the iron formulation ! In addition, patients are often advised to take the iron tablets on an empty stomach which also increases the risk of gastric irritation. I always advice patients to take the iron (tablets or liquid) with food !
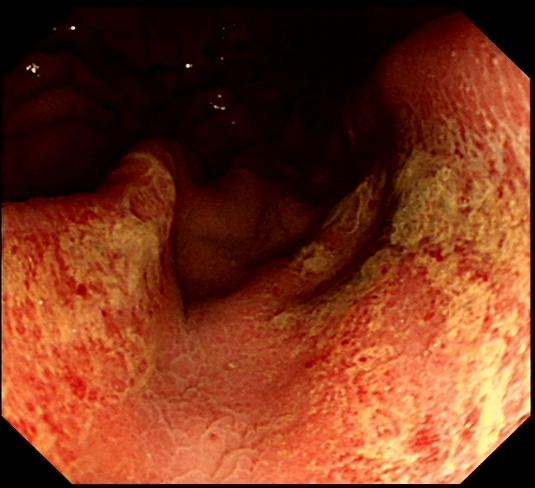
This 35-year-old man presented with a history of abdominal pain and weight loss. For the last 5 years he has suffered intermittent abdominal pains. He has recently been treated with lansoprazole for some weeks but with no response.
Hb 112 g/dl MCV 99 fl WCC 12.9 x 109 Platelets 395 x 109 Basal gastrin level: 180 pg/ml (normal <75) This is the patients gastric antrum. WHAT IS THE MOST LIKELY DIAGNOSIS?
■ Hp associated gastritis
Unfortunately, the wrong answer
■ Crohn's disease
You are correct (but do you know why?)
■ Zollinger Ellison's syndrome
INCORRECT!
■ Gastric lymphoma
INCORRECT!
■ Linitis plastica
INCORRECT!
explanation
Quite a nasty looking gastritis. All of the options seem to be on the table but what about the raised gastrin level? Could this be a case of ZE? Of course you can explain the elevated basal gastrin level! The patient is taking a PPI and low gastric acid levels will induce a positive feedback loop to raise the gastrin levels ! The second clue is the macrocytic anaemia... How do you explain this?
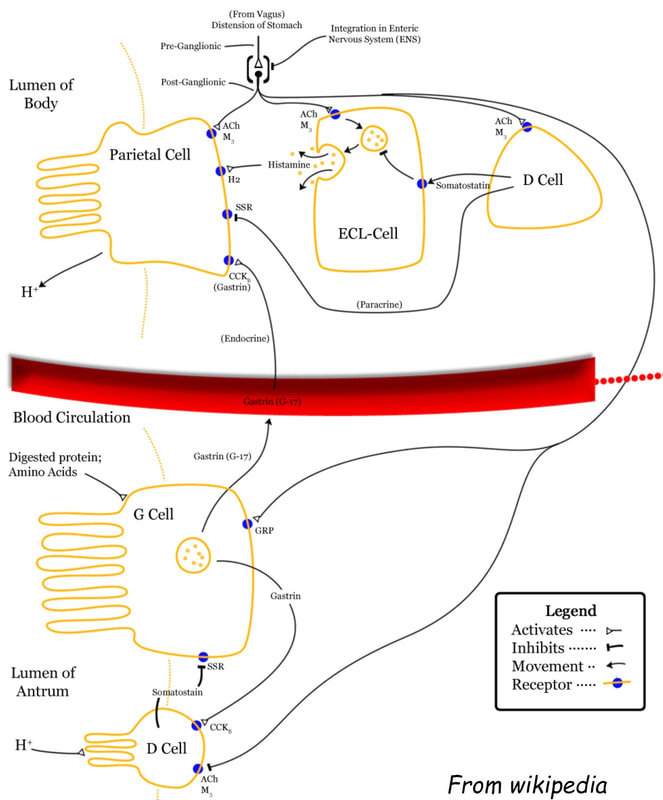
Of course the combination of abdominal pain and macrocytic is usually found in patients addicted to alcohol with recurrent attacks of pancreatitis. However, there is another possibility! The macrocytosis is due to vitamin B12 deficiency from terminal ileal Crohn's disease. The antral gastritis was also due to Crohn's! Courtesy ofWikipedia, there is a diagram below to remind you of the hormones involved in gastric acid secretion ! This patient is undergoing a gastroscopy because of abdominal pain. Some 20 years ago he underwent a distal gastrectomy for peptic ulcer disease. WHAT IS THE DIAGNOSIS?
■ a) Large hyperplastic polyp
No, there is more going on!
■ atrophic gastritis with malignant polyp
Closer to the truth but not the whole truth!
■ None of the above
What else could there be??!
explanation
You can tell that the mucosa must be atrophic by the mucosal xanthelasma at about 25seconds into the video. The polyp dangling at about 12 O'clock is covered with the same irregular surface as the base from which is arises. Finally, there is a large plaque-like area extending between 3-10 O'clock which does look concerning. It goes all the way up to the small bowel anastomosis. In general, large plaque-like cancers in the stomach do look intimidating but is usually intramucosal disease, provided that the appearance is uniform. The subpedunculated polyp indicate more 'heterogenous' disease. The obvious question is now, should we offer the patient an attempt at endoscopic resection or a 'completion gastrectomy'? The second part of that particular question is of course, 'is the patient actually a candidate for a completion gastrectomy?' At the time of my endoscopy, I didn't like the wide extend of the cancer and its heterogenous appearance. To also convince our MDT, I removed that subpedunculated polyp which was confirmed as intestinal-type intramucosal adenocarcinoma which was mainly 'moderately well differentiated' but with 'focal poor differentiation'. Predictably, the deep margin was positive, as the surface structure of both the polyp and the underlying gastric mucosa look the same. Although both an EUS and CT were reassuring, because the patient was a surgical candidate and also because of the large area of heterogenous disease, extending all the way up to the small bowel anastomosis, the patient was in the end offered a gastrectomy rather than an endoscopic resection. Ultimately, histology confirmed a very 'heterogenous disease' with areas within this 13cm lesion containing:
Is there any evidence that heterogenous gastric cancers (i.e. cancers with a mixed histology) are less likely to be cured by endoscopic resection? Actually there is! Ozeki et al looked at more than 3000 endoscopic resections for early gastric cancer and found that lesions with a mixed histology was significantly less likely to be cured by ESD (35-42% cure rate vs 76-92% cure rate). Ozeki Y. Mixed histology poses a greater risk for noncurative endoscopic resection in early gastric cancers regardless of the predominant histologic types. Eur J Gastro Hep 2021;32(2);186-193 Fortunately, there was no nodal disease. Almost everyone got this right when first published on our FoE facebook group.
