|
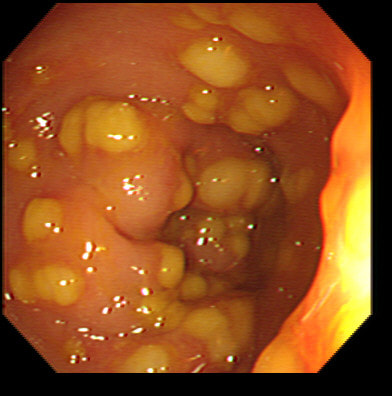
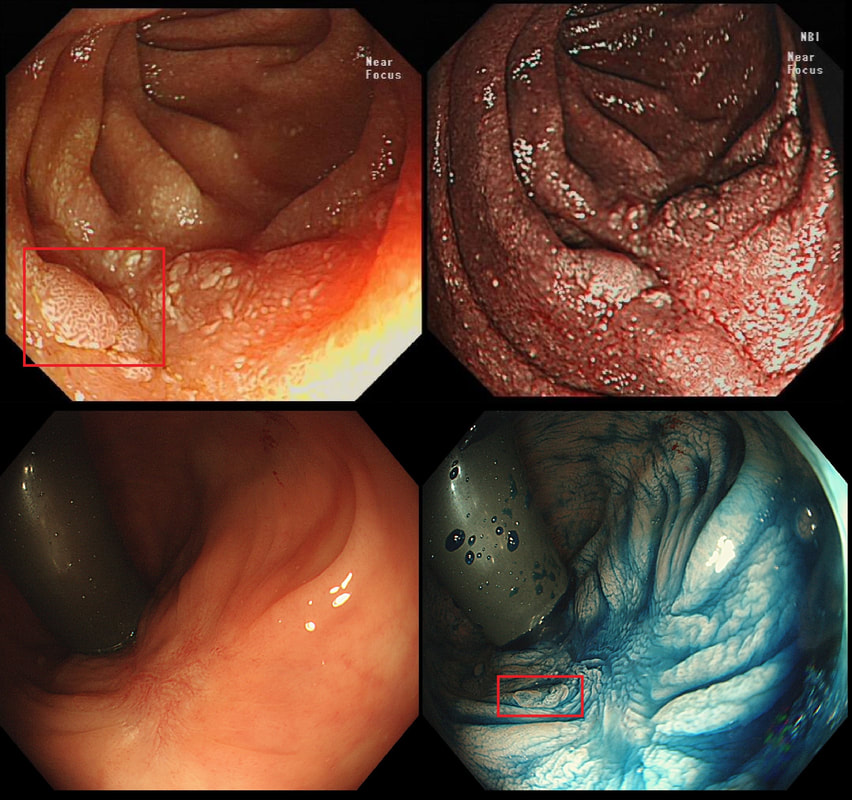
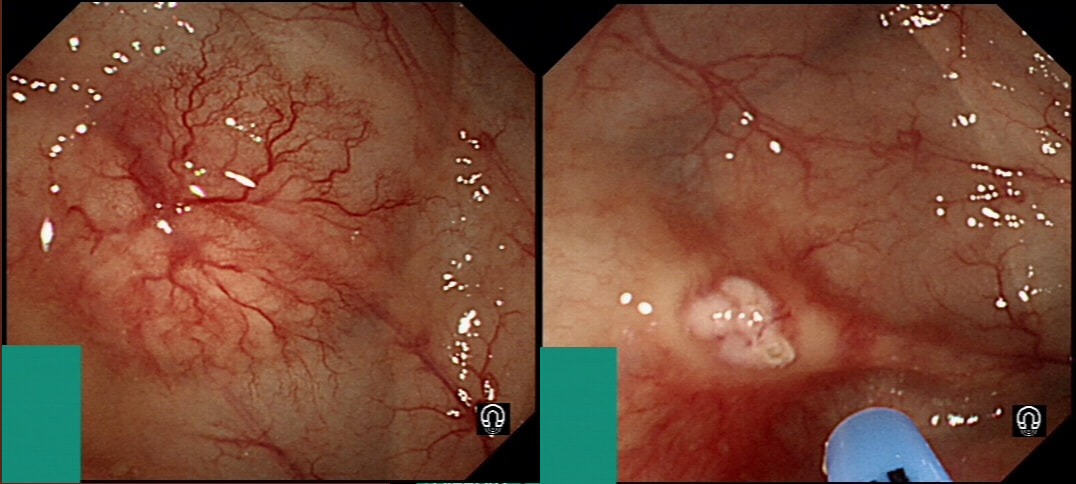
This is the sigmoid mucosa of an elderly inpatient who has developed diarrhoea.
WHAT SINGLE TEST WOULD YOU RECOMMEND NEXT?
■ FBC
Of course it's not sensitive nor specific and does not exclude diagnosis if normal. Nevertheless, a very high WBC level is often found in fulminant pseudomembranous colitis.
■ Obtain a set of stool cultures
Probably not a priority and they should have done it already!
■ ELISA for C.diff toxin in the stool
Yes! Results available within a few hours with sensitivity of 65%-85% and specificity 95%-100%
■ Stool PCR for C.diff
Seems like a sensible investigation in patients with unexplained, new-onset diarrhoea but perhaps not the single most important test...
■ Abdominal X-ray
Important IF there is significant abdominal distension
explanation
Of course this is a case of Clostridium difficule associated colitis (pseudomembranous colitis). Patients usually present with diarrhoea, abdominal pain, and leukocytosis, and a history of recent antibiotic use. Other common symptoms include fever, abdominal tenderness, and distension.
In a symptomatic patient with typical colonic pseudomembranes such as in this case, arguably it would be sensible to recommend that treatment is immediately started. Stool testing should be considered in any patient with unexplained, new-onset diarrhoea (defined as 3 or more unformed stools in 24 hours in a patient not taking laxatives). Your local institution will probably have protocols for how patients should be investigated and all of the above test may well be part of the algorithm. When I was on the wards, toxin tests were favored over culture for diagnosis of C.diff because it was the toxins which mediate disease. detection of toxins was faster and correlated better with symptoms. However, there was a move towards 'molecular tests' (PCR for the bacterium) from 2009 because of concern that patients with C.diff could be missed by toxin tests. Of course, this raised the question; do toxin-negative patients with a positive C difficile PCR test result require treatment? Several studies have now indicated that about half of the patients with positive PCR test for C. diff do not experience adverse events without treatment and do not need treatment. For this reason, PCR testing for C.diff should not be used as the stand-alone diagnostic test. Instead it's patients with clinical disease (diarrhoea) AND a positive toxin assay who should be treated! There are lots of references for this statement and here is an open access article in JAMA: Treatment is to discontinue the responsible antibiotic and start therapy with oral vancomycin or fidaxomicin. Up to 50% of patients have a relapse after discontinuation of antibiotics, but most respond to a second course of therapy. In those who relapse again, faecal microbiota transplantation is very effective.
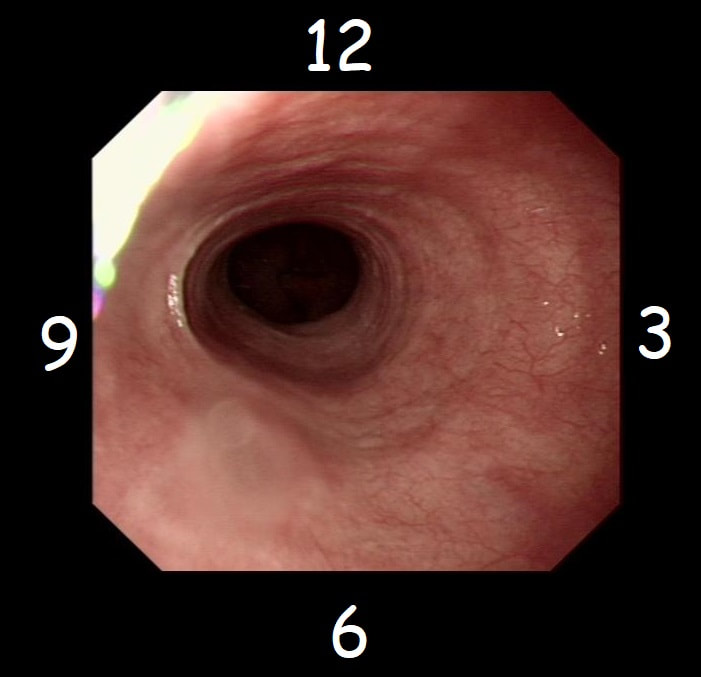
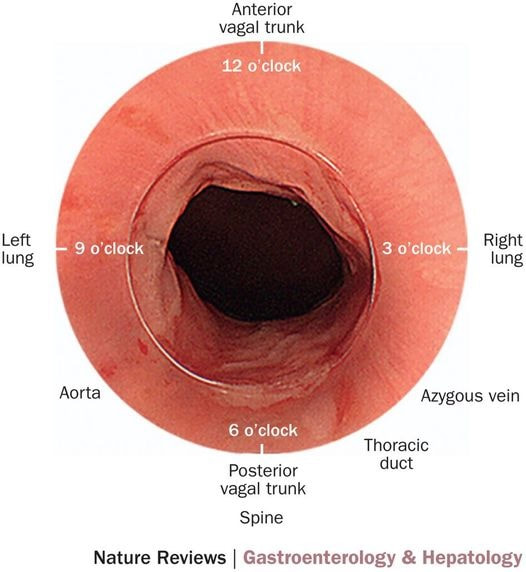
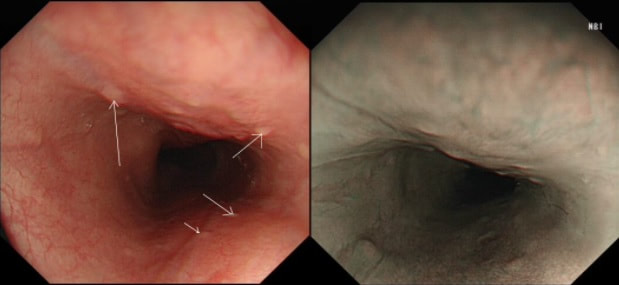
With POEM, knowing the landmarks outside of the oesophagus has become something which endoscopists are expected to be familiar with. Here is a quick self-test!
AT WHICH POSITION OF THE 'CLOCKFACE' DO YOU FIND THE:
■ Spine
6 o'clock
■ Anterior vagal trunk
12 o'clock
■ Posterior vagal trunk
6 o'clock
■ Thoracic duct
5 o'clock
■ Left lung
9 o'clock
■ Aorta
7-8 o'clock
■ Azygous vein
4 o'clock - runs in the posterior mediastinum, on the right side of thoracic vertebral column. It drains into the SVC, and returns blood from the posterior walls of the thorax and the abdomen.
