|
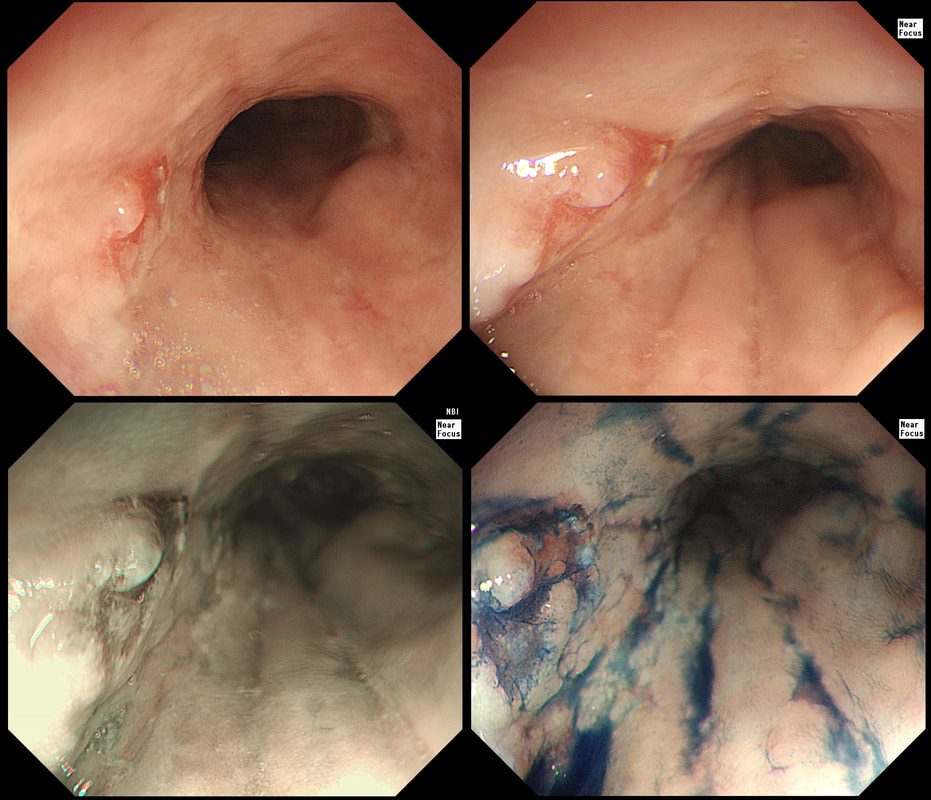
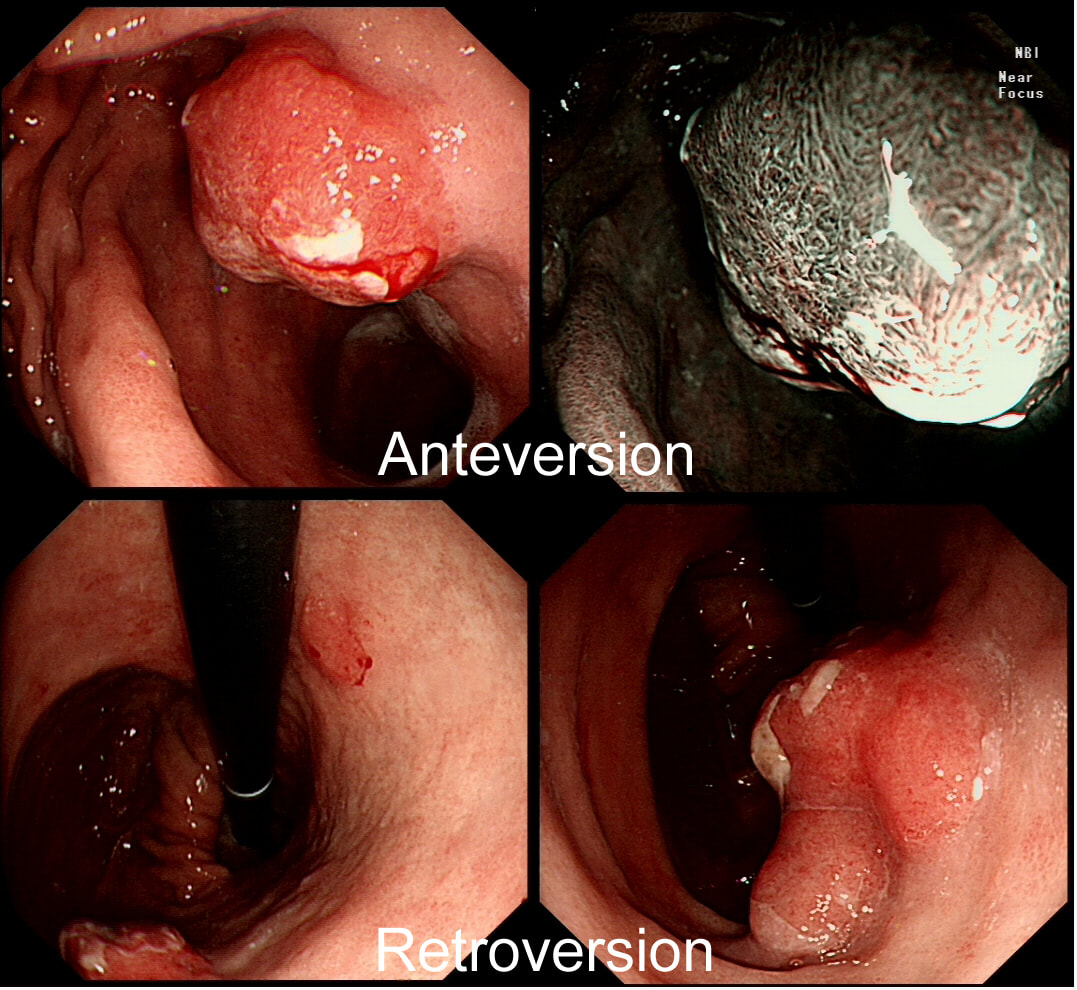
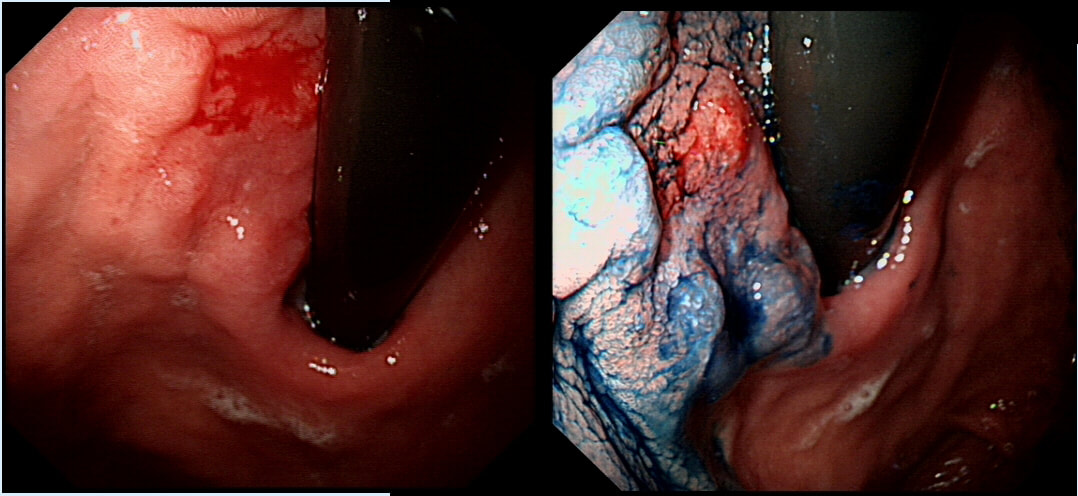