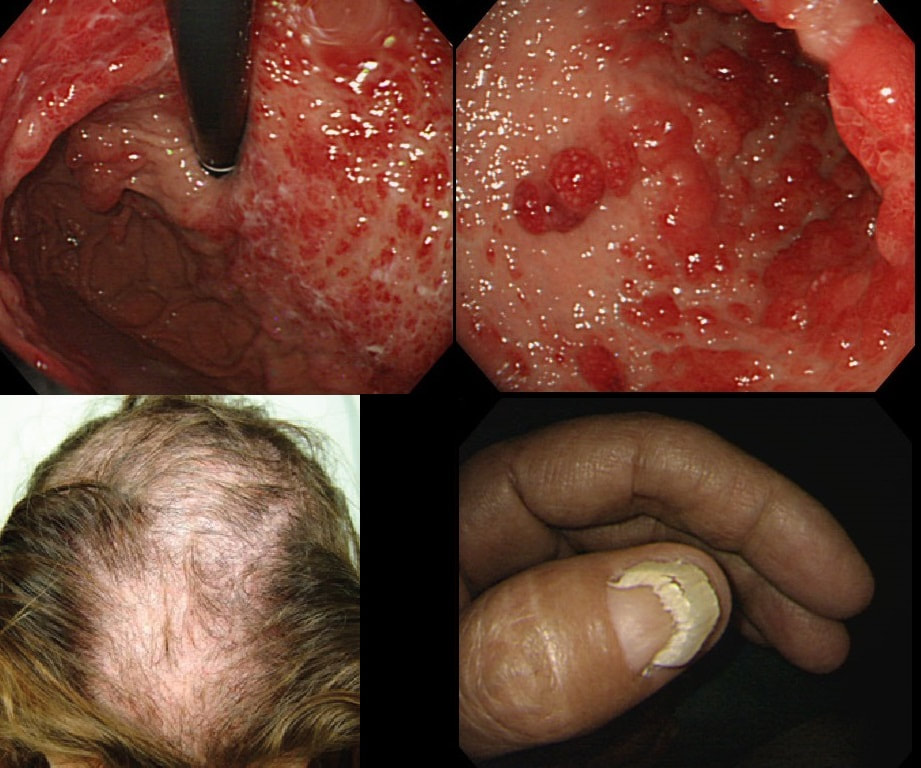
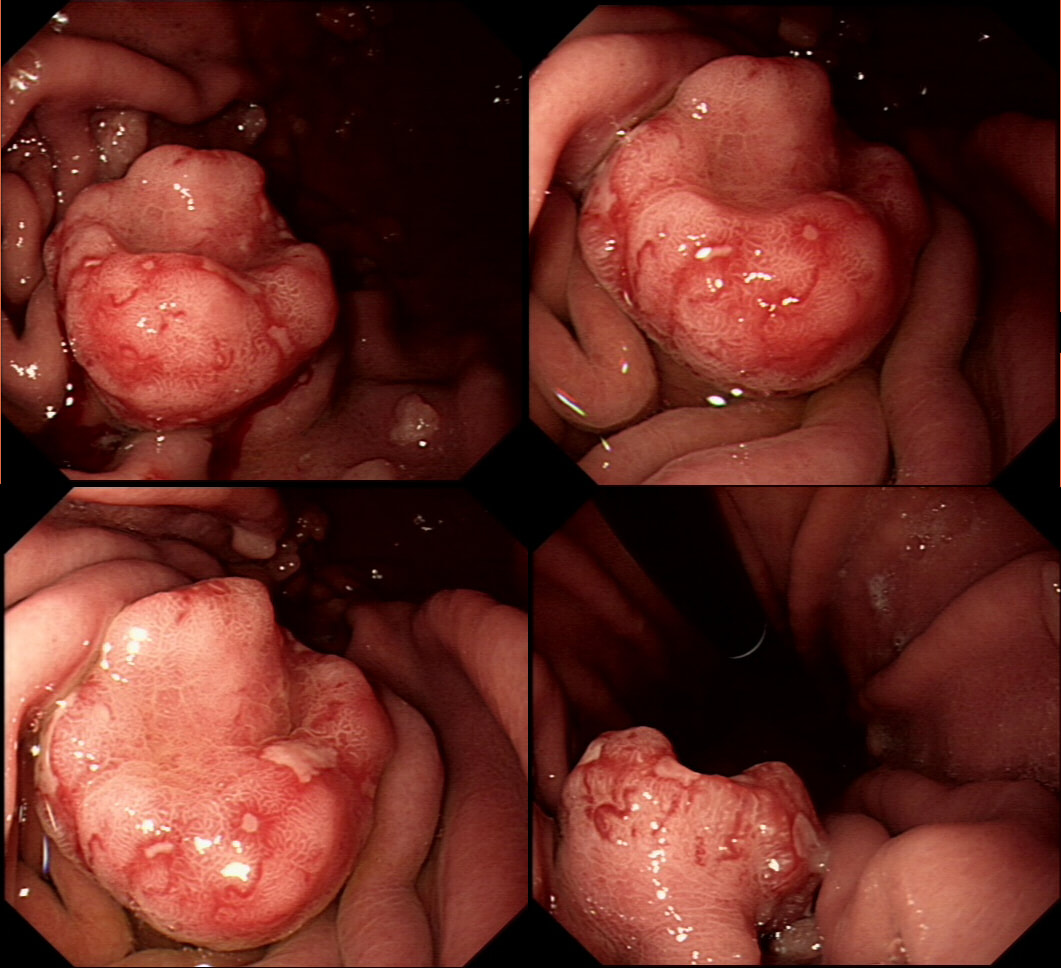
You are called into another endoscopy room to give an opinion on this gastric findings. It's a 65 year old man undergoing investigations for severe weight loss and anaemia. The anaemia was discovered by the dermatologists where he had been referred for investigations of suspected onychogryphosis and thinning of the hair.