This lesion was found at the top end of the colon. WHAT WOULD YOU DO NEXT?
■ Lesion is benign but it can't be removed endoscopically
Best to admit defeat than to fight against overwhelming odds
■ Lesion is benign and I will organise an EMR
Not sure that anyone could remove this endoscopically
■ Lesion is benign and I will organise an ESD
Ridiculous!
■ Lesion is benign and I will organise an underwater resection
It will also fail !
■ Lesion is malignant and requires a surgical resection
Polyp is large BUT looks benign!
explanation
This is a classical LST-G (laterally spreading tumour of the granular type). These lesions are always TVA's and almost always harbour LGD. Unfortunately, there are three problems which precluded an endoscopic resection; 1) there is a crazy alpha loop in the transverse colon which as usual proved impossible to remove, 2) the movement of the diaphragm continuously moves the lesion away and towards you and 3) the main part of the lesion is in the ascending colon but then extends across to the caecum where it almost kiss the appendix orifice. The moral of the story is to carefully consider BEFORE you start the resection. After you have started to attempt resecting a lesion such as this it becomes progressively more difficult to stop! It's like a gambler finding it increasingly difficult to walk away from the table as losses stack up. Because you have invested so much time and effort, it becomes difficult to stop and admit defeat! This was found in the sigmoid of a 35 year old lady presenting with abdominal pain and bleeding. She has been taking ibuprofen for her pain. WHAT IS THE MOST LIKELY DIAGNOSIS?
■ NSAID induced colitis
Possible but the pain came before starting the ibuprofen
■ Ulcerative colitis
Stricturing and linerar ulcers are not typical for UC
■ Crohn's disease
Most likely and don't forget to add your endoscopic diagnosis to the histology request form!
■ Diverticular stricture
Those rarely ulcerate and are usually MUCH tighter
■ Malignant stricture
Those post-inflammatory polyps are innocent!
explanation
The superficial, linear ulcers are typical of inflammatory bowel disease. Both ulcerative colitis and Crohn's disease can cause stricturing but of course Crohn's is more likely to. Those little nodules are post-inflammatory polyps, covered in normal crypts. In any case of colitis it's important to take a full set of samples, targeting not only areas which are visibly inflamed but also non-inflamed areas. Patchy inflammation is very suggestive of Crohn's disease but if you neglect to sample the non-inflamed areas, you will find that your pathologists will report a continuous colitis which would be more suggestive of ulcerative colitis. After all, they can only report on the samples given to them and may well not read your endoscopy report. This 75 year old man underwent a CTC because of a change in bowel habit which has confirmed a large caecal polyp. The patient has been referred for a snare polypectomy. WHAT WOULD YOU DO NEXT?
■ Abort the procedure and reassure the patient
Absolutely!
■ Take a full set of samples
Uneccessary and they will only show normal mucosa
■ Remove the polyp by EMR
Foolish!
■ Remove the polyp by ESD
Idiotic!!!
explanation
The location and the 'pillow sign' tells you that this lesion is a lipoma. We know from Shrisha Hebbar's Podcast that the ICV is a high risk location for any attempt at removing lipomas. He mentions a patient of his who presented with acute small bowel obstruction due to engorgement of an ICV lipoma after the placement of an Olympus ligation.
Furthermore, you wouldn't of course go ahead with an attempt at resecting a lipoma without due consideration of indications. How long has the patient had symptoms? Are they getting better? Are symptoms likely to arise because of this soft and floppy lesion. Probably unlikely and statistically speaking, this elderly patient is probably far more likely to have a change in bowel habit due to constipation. By the way, please take notice of the odd looking mucosal crypt pattern which is reminiscent of a TVA! This is the normal crypt pattern at the ICV at the transition to the villous surface in the terminal ileum! This video clip requires no introduction. No other abnormality was found. WHERE IS THE LIKELY SITE OF THE PROBLEM?
■ in the portal vein
Then you would expect BOTH oesophageal and fundal varices!
■ In the hepatic vein
In Budd-Chiary, there are no varices!
■ In the splenic vein
Isolated splenic vein varices - absolutely correct!
explanation
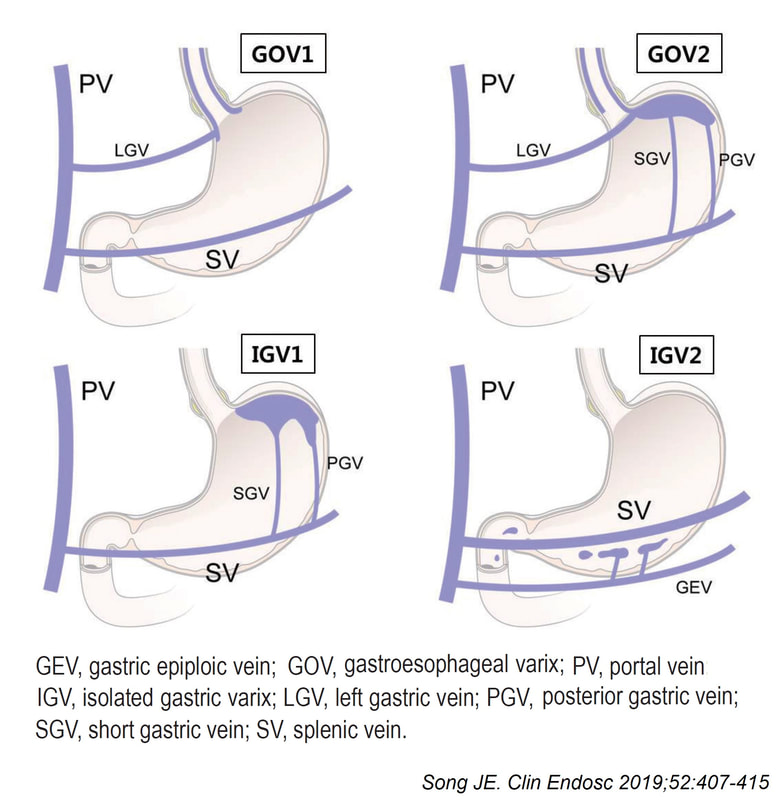
Quoting the 'Sarin Classification' of gastric varices, this is a case of an 'isolated fundal varix' (IGV1). In this case the underlying cause was splenic vein thrombosis following an admission for acute on chronic pancreatitis. The precise distribution of the varices helps you to pinpoint the location of the thrombus. The infographic below from Song et.al is brilliant:
Briefly, 1) oesophageal varices with or without fundal varices points to portal vein thrombosis 2) fundal varices ALONE points to splenic vein thrombosis 3) antral and/or duodenal varices points to a thrombus in the gastro-epiploic vein Brilliant !
This was an incidental finding in the mid-oesophagus. Subsequently an EUS is organised and the lesion is ultimately removed (histology in the last image)
WHAT IS THE DIAGNOSIS?
■ Neuroendocrine tumour
I've never actually seen an oesophageal NET
■ Granular cell tumour
But it isn't a small white submucosal disc?!
■ Leiomyoma
A shrewd guess!
■ GIST
These are VERY rare in the oesophagus (but common in stomach)
■ Melanoma
Usually look brownish endoscopically!
explanation
Leiomyomas are the most common submucosal lesion found in the oesophagus. No idea why they are so rare in the stomach, where GIST's are the most common submucosal lesion. Correspondingly GIST's are very rare in the oesophagus.
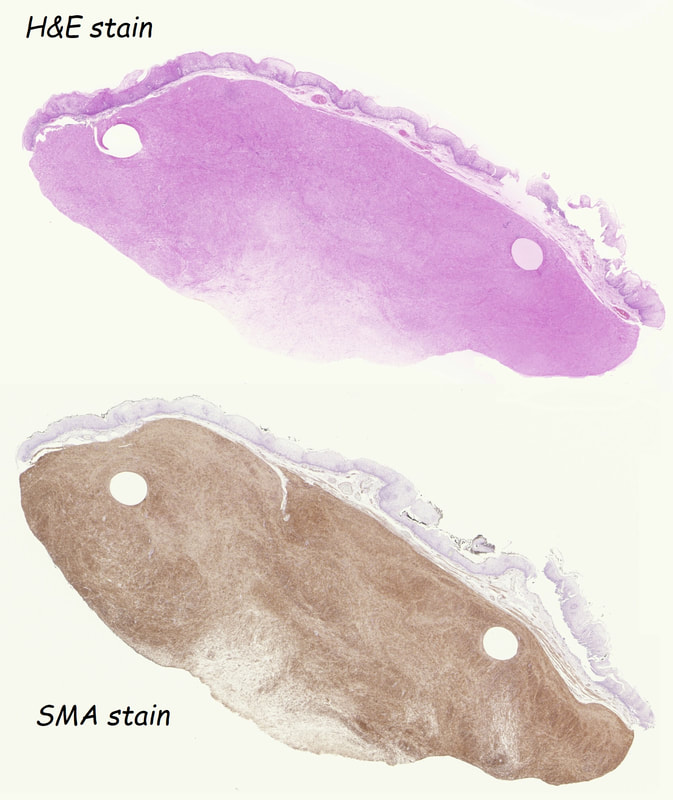
These usually arise from the circular muscle propria layer. On CT they are often calcified and there are of course no nearby nodes to suggest malignant disease. Indeed, malignant transformation is extremely rare and for this reason many centres would ignore a small submucosal lesion such as this, when found in an elderly person. Larger (around 5cm usually) and symptomatic lesions (which are more likely to be leiomyosarcomas) are enucleated surgically. "- They shell out like peas" according to one of our UGI surgeons. However, our local surgeons are also careful to point out that resection usually result in some damage to the muscle propria layer, which require repair as otherwise a diverticulum may develop. With endoscopists spending more time in the 'third oesophageal space' (i.e. the submucosal space), doing ESD's and POEM, the suggestion that we should also remove leiomyomas endoscopically has arisen. May seem like a good idea but I would advocate caution for the simple reason that endoscopically, we are unable to repair the defect we cause in the muscle propria layer ... Oh, how about that 'SMA stain' used in the histology. This is 'Smooth Muscle Actin' stain which does exactly what it says on the tin ...
This patient originally presented with a variceal bleed and was managed on ITU. Investigations confirmed alcoholic liver disease and the patient is entered onto a variceal ablation programme. At the first outpatient endoscopy, this is noted
WHAT IS THE LIKELY DIAGNOSIS?
■ Vocal cord granuloma
Looks like granulomatous tissue!
■ Mucus retention cyst
It doesn't look cystic !
■ SCC
Unlikely as wasn't noted at intubation
explanation
This is a vocal cord granuloma likely caused by irritation from the ET tube during the recent admission. Other causes include 'voice abuse', severe cough, vomiting, habitual throat cleaning, and the use of irritants such as tobacco and alcohol. Mucus cysts rarely develop on a vocal cord and look cystic rather than as granulomatous tissue as in this case.