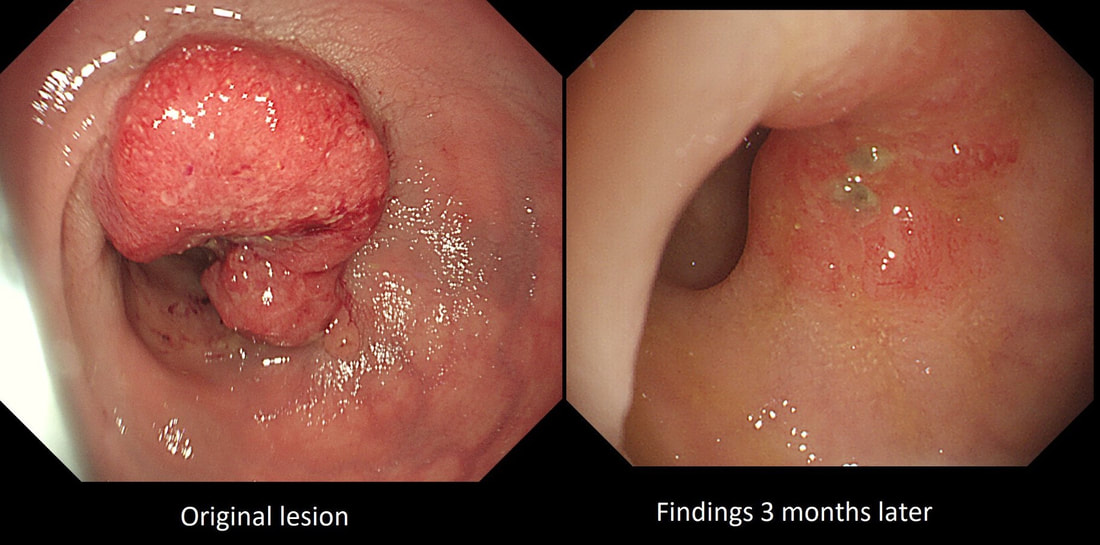
A year ago, this patient underwent RFA for a C5/M6 Barrett's harbouring HGD.