WHAT IS THE EXPLANATION FOR THE GASTRIC POLYPS?
■ Likely hyperplastic polyps
INCORRECT!
■ Likely hamartomatous polyps
CORRECT! Clearly you know what the underlying diagnosis is?
■ Likely florid GAVE
INCORRECT!
■ Likely lymphoma
Gastric Lymphomas are usually ulcerated
■ Likely linitis plastica
INCORRECT! (although I guess it could be)
explanation
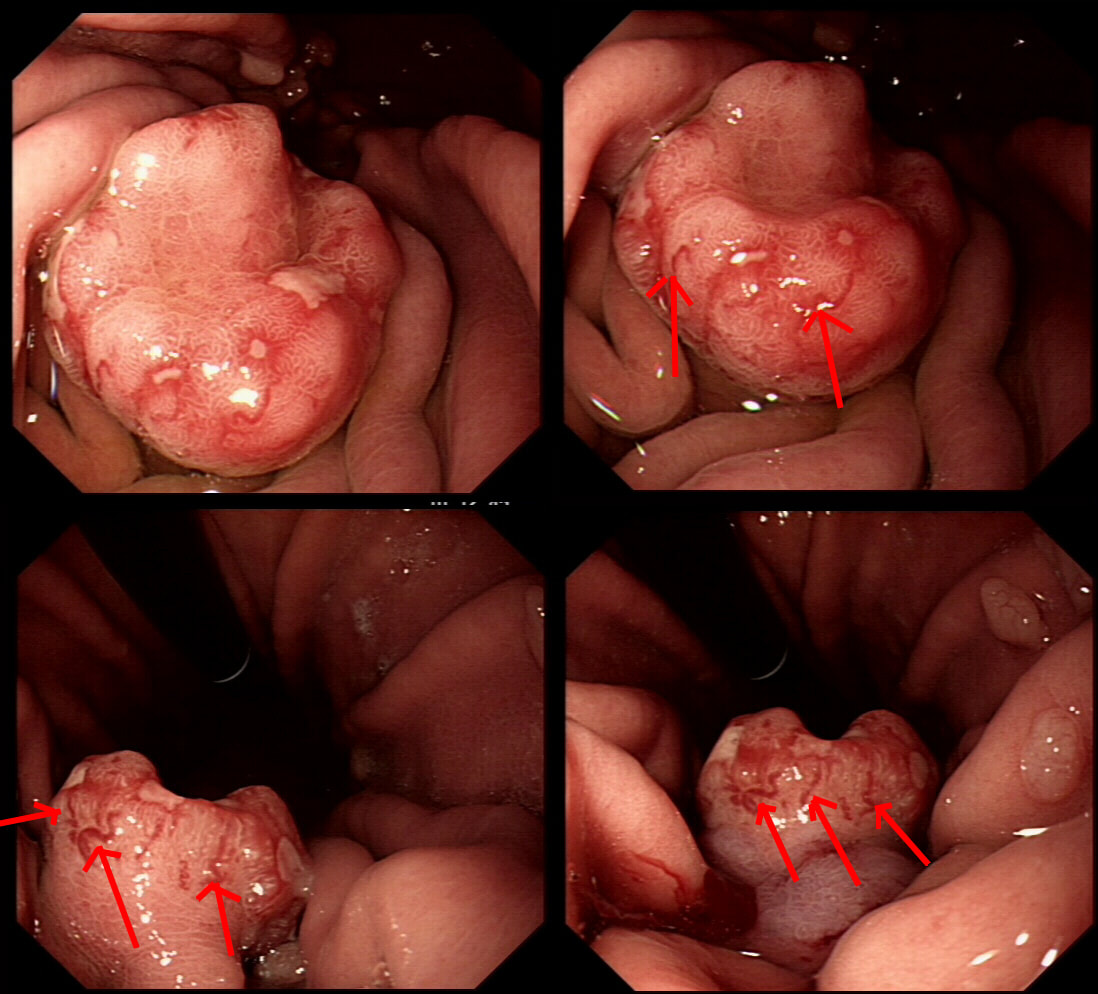
The nail dystrophy is not due fungal infection (onychogryphosis). In fact, this patient has Cronkhite-Canada syndrome. This enigmatic acquired syndrome is characterised by malabsorption, gastrointestinal polyposis, nail dystrophy, alopecia, cutaneous pigmentation, diarrhoea and weight loss. The nail dystrophy and skin pigmentation is clearly seen in the photograph. Other common symptoms include abdominal discomfort and a protein loosing enteropathy. The underlying cause is unknown. The cutaneous manifestations are probably all secondary to severe malnutrition due to diffuse small bowel mucosal involvement.
Histologically the mucosa is oedematous with dilated glands and an inflammatory cell infiltrate of the lamina propria. Gastrointestinal polyps are hamartomatous but may contain foci of adenomatous epithelium. The histopathologists will struggle in interpreting the biopsy findings and will mention things like 'marked foveolar hyperplasia', 'cystically dilated irregular glands', 'oedematous stroma' and 'scattered mixed inflammatory cells'. The differential diagnosis of such hamartomatous findings includes juvenile polyposis syndrome, Cronkhite-Canada syndrome, hyperplastic polyposis and Menetrier's disease. Of course, it will help them to know about the nails and hairloss etc!!! Malignant transformation has been reported and may not be as rare as initially thought . The prognosis is said to be poor but I'm not sure that this is still the case with 'modern management'. Interestingly, in this case a suspicious nodule was found (image below). It was removed endoscopically and found to be an early gastric cancer!