Initially, management is supportive as these often shrink within 6-12 weeks. Surgery or laser ablation is reserved for large lesions but is a little fraught is these tend to recur unless to underlying cause is addressed. Singers should be advised to warm up the voice before singing (or speaking), resting the voice in anticipation of future speaking obligations and to avoid singing at times of an upper respiratory infection. This excellent photograph of a vocal cord granuloma was actually supplied by Dr Muslim Alkafaji who has been a member of Friends of Endoscopy since 2014 !!!
I found this in the low rectum of an elderly lady undergoing a flexible sigmoidoscopy because of rectal bleeding. A quick check with the patient confirmed my suspicions.
WHAT IS THE DIAGNOSIS?
■ Rectal prolapse
The patient would know about that!
■ Solitary rectal ulcer syndrome
A good guess and patient would know about constipation and straining but actually not the correct answer
■ Likely serrated polyp syndrome
Hmm, what question would you ask patient? FH of CRC perhaps?
■ Likely FAP
Findings of a rectal pouch in FAP? No, that's not it!
explanation
On first glance this looks like a circumferential serrated polyp in the rectum. Of course, this would be somewhat odd and furthermore, unlikely that the patient would know anything about this.
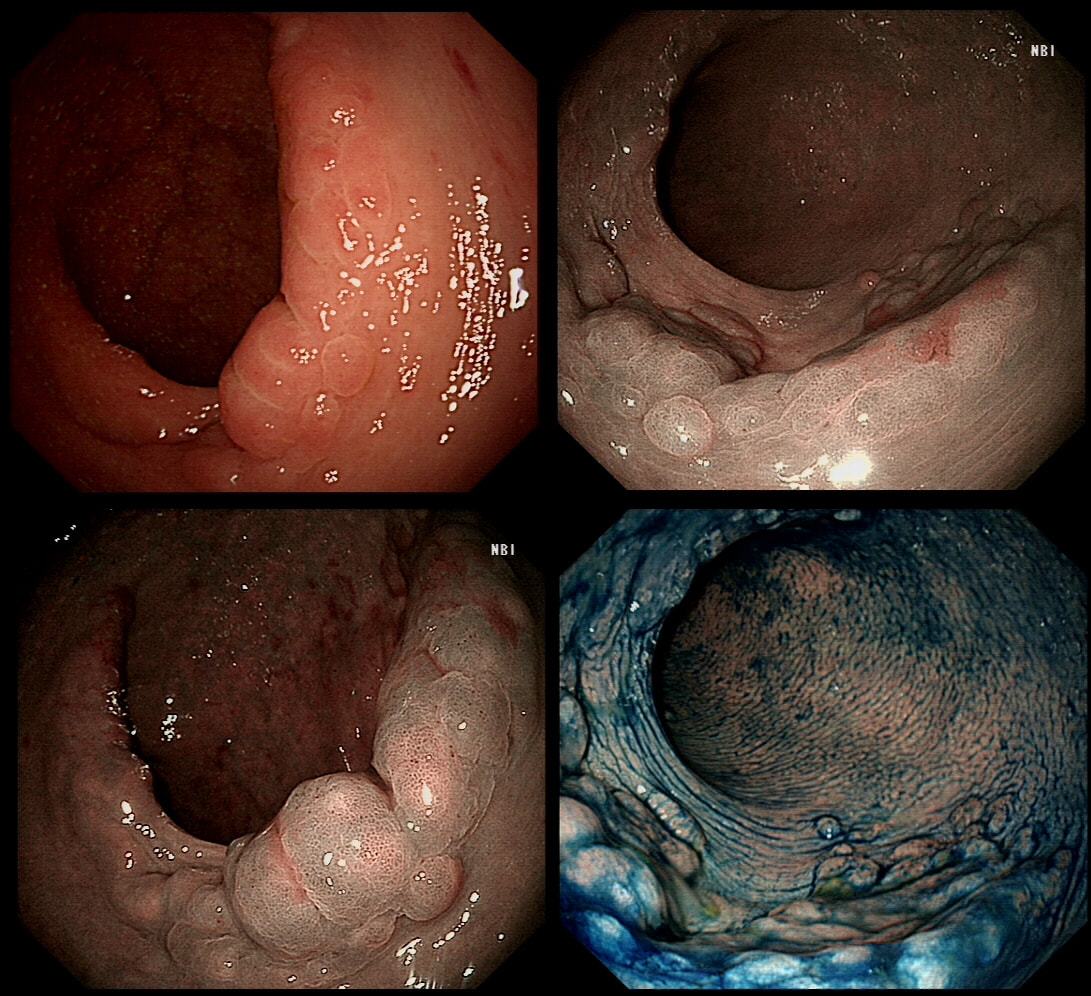
Actually, on direct questioning she did know about her prolapse! Another common endoscopic finding is an erythematous and oedematous patch, close to the anal margin. Just a bit of fun! Above are four types of benign polyps which can be found in the stomach, duodenum and colon respectively. I've used a 'plugin' which will hopefully make it easier to see the polyps on a small screen. BUT WHAT ARE THESE FOUR POLYPS ? A = B = C = D = You'll have to make up your mind and then click 'Explanation' below to reveal the correct answers ! EXPLANATION
The 3 polyps in column A are all adenomatous polyps. Flat and plaque-like in the stomach, pale in the duodenum and a sessile TVA in the colon. Column B shows NET's with the prominent vessels along it's side in the stomach and colon whilst the duodenal NET has the typical central depression. Column C shows 'hamartomatous' polyps seen in Peutz-Jeghers syndrome. There is hardly ever any dysplasia within these polyp and it's uncertain what the 'precursor lesion' is for these patients GI cancers Column D show inflammatory polyps which are typically angry red and sometimes with pretty white spots ! This patient has been referred for a dilatation WHAT IS THE AETIOLOGY OF THE STRICTURE?
■ Peptic stricture
Too membrane-like to be peptic
■ Schatzki ring
Of course it is - Well Done!
■ Eosinophilic oesophagitis
Strictures in EoE are long and tapered
■ Radiotherapy stricture
RXT strictures are at least a couple of cm in length
■ Malignant stricture
Of course it isn't !
explanation
This short almost membrane-like stricture is of course a Schatzki ring, named after the German-American physician Dr Richard Schatzki. It's usually easy to treat these with a balloon dilatation. This particular case is unusual as two dilatations were required. However, rather than going ahead with a second dilatation to 20mm (our largest balloon in Leeds), I decided to cut the membrane with the tip of a snare and the diathermy set on the normal sphincterotomy setting.
The uploaded video is a good example of this mishap when cutting across yourself. I don't think that I perforated the full thickness of the oesophagus but applied a clip just in case (I've never regretted placing too many clips).
These are the NBI images of a young woman complaining of chest pain, dysphagia and reflux symptoms. She also complains of feeling of a 'lump at the back of her throat'.
WHAT WAS THE ORIGINAL DIAGNOSIS?
■ Reflux oesophagitis
Would be an odd pattern surely?
■ Barrett's oesophagus
With squamous mucosa below?
■ Eosinophilic oesophagitis
EoE doesn't look brown on NBI !
■ Oesophageal inlet patch
Yes, but why is it getting smaller?
■ Squamous dysplasia
No dilated IPCL's !!!
explanation
This patient was found to have a large oesophageal inlet patch at the index endoscopy a year ago. Although his symptoms were rather mixed, we have spent 4 sessions on ablating her inlet patch with APC.
Of course, how often you find these will depend on whether you use NBI/FICE/BLE during extubation. Of course you should do because you are then less likely to miss squamous dysplasia! An interesting study reported that gastric inlet patches contained glucagon cells which would normally only be found in the embryo and subsequently disappear. To remind you, the hormone glucagon is normally produced by the alpha cells of the pancreas! Glucagon-like peptide-1 is also secreted by some cells in the ileum and colon. Anyway, the implication is that the gastric inlet mucosa is actually an embryonic left-over. Interestingly, there appears to be a lower prevalence reported in older populations, and some authors suggest that inlet patches may regress with age! Seems odd for a congenital condition? Endoscopic studies have found inlet patches in up to 10% of patients. Whether these give rise to any symptoms is contentious. It has been suggested that inlet patches may give rise to globus sensation due to irritation of the upper oesophageal sphincter by acid or mucus. Furthermore, others have suggested that H. pylori colonization of the heterotopic gastric mucosa causes symptoms. Others believe that it's not just the upper oesophageal sphincter which is abnormal but the entire oesophageal motility may be disorganised in these patients. However, prospective studies have usually not found that patients in whom an inlet patch is found, present with any different symptoms compared to patient without an inlet patch. Of course as anywhere in the body, cancer may develop. A study in Digestive Diseases & Sciences [2018;63(2):452-65] reported on 156,236 gastroscopies and may have found 3 cases of malignant conversion. Assuming that 10% of their patients had an inlet patch, that would suggest that you would have to regularly surveil 500 patients with an inlet patch over many years detect 1 cancer. If you grab 500 random people off a street and scope them annually for 5 years, wouldn't you pick up more than a single early oesophageal cancer? Seems like that lemon is not worth the squeeze? Finally, how about our patient? Over the period of one year, we spent about one hour ablating and completely getting rid of that large inlet patch (well two inlet patches really). The patient reported that symptoms were much better for a couple of months but then returned as before. Of course as she no longer has an inlet patch, we can now conclude that it never did contribute to her symptoms. Must admit that I was sceptical from the start! Show me a prospective RCT with evidence that ablation provides a clinically significant improvement in patients globus symptoms for more than a few months! Until I see that RCT, I will not be offering ablation for these patients. This lesion was found in the ascending colon. WHAT IS THE MOST LIKELY DIAGNOSIS?
■ Lipoma
Hmm looks too translucent
■ Lymphangioectatic cyst
Well it looks cystic doesn't it!!!
■ GIST
I predict that the pillow sign will be positive
■ Serrated polyp
Doesn't look like a polyp
■ Adenomatous polyp
But the crypt pattern is normal !!!
explanation
The apparent lesion is covered with a normal layer of mucosa. In addition, it seems very soft. However, the lesion looks more translucent than yellow! Of course, it's another lymphangioectatic cyst ! Naturally, you confirm this by giving it a prod with your biopsy forceps. If soft, its either a lipoma or a cyst. If firm, it's either a GIST, leiomyoma or a metastasis (I guess).