WHAT IS THE MOST APPROPRIATE MANAGEMENT?
■ APC ablation
INCORRECT, the lesion is NOT FLAT!
■ RFA
INCORRECT, because the lesion is too worryingly nodular
■ EMR
CORRECT, the only way to make sure!!!
explanation
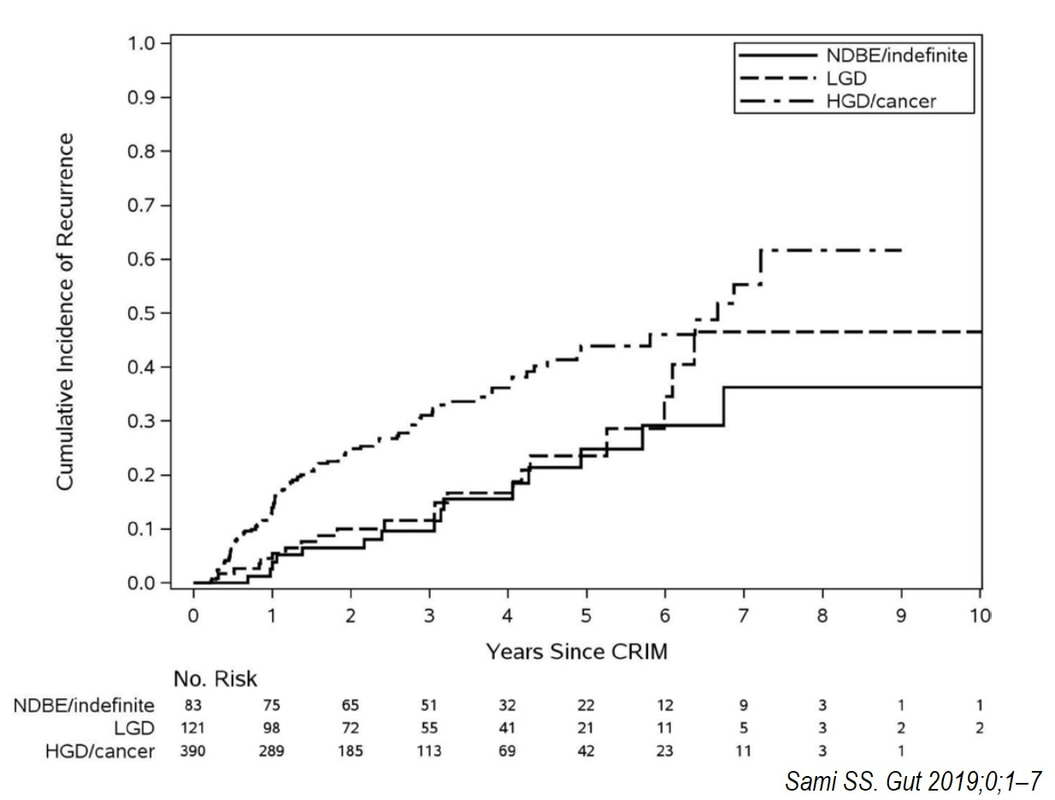
This is the problem with RFA, it doesn't last forever! After 5-6 years, about half your patients will have relapsed. Predictably, it's the patients with the most unstable changes who are the most likely to relapse (see graph below from Dr Sami's landmark paper). This is the reason why surveillance at 12 month and then 3 yearly is recommended by the BSG when the 'index histology' was LGD. However, after HGD or IMca, surveillance after 3 months, 6 months and then annually is recommended. Click here to read those recommendations in full.