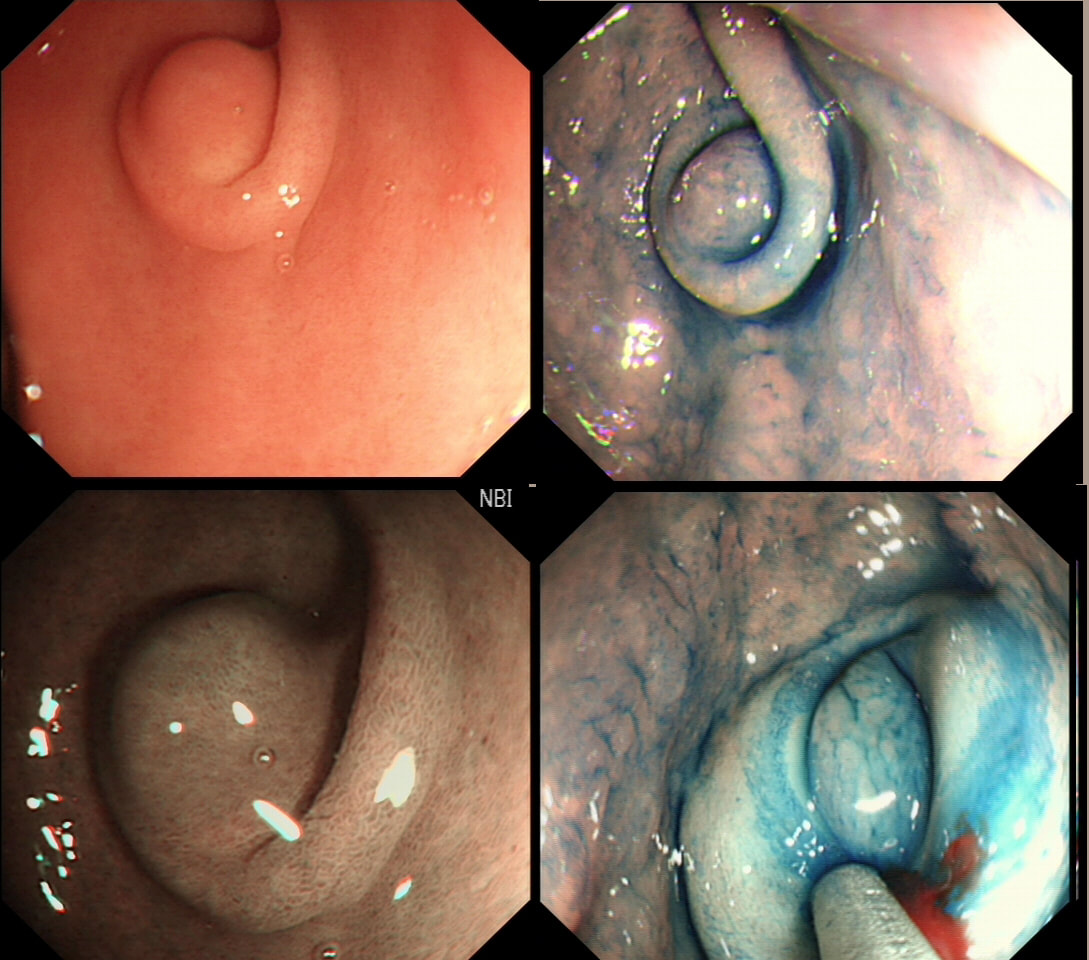
This antral lesion was found in a 25 yr old man undergoing gastroscopy for dyspepsia. Surface biopsies have only revealed a normal gastric mucosa.
WHAT WOULD YOU ADVICE?
■ This lesion does not require removal
Damn right it doesn't!
■ Another set of samples is required first
INCORRECT as these will only show normal mucosa again
■ an EUS is first required
INCORRECT! Because you will not be removing it anyway!
■ a CT should be done first
Don't be stupid!
■ I'll go ahead with the EMR/ESD
INCORRECT! Doing what you are told to do is not always the right thing do to !
explanation
This is a typical appearance of an 'ectopic pancreas' in the classical location along the greater curve of the antrum. The lesion even has a central dimple where the pancreatic duct finishes and a beautiful fold draped around it (which may not always be present in these lesions of course).
Sometimes I am asked to remove these for the 'reassurance' or to obtain a 'definite diagnosis' or because it's causing pain. Pain due to pancreatitis arising in these has been reported but is probably less common than finding that the rocking horse has shat on the floor. When the endoscopic appearances are typical, I don't believe that any further 'confirmation' is necessary. At Gastroscopy, I report these as: "small pancreatic rest was noted in the antrum. This is a normal, incidental finding of no significance" If you foolishly try to remove these, you will find that the endoscopic resection is actually quite difficult. The central pancreatic duct tethers the lesion down and therefore these never elevate well. You can see that in the bottom row image on the right. After struggling, you will end up with a mucosal defect and a central pink area (see image below). The resected fragments will only contain normal gastric mucosa and you will have to either dig deeper (have you heard of the expression: "to dig an even deeper hole for yourself?") or take some samples from the central pink area to capture some pancreatic duct tissue. If the referring team are steadfast that "proof is needed that the lesion is innocent", an EUS is safer and easier than an attempt at endoscopic resection.