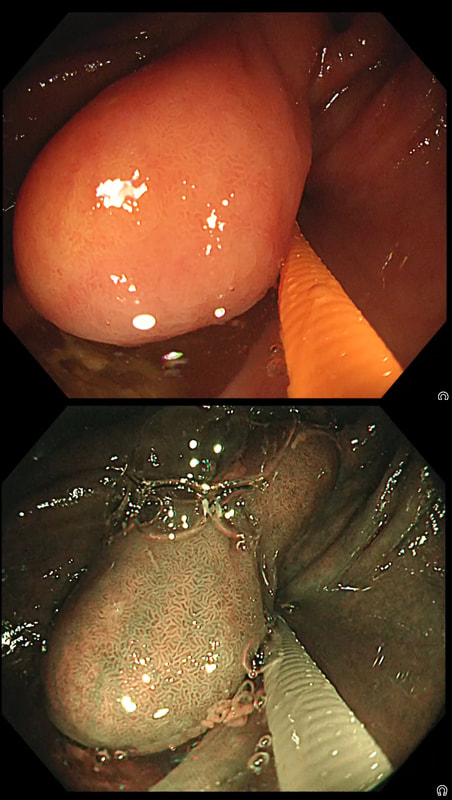
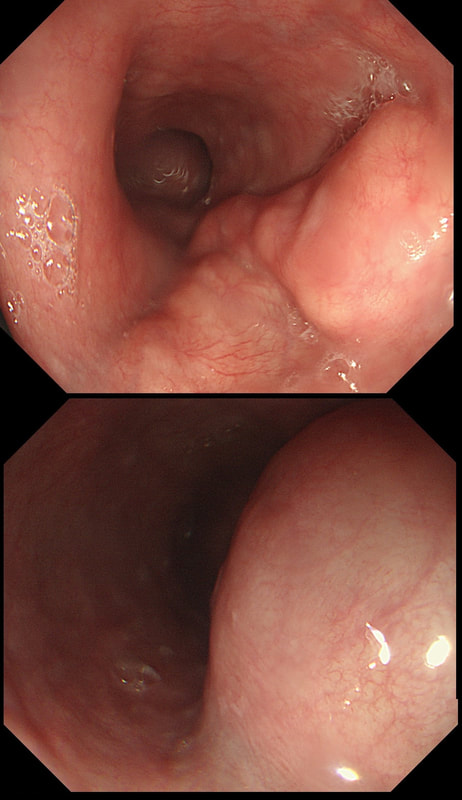
This lady is undergoing a gastroscopy
WHAT IS THE LIKELY DIAGNOSIS?
■ Cirrhosis and portal hypertension
Look again!
■ Splenic vein thrombosis
Gives you gastric varices!
■ Hepatic vein thrombosis
Pts present with ascites and normal LFT's!
■ SVC obstruction
Too easy perhaps?
explanation
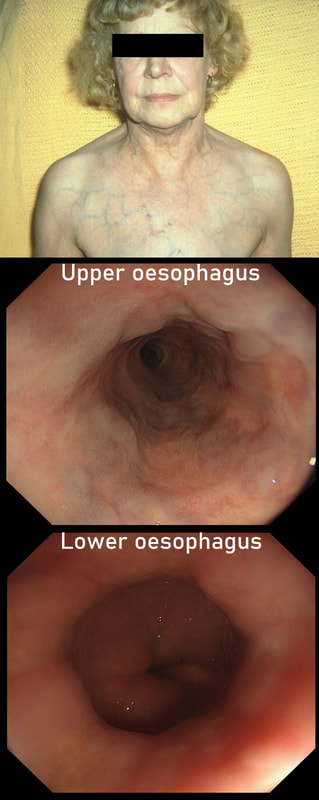
‘Downhill oesophageal varices’ are associated with superior vena cava (SVC) obstruction or obstruction or as in this particular endoscopic case, a clot in the right brachiocephalic, subclavian and axillary veins. I thought that just showing the oesophagus would make the question too difficult and therefore added the photo of a patient with SVC obstruction. Perhaps this made it too easy ☺!
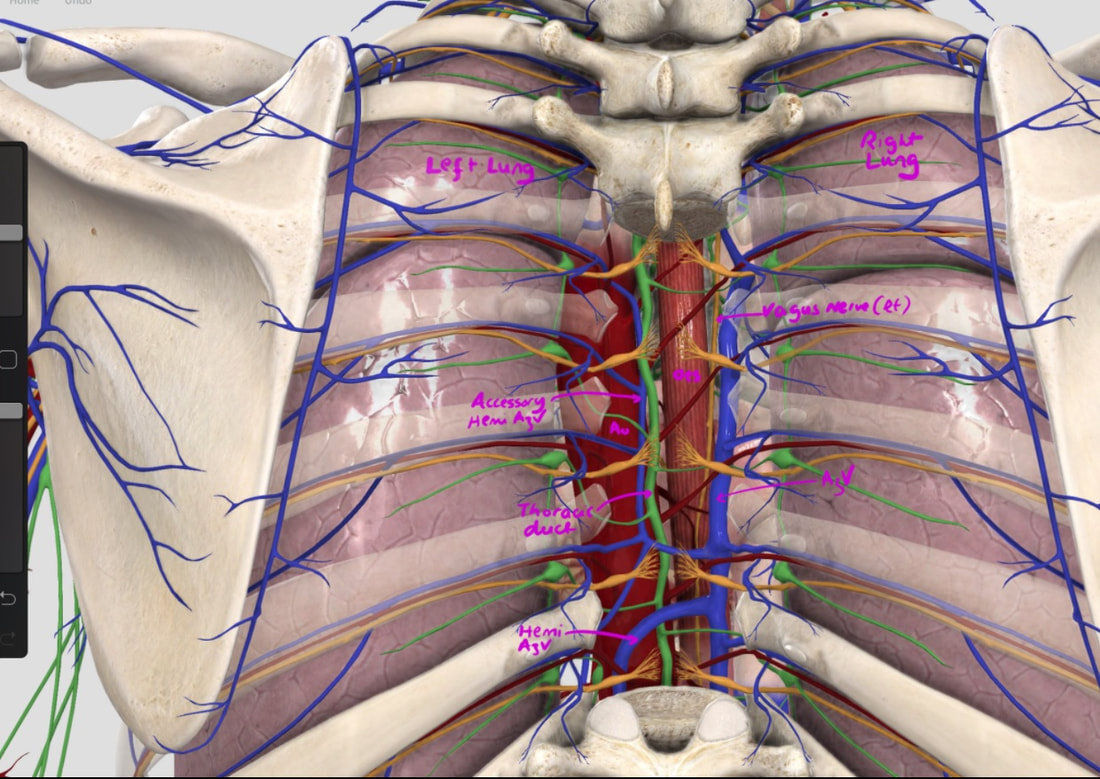
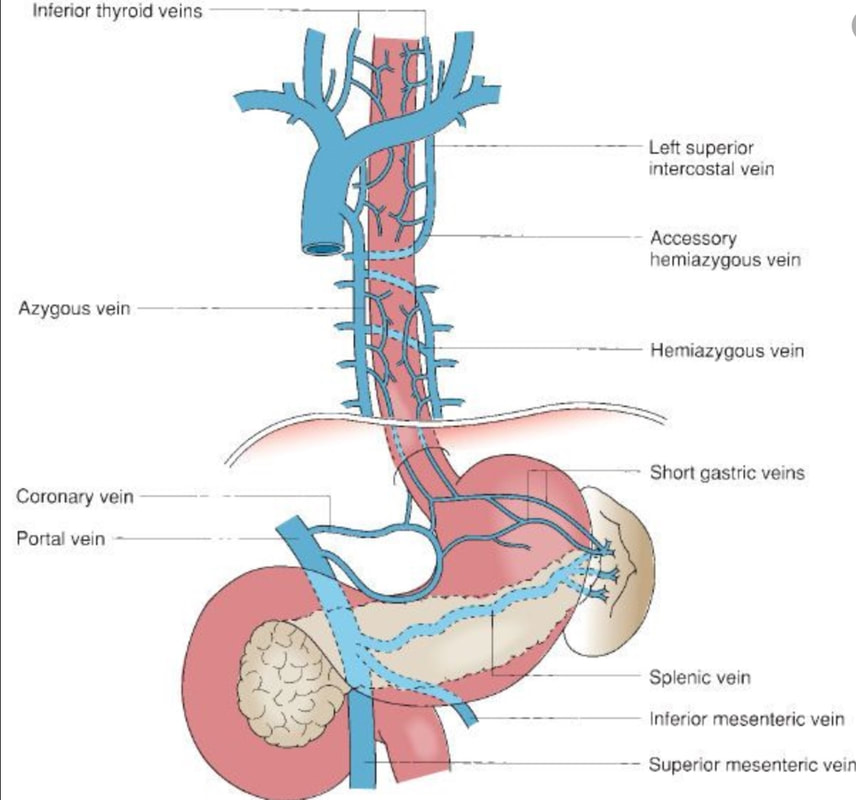
Venous drainage of the oesophagus is through the azygos and hemiazygos veins (see below). When there is thrombus, retrograde flow pushes blood towards the heart via collateral channels. If the obstruction is above the level of the azygous vein, varices develop in the upper oesophagus. However, if the clot is below, or involves the azygous vein, varices will form along the entire oesophagus. Bleeding is rare as varices in the proximal oesophagus are situated deeper within the submucosa than distal varices. However, if you are asked to deal with bleeding, you need to place bands at the proximal end of the varix. Sclerotherapy is contraindicated in the high oesophagus because of the risk of spinal cord infarction and vertebral infarction. Glue injection should also be avoided as there has been a case report of a massive PE.
This was polyp found on the greater gastric curve.
WHAT IS THE LIKELY DIAGNOSIS?
■ Hyperplastic polyp
No! Those look like mushrooms usually
■ Fundic gland polyp
No, those look like frogspawn when you look closely
■ Adenomatous polyp
Absolutely, to be specific an 'intestinal type adenoma'!
■ Early gastric cancer
It still has a crypt pattern!
■ Invasive gastric cancer
Impossible as it still has a crypt pattern!
explanation
Clearly there is a discrete nodule containing some small round crypt openings. If a dysplastic (mutated and genetically unstable) lesion produces a visible lesion, the pathologist calls it an 'adenoma'. Dysplasia in a flat mucosa is just called 'gastric dysplasia'. Of course in the colon there is a long established 'adenoma → carcinoma' sequence first proposed by Basil Morson a famous British pathologist. He estimated that about 2/3 colorectal cancer must be arising from an adenomatous precursor. What gives rise to the rest remained a disputed mystery until Jeremy Jass, another famous pathologists (and I really nice guy as I found, meeting him at a conference) proposed that serrated polyps may also give rise to cancer [Histopathology 2006;49:121-31]. His suggestion and data to support it, didn't really go anywhere for some 10 years but after he died in 2008 of glioma, the 'serrated pathway' to bowel cancer has become accepted.