Endoscopically, it can be difficult to spot a small rim of Barrett's returning, which over time slowly pushes the squamo-columnar junction proximally. In most cases, this is how Barrett's return over time. It's for this reason that we advice taking 4 biopsies from the columnar side of the SCJ'n. If your pathologists report dysplasia in any of these samples, a "top-up" RFA may well be indicated. Less commonly patients develop a nodule emerging from the 'neo-squamous mucosa' as in this case. Clearly you need to resect this for a full histological analysis. Ablation would be like sweeping it under the carpet! The nodule was removed by EMR and histology revealed an intramucosal cancer! Rather worrying that this patient's disease has in some sense, 'progressed' after RFA. Prof Rebecca Fitzgerald's group at Cambridge has shown us that the DNA is pretty thoroughly mashed up by the time the mucosa looks dysplastic down a microscope. Is it really possible for RFA to 'clean up' and remove all those chromosomal translocations ? This patient is undergoing a gastroscopy because of abdominal pain. Some 20 years ago he underwent a distal gastrectomy for peptic ulcer disease. WHAT IS THE DIAGNOSIS?
■ a) Large hyperplastic polyp
No, there is more going on!
■ atrophic gastritis with malignant polyp
Closer to the truth but not the whole truth!
■ None of the above
What else could there be??!
explanation
You can tell that the mucosa must be atrophic by the mucosal xanthelasma at about 25seconds into the video. The polyp dangling at about 12 O'clock is covered with the same irregular surface as the base from which is arises. Finally, there is a large plaque-like area extending between 3-10 O'clock which does look concerning. It goes all the way up to the small bowel anastomosis. In general, large plaque-like cancers in the stomach do look intimidating but is usually intramucosal disease, provided that the appearance is uniform. The subpedunculated polyp indicate more 'heterogenous' disease. The obvious question is now, should we offer the patient an attempt at endoscopic resection or a 'completion gastrectomy'? The second part of that particular question is of course, 'is the patient actually a candidate for a completion gastrectomy?' At the time of my endoscopy, I didn't like the wide extend of the cancer and its heterogenous appearance. To also convince our MDT, I removed that subpedunculated polyp which was confirmed as intestinal-type intramucosal adenocarcinoma which was mainly 'moderately well differentiated' but with 'focal poor differentiation'. Predictably, the deep margin was positive, as the surface structure of both the polyp and the underlying gastric mucosa look the same. Although both an EUS and CT were reassuring, because the patient was a surgical candidate and also because of the large area of heterogenous disease, extending all the way up to the small bowel anastomosis, the patient was in the end offered a gastrectomy rather than an endoscopic resection. Ultimately, histology confirmed a very 'heterogenous disease' with areas within this 13cm lesion containing:
Is there any evidence that heterogenous gastric cancers (i.e. cancers with a mixed histology) are less likely to be cured by endoscopic resection? Actually there is! Ozeki et al looked at more than 3000 endoscopic resections for early gastric cancer and found that lesions with a mixed histology was significantly less likely to be cured by ESD (35-42% cure rate vs 76-92% cure rate). Ozeki Y. Mixed histology poses a greater risk for noncurative endoscopic resection in early gastric cancers regardless of the predominant histologic types. Eur J Gastro Hep 2021;32(2);186-193 Fortunately, there was no nodal disease. Almost everyone got this right when first published on our FoE facebook group.
This suspicious looking 3cm gastric nodule was referred for an endoscopic removal after a CT and EUS had been reassuring. No samples have been taken as not to impede the resection
WHAT WOULD YOU DO NEXT?
■ First sample the nodule
Good idea but not good enough!
■ Sample nodule, antrum and gastric body
YES! To tell if this is a type III NET
■ Go ahead and resect as planned
NET's >2cm should be considered for surgery first!
explanation
This is a gastric neuroendocrine tumour (NET). You can tell because of the dilated vessels along its side (see below). A typical feature of all gastric NET's.