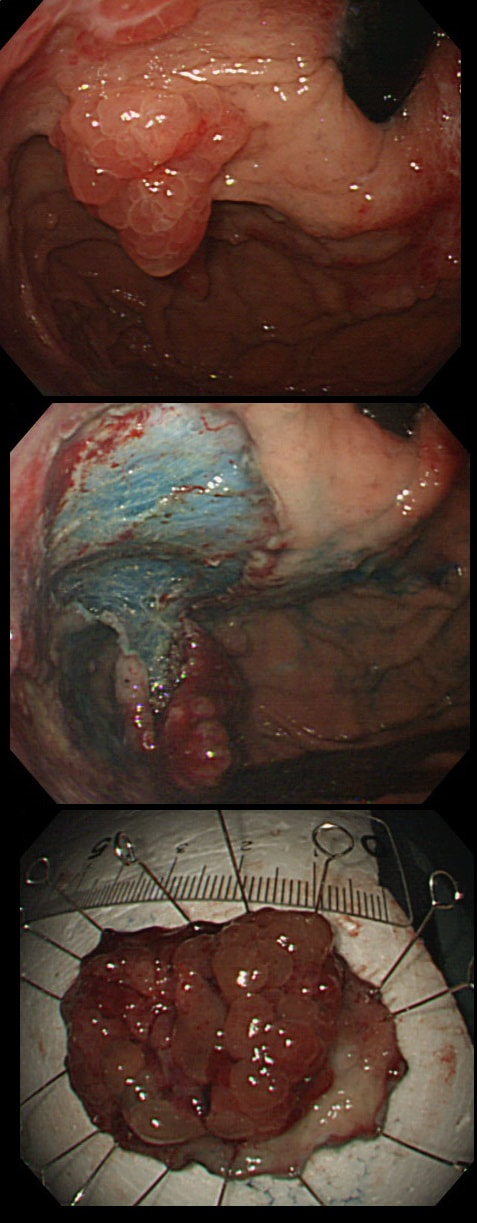
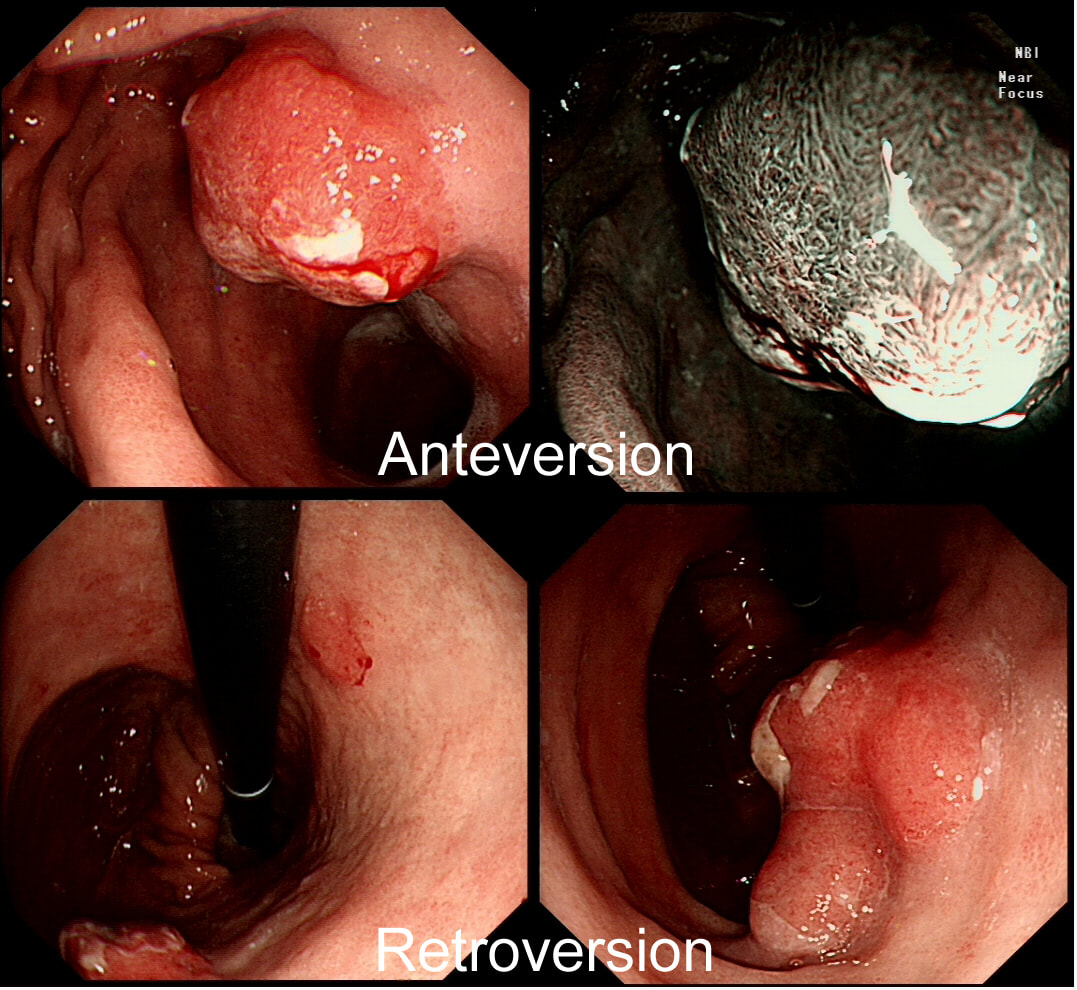
This suspicious looking 3cm gastric nodule was referred for an endoscopic removal after a CT and EUS had been reassuring. No samples have been taken as not to impede the resection
WHAT WOULD YOU DO NEXT?
■ First sample the nodule
Good idea but not good enough!
■ Sample nodule, antrum and gastric body
YES! To tell if this is a type III NET
■ Go ahead and resect as planned
NET's >2cm should be considered for surgery first!
explanation
This is a gastric neuroendocrine tumour (NET). You can tell because of the dilated vessels along its side (see below). A typical feature of all gastric NET's.