Anyway, in the stomach things are even more complicated! There are at least 4 types of adenomas:
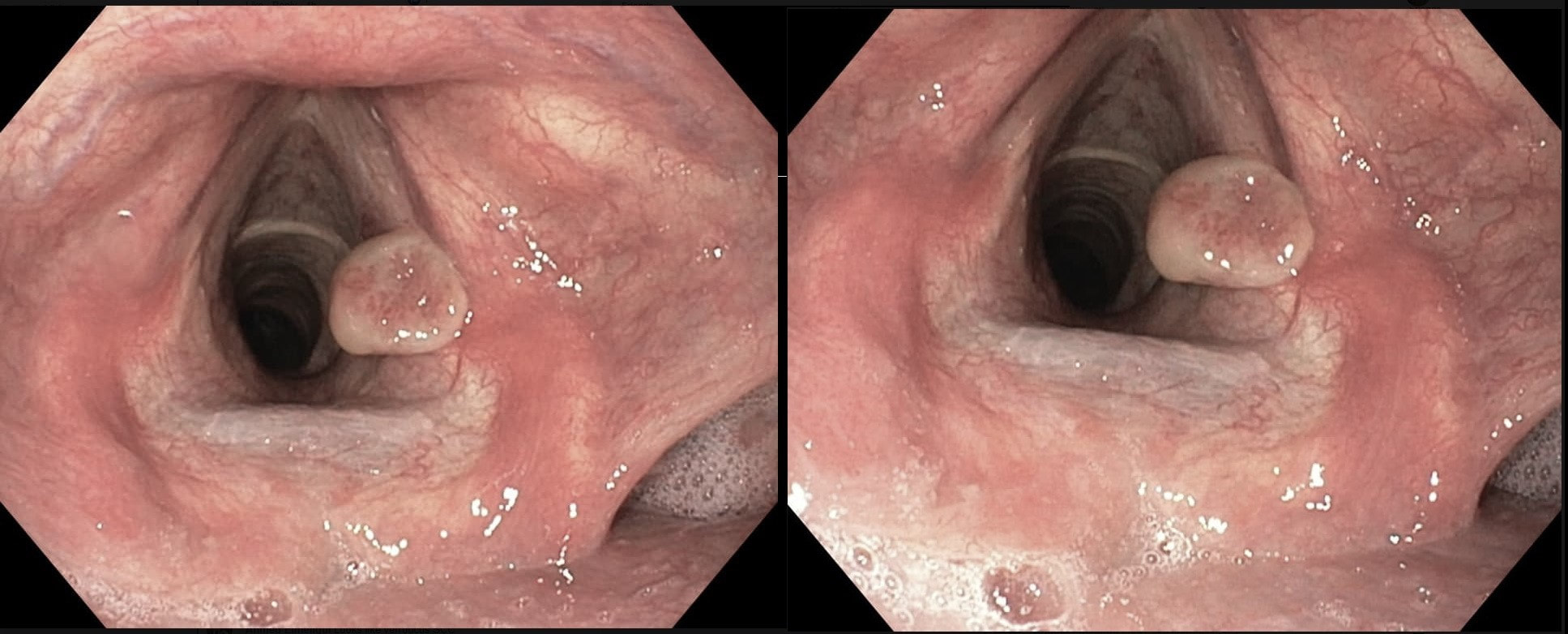
Of course, not every gastric polyp is adenomatous. Most are hyperplastic and look angrily red (because they have lots of capillaries) and often with white fibrin caps, making them look a little like mushrooms and may have a villiform surface reminisient of TVA's in the colon. Another common type are the 'Fundic gland polyps' which are full of cystic spaces and therefore look a little like frog-spawn. These usually develop in patients on PPI's but also in FAP. Polyps may also be hamartomatous as in 'Peutz-Jeghers syndrome', 'Cronkhite-Canada syndrome' and 'Juvenile polyposis'. By the way, the word 'Hamartomatous' implies that the polyp has arisen from overgrowth of multiple aberrant cell lineages rather than from a single, mutated precursor cell as in an adenoma. And then of course there are neuroendocrine tumours (NET's). A whole different thing which has a complete podcast dedicated to it. You can tell that a gastric polyp is a likely NET as they usually have little vessels crawling up it's sides. Finally, the lesion may be a gastric metastasis most commonly from breast, kidney, melanoma or HCC. By the way, there is a pitfall here as 'lobular breast cancer' mimics diffuse type gastric cancer. For this reason you should inform the pathologist if the patient has a history of breast cancer and you suspect a metastasis! This was an incidental finding in the pharynx WHAT IS THE DIAGNOSIS?
■ Papilloma
Too large probably
■ Retention cyst
Absolutely right!
■ Small SCC
Smooth, round and without dilated IPCL's !?!
explanation
This is a 'Pharyngeal retention cyst'. It was an incidental finding and the patient had no symptoms relating to this. These are the most common benign lesions in the pharynx, usually arising behind the epiglottis (the area is called the valleculae) or more to the side as here. The cysts are thought to result from dilatation of mucus glands secondary to retained secretions and chronic inflammation. They are usually incidental findings although larger cysts (>1 cm) may cause dysphagia, coughing or dysphonia. Complications include ulceration and secondary infections.
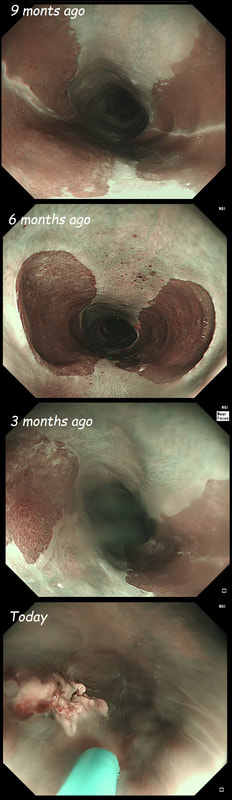
Two EMR sites. The top in the duodenum and the bottom in the rectum
ARE THE SCARS 'CLEAN'?
■ Both sites look OK
No they don't!
■ Local recurrence in duodenum
You are half correct!
■ Local recurrence in duodenum
You are half right !
explanation
The teaching point behind these two images is the simple message that it's impossible to see a local recurrence with just 'white light'. You must assess the scars with NBI or dye sprye or both (I do)! In addition, remember that most local recurrences are to be find at the most 'difficult to see edge'. Perhaps you can only see a local recurrence in retrovertion?
In these particular cases both have a local recurrence (see below).
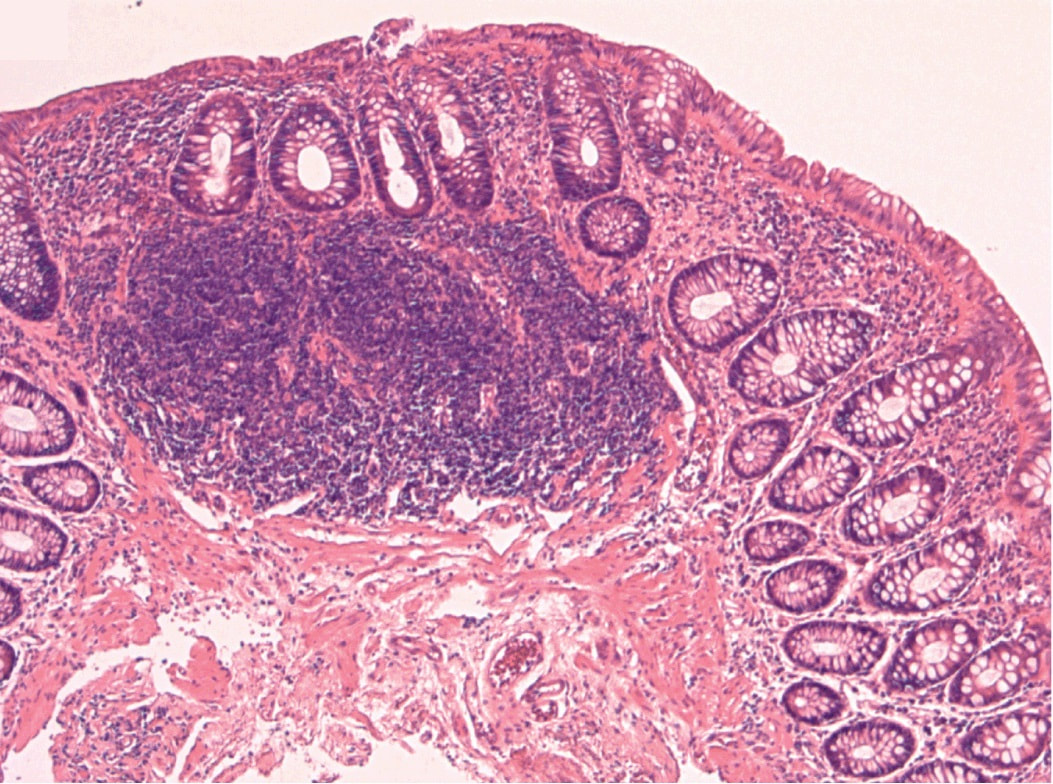
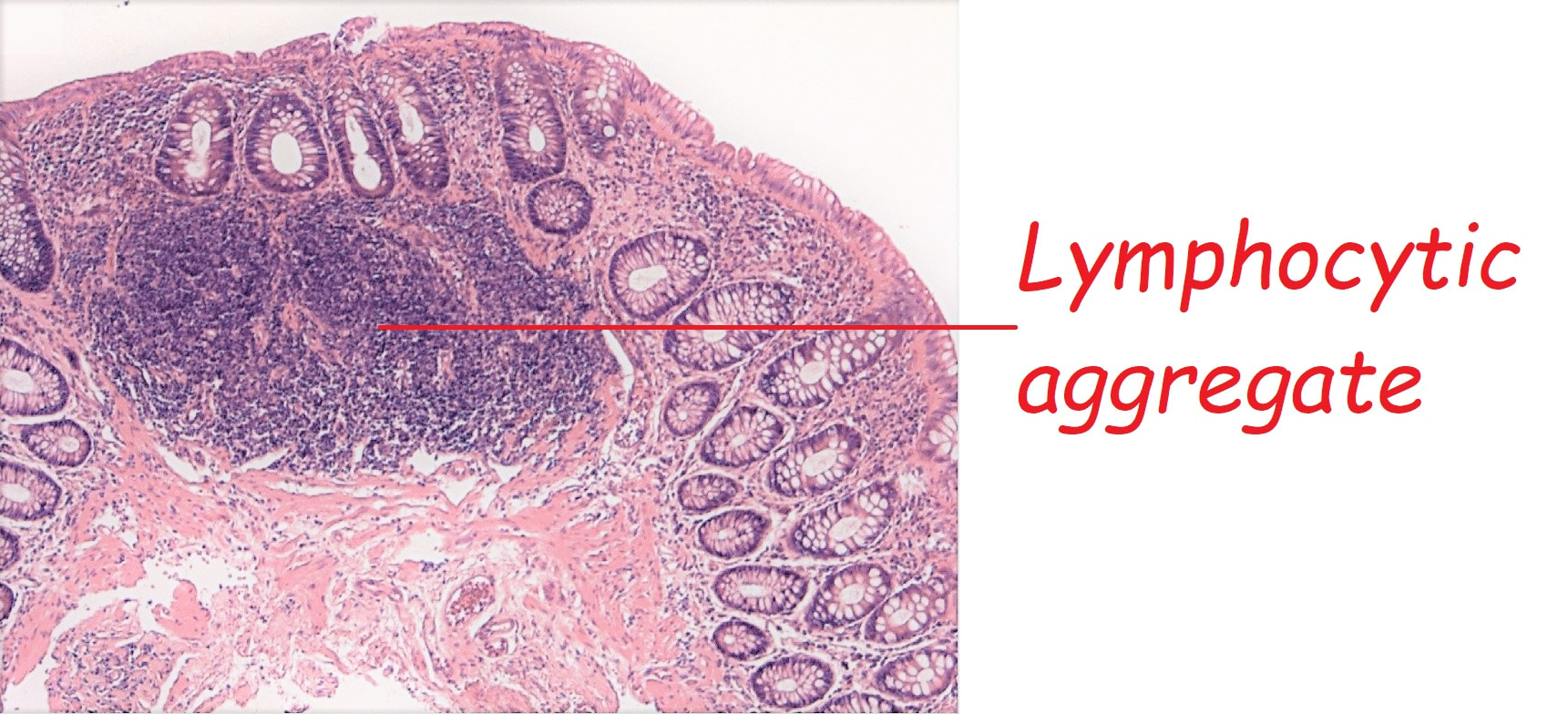
These small spots are a common finding within the oesophagus but
WHAT ARE THEY?
■ Glycogenic acanthosis
Good guess but you haven't understood the histology
■ Aggregates of lymphocytes
Absolutely!
■ Xanthelasmata
Only found in the oesophagus!
■ Hyperkeratosis
Looks different remember?
explanation
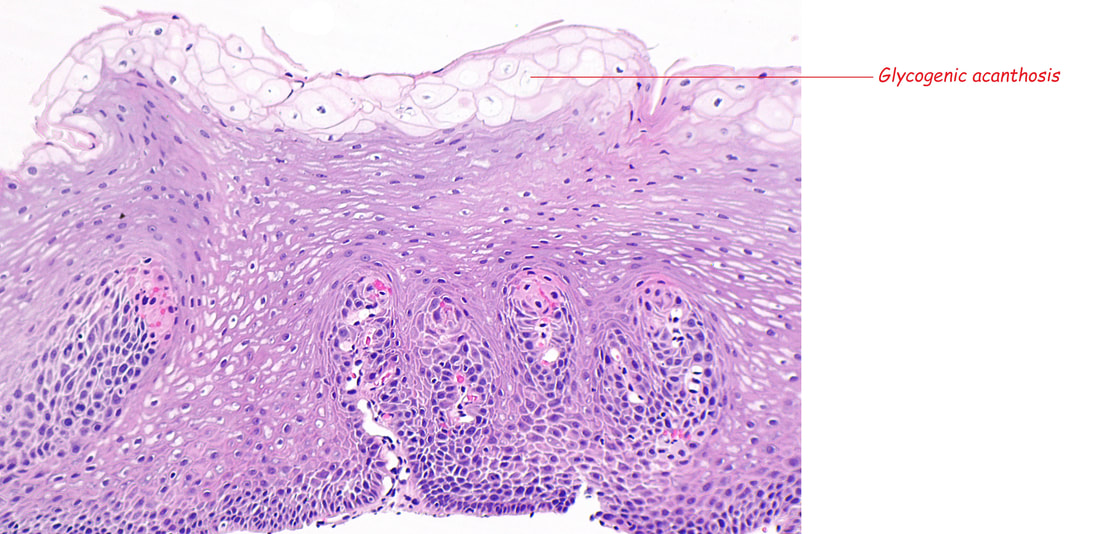
These are tiny lymphoid aggregates which are sometimes arranged as longitudinal chains mainly, near the ducts of oesophageal glands. These are most prominent in childhood and they regress. The main differential diagnois is that of 'glycogenic acanthosis' is a benign finding which becomes more common as we age. Histologically, they are composed of hypertrophied stratified squamous cells filled with glycogen and has no link with 'leukoplakia', and has no malignant potential. Below you have histologies of lymphocytic aggregates vs glycogenic acanthosis which tells a thousand words!
This was found in the distal oesophagus of a patient with dyspepsia. You take some samples.
WHAT WILL YOU TELL THE PATIENT?
■ This is a benign finding
Yes it is but do you know what it is?
■ We'll see what biopsies show
It's' a spot diagnosis!
■ This may well need EMR
Very unlikely unless those IPCL's look odd
■ Staging investigations are likely next
Can't imagine!
explanation
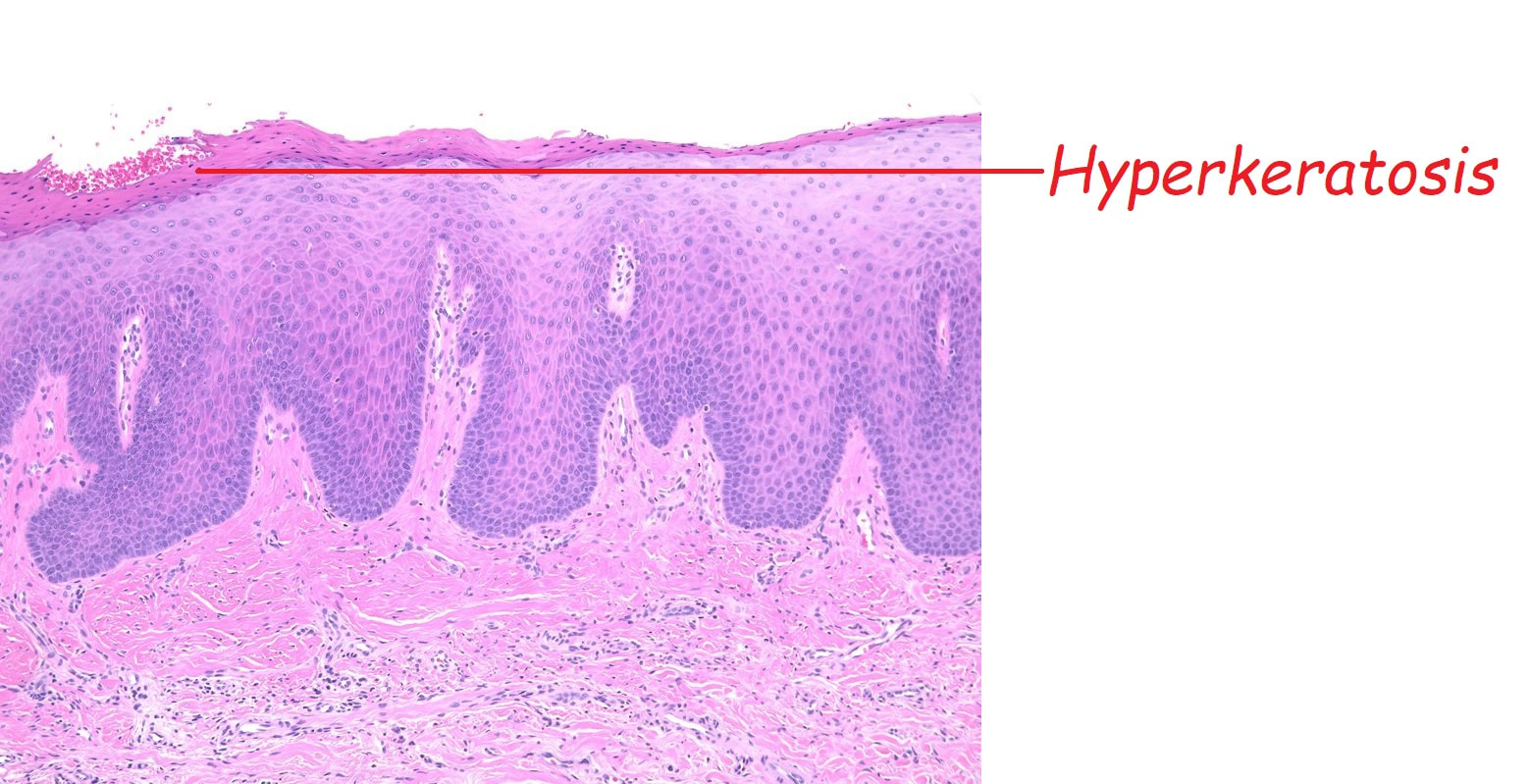
This may be a good example of 'we don't see' because we don't know what it is. Intuitively, we all blank this because it's a benign finding. But what is it? It's actually a patch of squamous hyperkeratosis with some underlying basal cell hyperplasia. The normal squamous basal cell layer is 1-4 cells thick but, in reflux oesophagitis thic increase up to 75% of the epithelial thickness. However, many cases of reflux oesophagitis does not exhibit basal cell hyperplasia and conversely, there are cases which will not be due to reflux disease as in this case!
In rodents (!), the finding has been linked with vitamin deficiencies and zinc deficiency. Of course, squamous hyperkeratosis in the mouth is associated with squamous neoplasia [ J Clin Pathol 2005;58:1325–1327 ] and for this reason, I always take some samples. However, I am yet to find a single case of dysplasia within a patch of oesophageal squamous hyperkeratosis !!!
This lesion was found in the transverse colon of a patient with a change in bowel habit
HOW WILL YOU MANAGE THIS?
■ I will ignore it
Brilliant, so would I !!!
■ Apply APC in the centre
Yes but only if patient has an IDA !
■ Apply APC from the periphery and working inwards
Advocated by some but life's too short!
■ Inject saline below and then apply APC
Overkill I think ...
■ Apply a clip to the centre
Doesn't work!
explanation
Angioectasia (formerly called 'angiodysplasia but of course these are not 'dysplastic'/ pre-malignant) are an acquired vascular malformation associated with advancing age, aortic stenosis, Von Willebrand Disease and chronic kidney disease. If a patient asks me, I usually put it more diplomatically as 'these come with maturity'. Seems kinder!
They are supposedly a little more common on the right than the left side. IF the patient is anaemic, APC is the treatment of choice. I have tried clips but they don't work. Be careful though as APC has been linked with delayed perforation in up to 1% of cases [Olmos J A. Dis Colon Rectum. 2006;49:1507–16]. I turn the APC down to 30 watts only and set the machine to 'pulsed APC'. Even so, please be sensible and stop after a maximum of a few seconds of heat! Some first raise the lesion with saline but first I don't think that this is necessary unless you are careless with the APC and secondly, I runs the risk of the underlying central, feeding vessel not actually getting ablated. Of course you could also use a 'coagulation grasper' if you are willing to pay €250 for the same outcome. Alternatively, you may consider a 'hot biopsy forceps' but of course these are now 'banned' and could land you in deep trouble if there is problem. Must admit that I use them instead of coagulation graspers because I'm too stingy to pay the €250. The correct answer in this case is probably not to do anything as the patient is not anaemic (and presumably has never been anaemic in the past). Secondly, I would advocate that you apply heat to the centre of the lesion only as those spidery legs will then disappear as in the example below.