To remind you, gastric NET's are classified as; type I (70% - 80% of gastric NET's) linked with hypergastrinaemia secondary to an atrophic gastritis and classically appearing as multiple, small gastric nodules. The rare type II gastric NET account for about 5-8% is associated with hypergastrinaemia from a gastrin-secreting tumour such as in the MEN-1 syndrome or the Zollinger–Ellison syndrome. Finally, the type III NET (20%) is solitary, larger nodules with a high mitotic index arising in a healthy gastric mucosa. These are the ones not to miss as they need a cancer-like gastric resection particularly if 2cm or larger in size ! Sampling the nodule will confirm that it's a NET and give you it's 'mitotic index'. Grade III lesions (20% mitotic index) should certainly be resected surgically. However, also sampling the antrum and body is even better because if the mucosa is found not to be atrophic, this confirms that the lesion is a type III gastric NET !
Previous sampling of this gastric nodule has confirmed HGD and an EMR has been organised.
WHAT IS THE MOST APPROPRIATE MANAGEMENT?
■ Take further set of Samples!
No, because it's unlikely to change anything!
■ Organise staging CT and EUS first!
YES! This may well be invasive cancer!
■ Go ahead and resect it!
INCORRECT!
explanation
The correct approach would be to slow down a little! Pathologists would always like more samples but actually, surface samples may not be a true reflection of what is nestling deep within this lesion! This lesion is a little over 3cm in diameter and it would be very surprising if it wasn't an invasive cancer after all. Furthermore, there is a suspicious looking red, flat patch close to the shaft of the endoscope in the bottom row image on the left.
Ultimately, this patient underwent a gastrectomy which confirmed that the large nodule was indeed an invasive adenocarcinoma invading as deep as the muscle propria layer (T1b). In addition, there was multifocal LVI and a nearby involved node (N1). That red patch turned out to be a spot of HGD. Because EUS found a suspicious node, a gastrectomy was the most appropriate therapy. Of course, we don't normally organise a CT and EUS for endoscopically resectable lesions which are not thought to be malignant. Of course biopsies have indicated that this is HGD only. However, the lesion is chunky and its unthinkable that it's HGD only. More surface biopsies may or may not reveal the true nature of the lesion. And frankly it doesn't really matter because endoscopically this is likely to be an invasive cancer. It's a dysplastic nodule about 3cm in diameter and probably close to 2cm in height !!! Of course, the pathologists always want more samples but with a lesion of this size, of borderline resectability, a CT and EUS is more reassuring than further samples. The EUS revealed a chunky node and the patient had an appropriate gastrectomy. How about a a test-lift? Unfortunately, test-lifts are difficult to interpret in the stomach and is likely to be 'borderline' committing you to 'having a go' trying to resect the lesion endoscopically. Of course, there is nothing wrong with an 'attempt at EMR/ESD' but the point is that first you would like to have the reassurance of a normal CT and EUS. Of course, you would probably take another set of samples rather than just looking at the lesion but whatever the pathologists say, it will not trump the endoscopic diagnosis of; ' invasive cancer'. But why not just 'lob it off' for a definite diagnosis as well as staging? The reason is that a ESD resection of a cancer is very difficult. This is because the semi-translucent lifting plane disappears due to the desmoplastic reaction and you end up cutting blindly through opaque beige coloured tissue. Perforating during the resection of a cancer is potentially devastating for the patient as it can upstage the lesion to 'disseminated disease'. As the stakes are very high in cancer, it would be inappropriate to subject the patient to a significant risk with only a slim chance of benefitting.
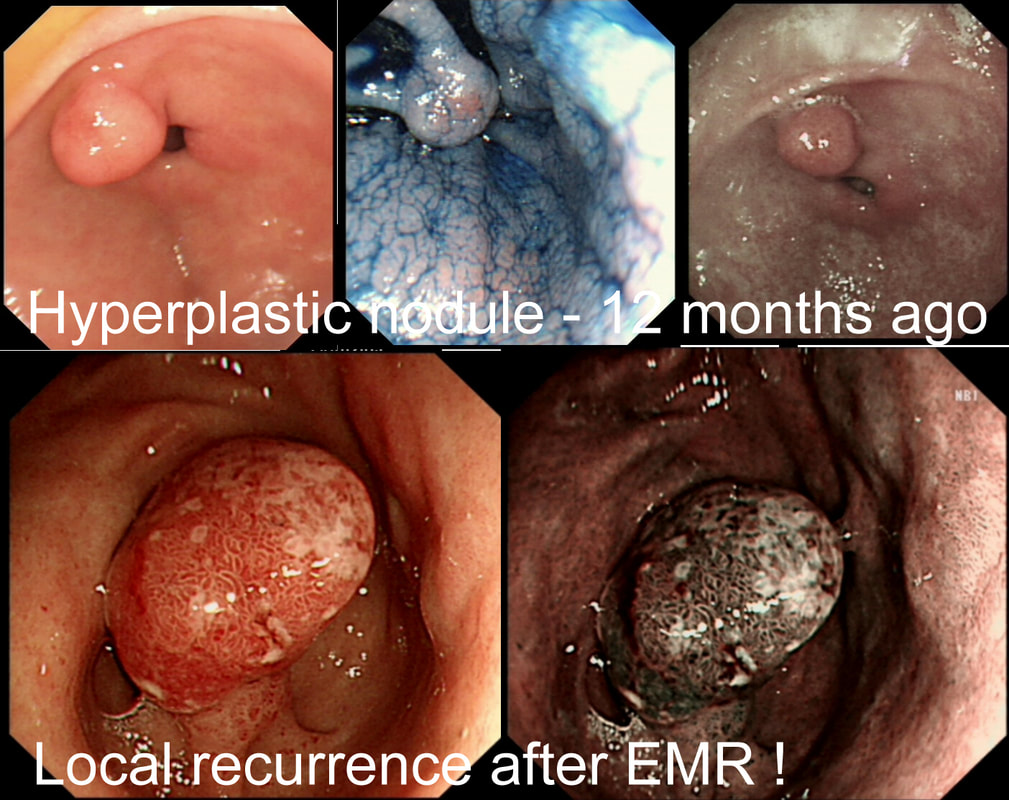
I removed this pre-pyloric nodule about a year ago. Histology confirmed an inflammatory/hyperplastic polyp without dysplasia. Now the thing has grown back!?