By the way, it's not described in literature but there are actually two types of angioectasia. There are the 'spider naevi like' angioectasia as in the case above but angioectasia more commonly appear as red blobs as in the example below. Must admit that I don't know why. Must have something to do with the aetiology ...
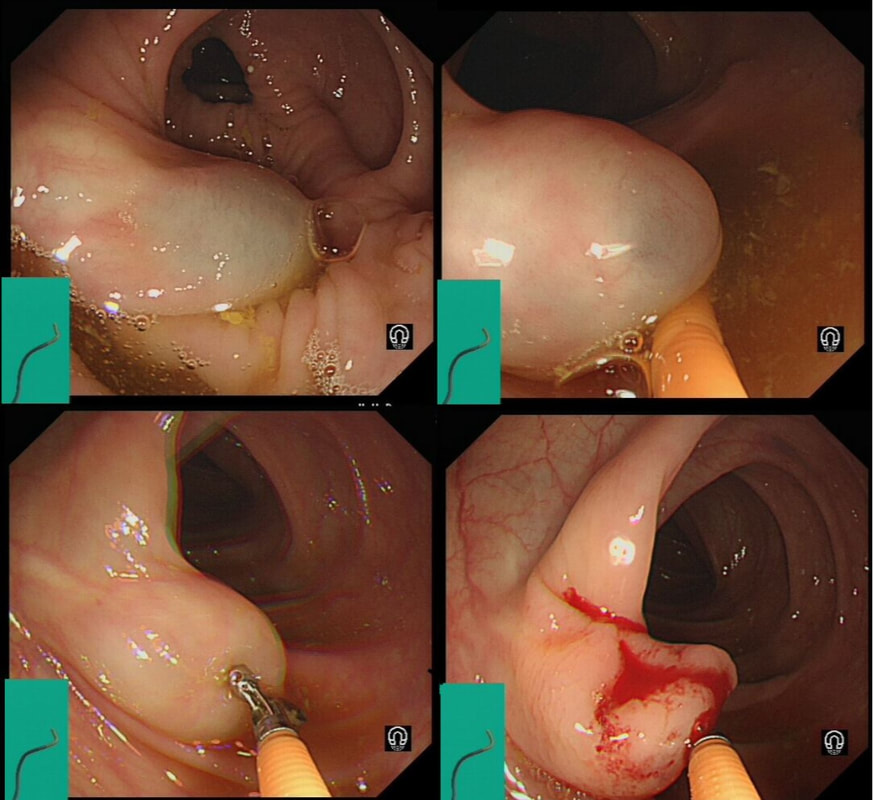
Large polyp at the recto-sigmoid junction on a short stalk which I have pre-injected.
WHICH DIATHERMY SETTING WOULD BE MOST APPROPRIATE?
■ Pure cut, effect level 1
it will bleed like stink !
■ A blended cut, effect level 4
Absolutely, steady does it!
■ Coagulation, effect level 1
Too much heat and too little cutting!
■ Coagulation, effect level 4
WAY too much heat and faaar too little cutting!!!
explanation
Endoscopy is an "art" and there is certainly more than one way of resecting polyps. A large polyp (turned out to be a TVA+LGD) could only be sustained by a plentiful supply of blood and therefore immediate bleeding is the most important concern. Removing it piecemeal could reduce the risk of bleeding but would be inappropriate as there is a risk of cancer, nestling deep inside the polyp head.
As you know, I am not a fan of placing clips beforehand. This is polyp is a good illustration why. A few clips on the stalk would get in the way, may lead the current to the nearby colonic wall and are unlikely to compress the vessels DEEP inside that stalk.
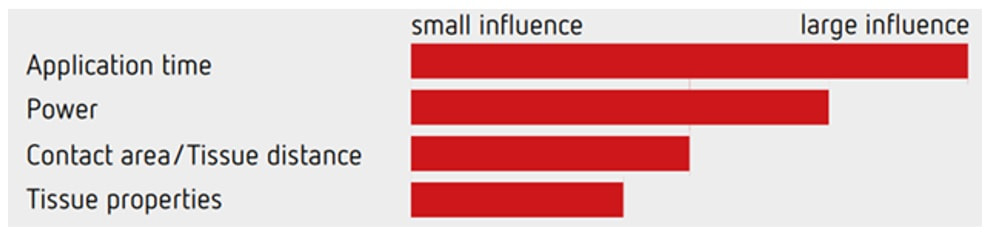
The graph above illustrate the four factors which influence the speed of your cut! The most important is the speed of closing the snare, the second is the power setting of your diathermy, the third is the thickness of your snare (thicker, braided snares take longer to cut through a polyp and will impart more heat) and finally the tissue properties (fatty tissue is slower to cut and I guess that a fat patient will require more electricity for the same about of cutting power).
Of course you can decide on the speed of snare closure but closing that snare yourself, or asking your assistant to for example count to 10. By the way, a good tip is to ask your assistant to stop applying pressure on the snare handle when the snare is visibly starting to cut through the stalk. In my experience the 'inertia' in the system will continue the snare closure at the correct speed whilst continuing to close the snare handle often cuts the centre too quickly. Anyway, this thick stalk could never by cut through with the blue pedal coagulation current. It's simply too thick and you would end up imparting an enormous amount of heat to the tissues. You may consider 'pre-coagulating' the stalk by first applying some coagulation power and then finish with the yellow pedal. It sounds clever but I suspect that the last bit which gets 'cooked' is the centre which carries the vessels. Therefore I hardly ever do it anymore. A 'pure cut' (called 'AutoCut' by ERBE and 'PureCut' by Olympus would be inappropriate as it would cut the stalk too quickly leading to immediate bleeding. You would only use pure cut setting for sphincterotomy, cutting a short oesophageal stricture etc. This leaves you with the correct answer which is B !
Just found another colonic lesion demonstrating the pillow sign.
WHAT IS THE DIAGNOSIS?
■ Lipoma
Doesn't it look translucent?
■ Lymphangioectactic cyst
Yes! Pillow sign + semi-translucent = cyst
■ Phlebectasia
A dilated vein? Nope!
■ Blue rubber naevus syndrome
The naevi are dark and firm!
■ Lymphangiosarcoma
Should then have a large solid component
explanation
Just when I'd say that all lesions exhibiting the 'pillow sign', I realised that the exception are 'lymphangioectatic cysts' (lymphangiomas) !
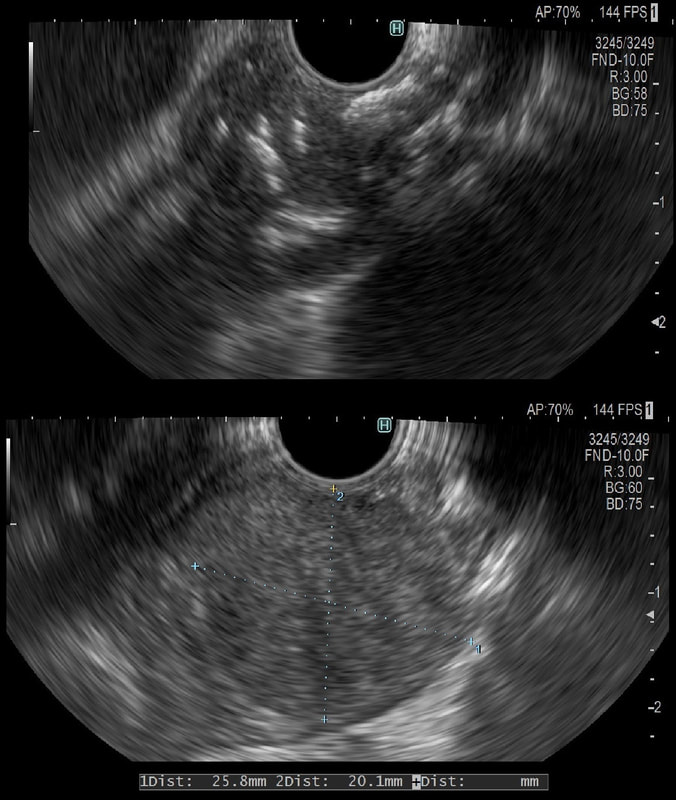
These are thought to be developmental malformations which can be found anywhere in or on the body. Of course they are entirely harmless. In fact the only harm they cause is by being poked about! There has been a reported case of peri-colic abscess formation following biopsy [Krishna SG, Endoscopy. 2012;44(Suppl 2 UCTN):E104–5]. Therefore, either biopsy or a 'de-roofing EMR' should probably be avoided. The best way to confirm the diagnosis, if it's in doubt is by EUS. It will show several cystic spaces confined to the submucosa and without any nearby nodes. There have been reports of lesions presenting with abdominal pain, and when pedunculated endoscopic resection may be possible [Case Rep Gastroenterol. 2017 Jan-Apr; 11(1): 178–183]. Must admit that I remain a little sceptical. Each case would have to be assessed on its own merit I guess !