WHAT IS LIKELY TO BE THE APPROPRIATE MANAGEMENT?
■ Take a full set of biopsies
INCORRECT!
■ Remove the local recurrence by snare
INCORRECT
■ Eradicate the Helicobacters
CORRECT!
EXPLANATION
In most cases, gastric hyperplastic polyps are caused by Helicobacter pylori. However, because of the 0.6% - 2.1% risk of cancer arising within a hyperplastic polyp in the stomach, I do remove the larger ones. However, a year ago, this proved to be a hyperplastic polyp caused by Hp. The polyp recurred because the underlying cause had not been dealt with. This patient needs to have his Hp eradicated!!
A study published in Digestive Diseases & Sciences 2020;65(12):3652-59 confirmed that all 14 patients had complete regression of their hyperplastic gastric polyps 10 months after eradication of their Helicobacter’s compared with none of the 13 patients in the non-eradication group.
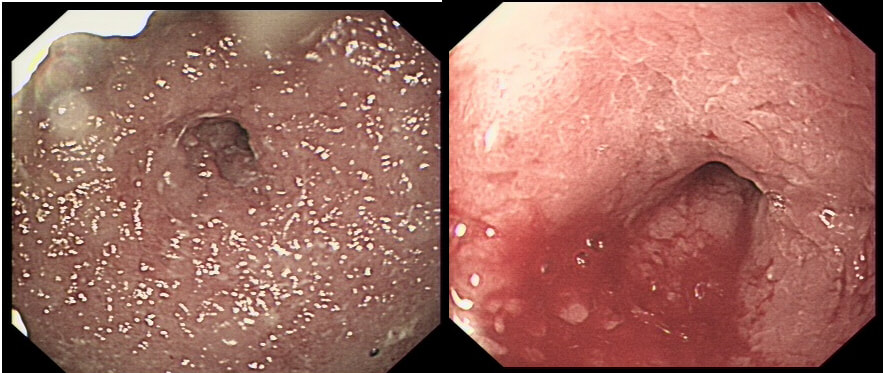
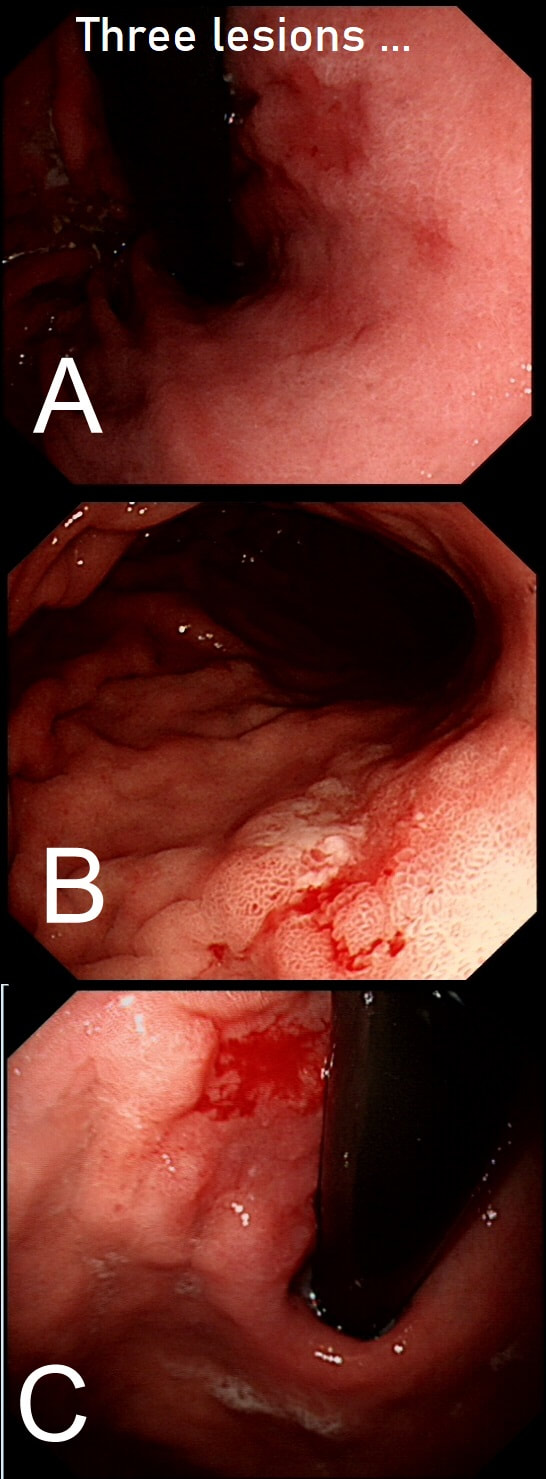
Three gastric lesions (A,B and C) in three separate patients all undergoing a gastroscopy because of iron deficiency anaemia
WHAT IS YOUR ENDOSCOPIC DIAGNOSIS?
■ BENIGN GU's
INCORRECT!
■ GASTRIC ADENOMAS
INCORRECT
■ EGC's
CORRECT!