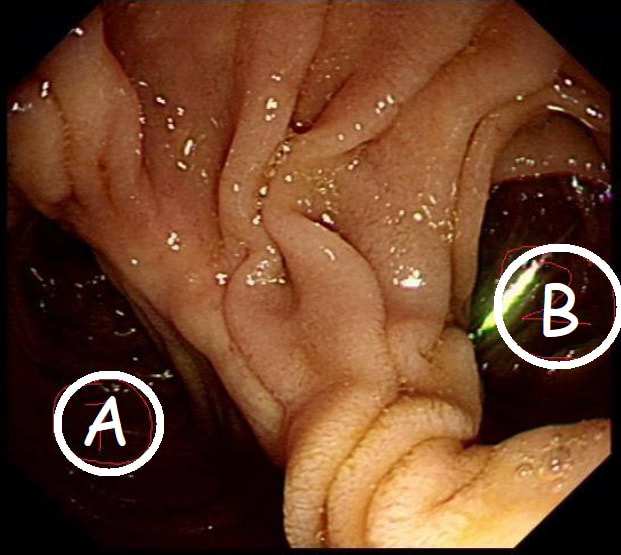
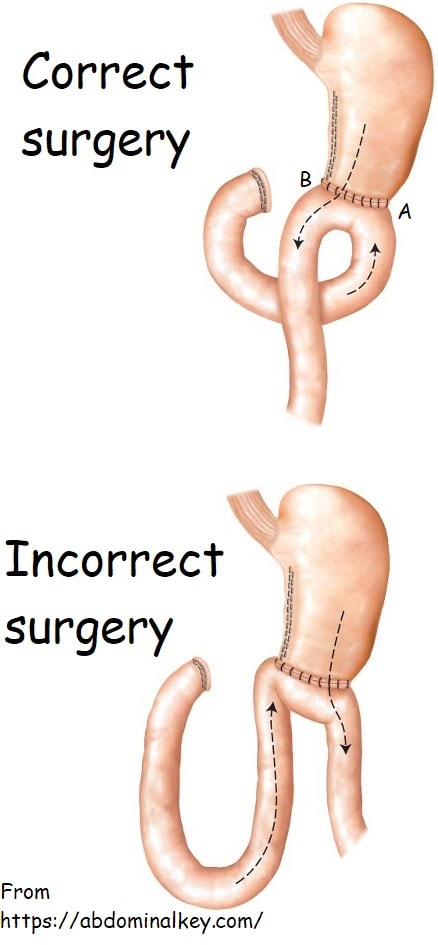
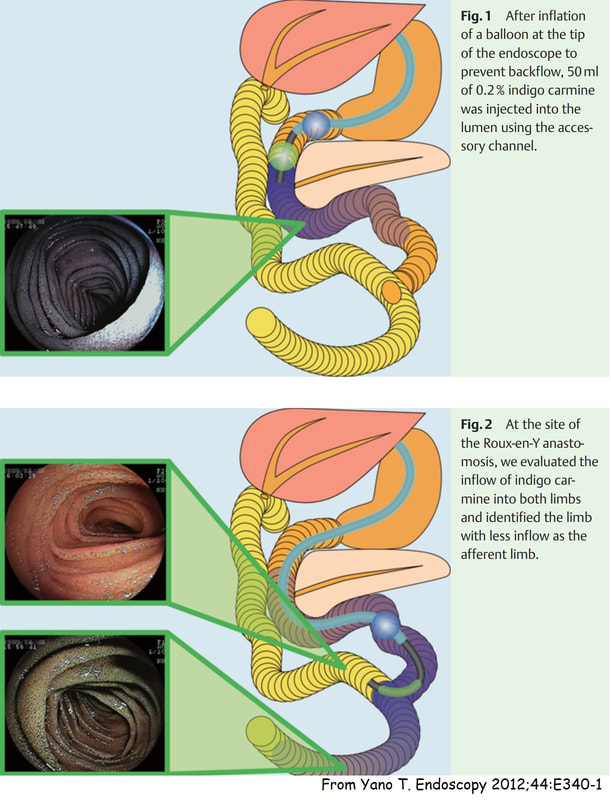
This is the gastric anastomosis of a patient who has previous undergone a Bilroth II distal gastrectomy.
WHICH PATH IS MOST LIKELY TO LEAD TO THE AMPULLA?
■ A
Seems wrong but it's right!
■ B
Seems right but is usually wrong!
explanation
At a 'correct' Bilroth II operation, the 'afferent limb' (taking bile to the stomach) should be placed on the greater curve (path A in the photo) and the efferent limb (taking food away from the stomach) should be on the lesser curve (path B). The reason why surgeons don't to it the other way around is that kinking and obstruction is then more common.
It's something of a trick question as at Enteroscopy, you would aim to get down the efferent limb (B). However, if your aim is to reach the duodenal ampulla, you should choose route A ! Dr Yano proposed a trick was [Yano T. Endoscopy 2012;44:E340-1] to help you at Enteroscopy. You inflate the 'distal balloon' (assuming that you are using a double balloon enteroscope of course), and instil 50 mL of indigo carmine solution beyond the tip of the scope. Peristalsis will not favour the afferent limb and the afferent limb will have less indigo carmine dye. Even when you get to the distal 'Y' in the Roux-en-Y', it will also help you (see below). The accuracy of this method is said to be 80%. However, I've come up with an alternative method! Give your patient a cup of 50ml of dilute indigo carmine dye to drink some 30 minutes before your examination. Of course you must warn him that the urine will be blue/green for the rest of the day. If all goes to plan, at Enteroscopy you will find that "the route which is blue is the way to go" ☺ !
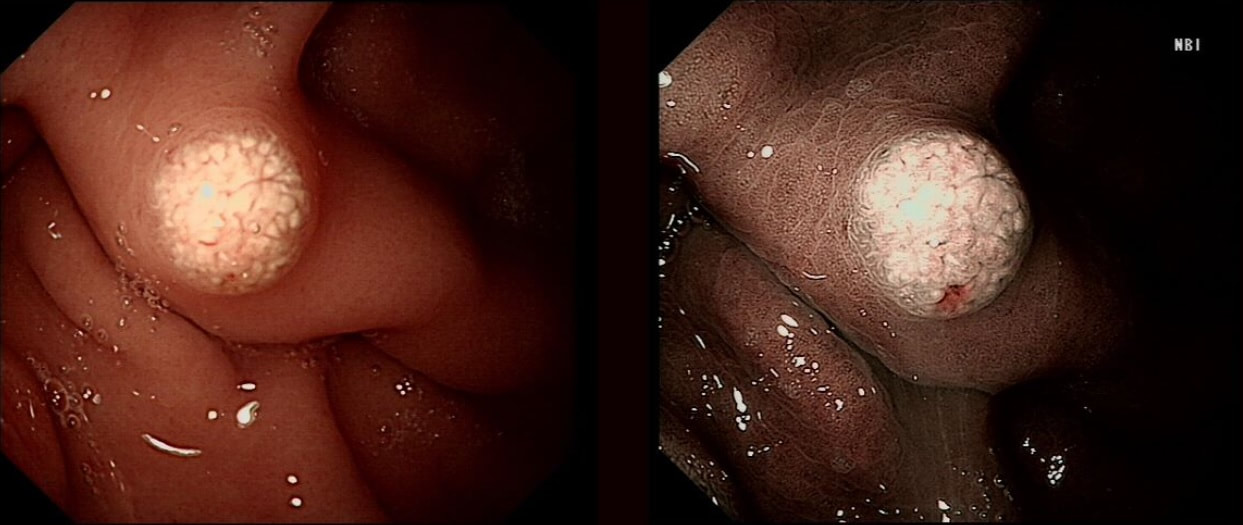
This was noted on the greater gastric curve
WHAT DOES IT MEAN FOR YOUR MANAGMENT?
■ I'll take a biopsy
No harm in this!
■ I'll check the patients cholesterol levels
No more likely to be abnormal than avg!
■ I'll check the patients urate levels
Don't think that you can get tophi in the gut!
■ I'll look carefully for EGC's
Brilliant!
■ I'll remove it by EMR
You are wasting your time!
explanation
Of course this is a 'Gastric xanthelasma' which histologically consists of macrophages stuffed full of lipid. There isn't a strong link with abnormal serum lipids but there is a strong link with gastric atrophy and gastric intestinal metaplasia ...
You will be familiar with 'Correa's hypothesis' and will know that widespread gastric atrophy and intestinal metaplasia is what's left when Helicobacters has run ravage for decades, leaving the stomach atrophied, achlorhydric and at risk of developing early gastric cancer! I used to regard gastric xanthelasma as a 'curiosity' only but actually, they are signposts that you should slow down and look VERY carefully for EGC's !!!
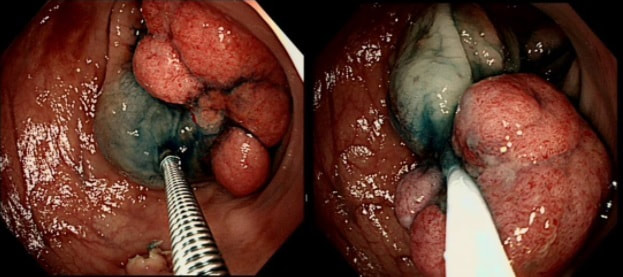
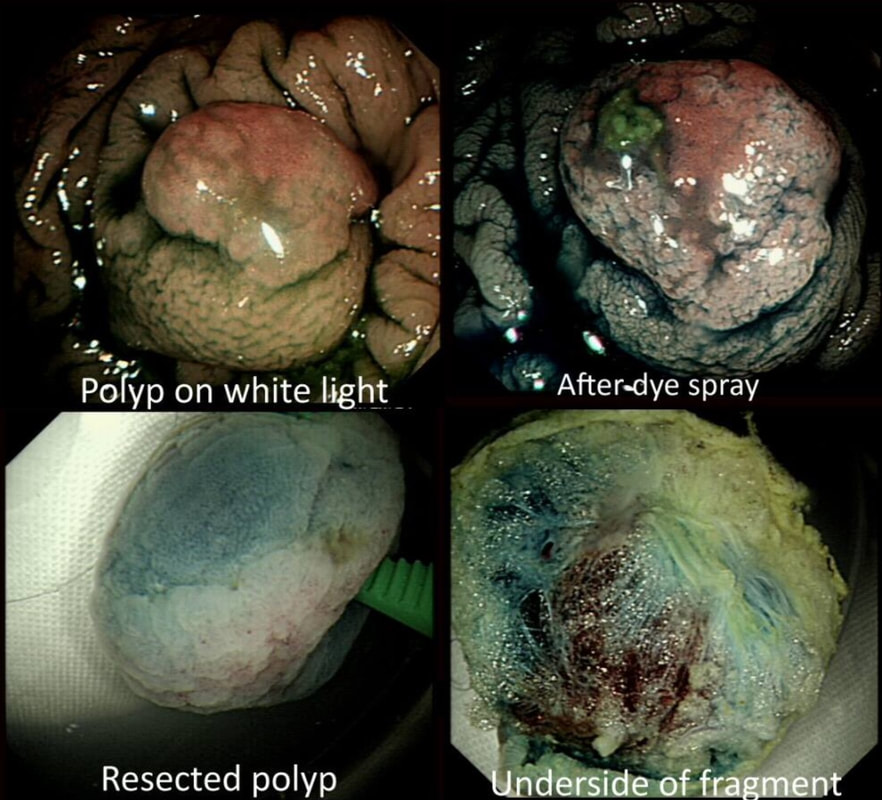
This polyp was removed from the ascending colon. Histology confirmed that the lesion was a TA+LGD. However, it also mentions an underlying lipoma ...
WHICH STATEMENT DO YOU AGREE WITH?
■ There was no lipoma!
That's right!
■ Hmm, may have missed that?
You didn't miss anything!
■ Yes, this was noted at the time
The submucosa is often 'fatty' in the right hemicolon!
explanation
It's common for the submucosa on the right side of the colon to be laden with fat. In fact, you can see the fat globules in the bottom-right image where the lesion has been turned up-side-down. It can be a little alarming to see fat floating about in the mucosal defect after the resection . You may think that you have perforated but it's normal !
|
Categories
All
|