EXPLANATION
These are all intramucosal gastric cancers. The typical scenario of an early gastric cancer is:
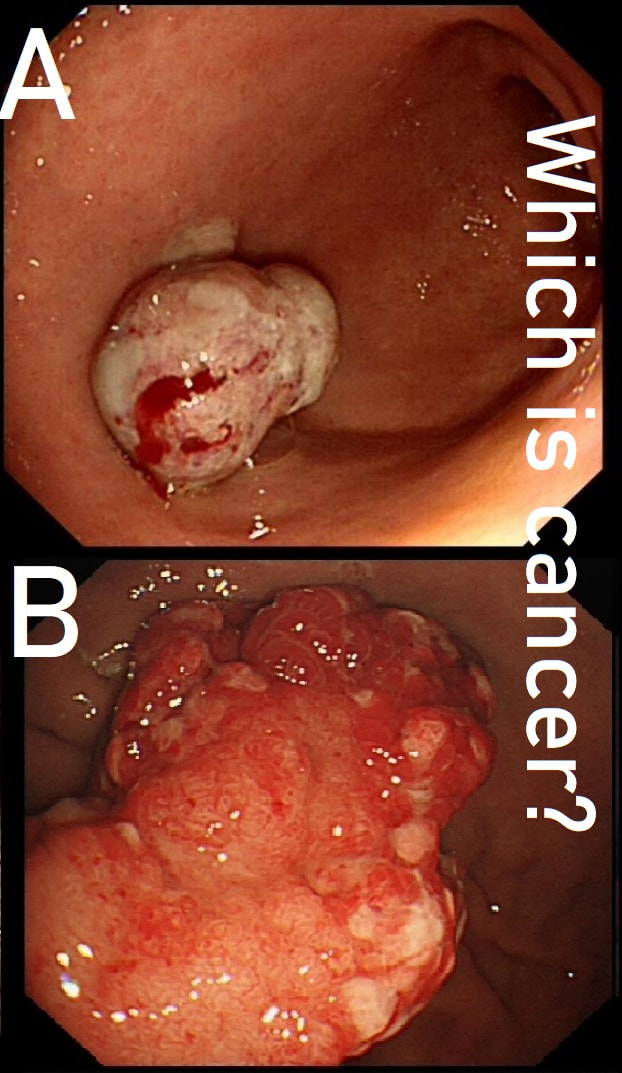
These two antral polyps look very different. That's because one is a polypoid cancer and the other is an innocent hyperplastic polyp.
WHICH IS WHICH?
■ A is malignant
Yes! A was poorly differentiated gastric carcinoma!
■ B is malignant
Surprisingly, B is actually the innocent hyperplastic polyp
explanation
Most (73%) thought that the hyperplastic polyp (polyp B) was the malignant one. Although large, the angry red colour and white spots are both strongly suggestive of a hyperplastic polyp.
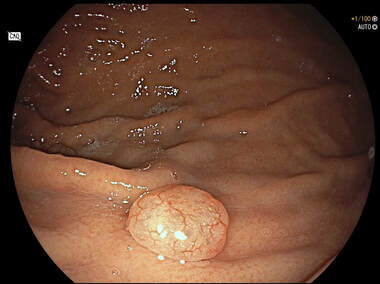
Interestingly, in both cases, the patient presented with anaemia. Of course, when the patient undergoes the endoscopy because of anaemia, any gastric polyp should be viewed with suspicion. Although the lack of surface crypts was disconcerting, as polyp A did not seem to be firmly attached, it was removed as a single fragment (of course). Histology confirmed that the lateral and deep margins were clear and there was no lymphovascular invasion (LVI). In the subsequent 'cancer meeting', it was pointed out that 'poor differentiation' may be considered an indication for surgery. This would indeed be the case elsewhere in the GI tract. However, in the stomach, poor differentiation 'on its own' (clear margins and no LVI) is 'allowed' provided that margins are clear, there is no invasion into the deepest layer of the submucosa and, most importantly, there is no LVI. Emerging data highlights LVI as THE MOST IMPORTANT predictive factor anywhere in the GI tract. The original ‘Japanese Standard Gastric EMR criteria’ gave the go-ahead for resection of well differentiated cancers up to 2cm provided that there was no surface ulceration. Nowadays, most of us would apply the 'Japanese Extended criteria’ which allows us to attack well differentiated EGC's up to 3cm in size even if there is some superficial ulceration of the surface. Pertinent to this case, the extended criteria also allows us to attempt to resect poorly differentiated cancers up to 2cm provided that there is no surface ulceration. This small polyp was found on the greater curve of a patient undergoing a gastroscopy because of indigestion. WHAT TYPE OF POLYP IS THIS? a) cystic fundic polyp b) hyperplastic polyp c) adenomatous polyp d) neuroendocrine tumour e) malignant polyp Explanation
Of course, this is a 'Fundic Gland Polyp' / 'Cystic Gland Polyp' (FGP). They are usually multiple, somewhat transparent sessile polyps, usually in the 1-5mm in diameter and located in the body and fundus. Histologically there are cystically dilated glands lined by gastric body type mucosa. Of course, there is no need to sample these. A study of patients reported that they can be diagnosed with a high degree of accuracy based on endoscopic appearance alone (J Clin Gastroenterol 2003;36:399-402). The sporadic polyps are caused by activating mutations of the beta-catenin gene. Not sure how an APC gene mutation (you may remember are involved in FAP), could give rise to FGP’s though !? I have seen a couple of cases of cancer developing within a FGP. In both cases the patient had FAP and the polyps were markedly larger than all the surrounding cystic fundic polyps. There have been case reports of dysplasia arising within sporadic fundic gland polyps. A few series have reported a <1% risk of sporadic FGPs harbouring dysplasia (Am J Surg Pathol 1998;22:293-298, Eur J Gastroenterol Hepatol 2003;15:1153-6, Endoscopy 27;32-37:1995). I must admit that I don't believe the figure of <1% risk of dysplasia in a sporadic FGP. I suspect that these series consist of rather selected cases. Of course by only selecting polyps 10mm or larger, you would be filtering away 99% of the FGP's, leaving only the 'far above average risk lesions' behind. Sporadic FGPs are not associated with either H.pylori gastritis or atrophic gastritis. In fact, it has been proposed that H. pylori infection may have an inhibitory effect on the development of FGPs. |
Categories
All
|