To remind you, gastric NET's are classified as; type I (70% - 80% of gastric NET's) linked with hypergastrinaemia secondary to an atrophic gastritis and classically appearing as multiple, small gastric nodules. The rare type II gastric NET account for about 5-8% is associated with hypergastrinaemia from a gastrin-secreting tumour such as in the MEN-1 syndrome or the Zollinger–Ellison syndrome. Finally, the type III NET (20%) is solitary, larger nodules with a high mitotic index arising in a healthy gastric mucosa. These are the ones not to miss as they need a cancer-like gastric resection particularly if 2cm or larger in size ! Sampling the nodule will confirm that it's a NET and give you it's 'mitotic index'. Grade III lesions (20% mitotic index) should certainly be resected surgically. However, also sampling the antrum and body is even better because if the mucosa is found not to be atrophic, this confirms that the lesion is a type III gastric NET !
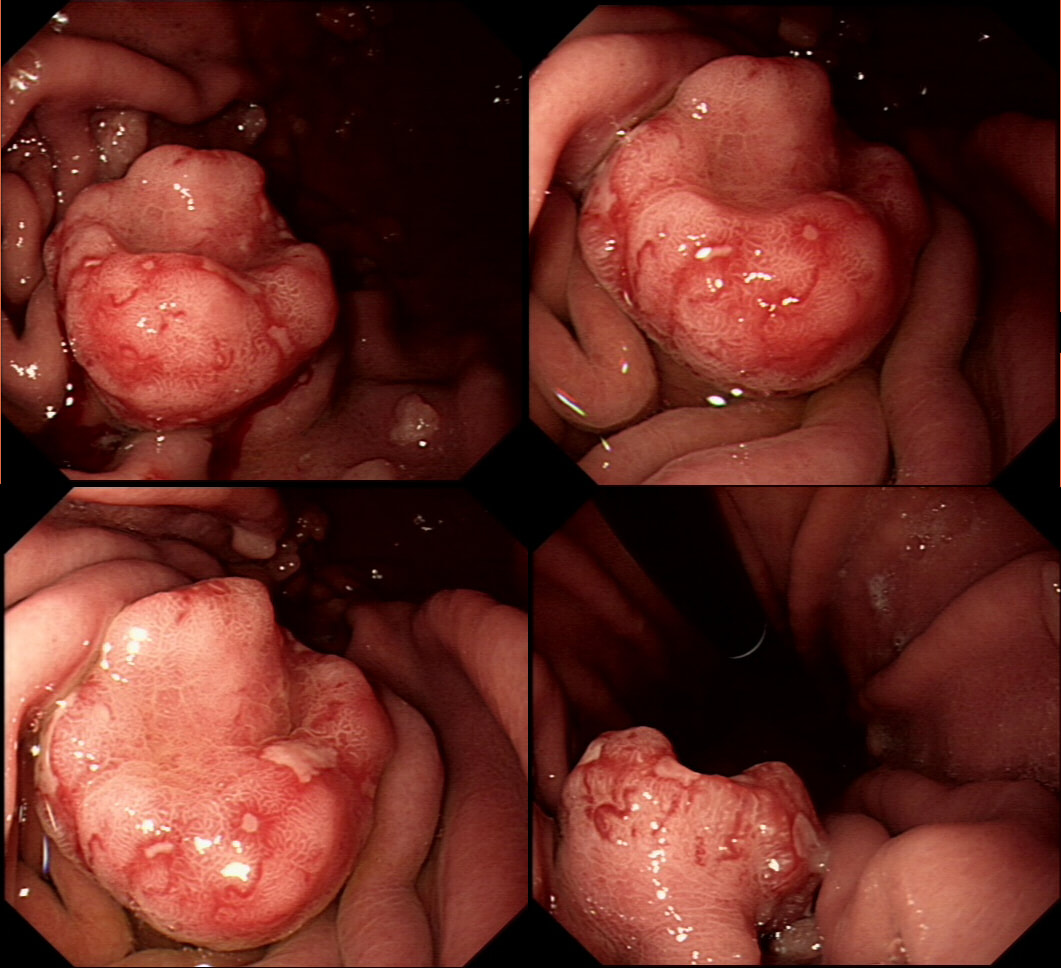
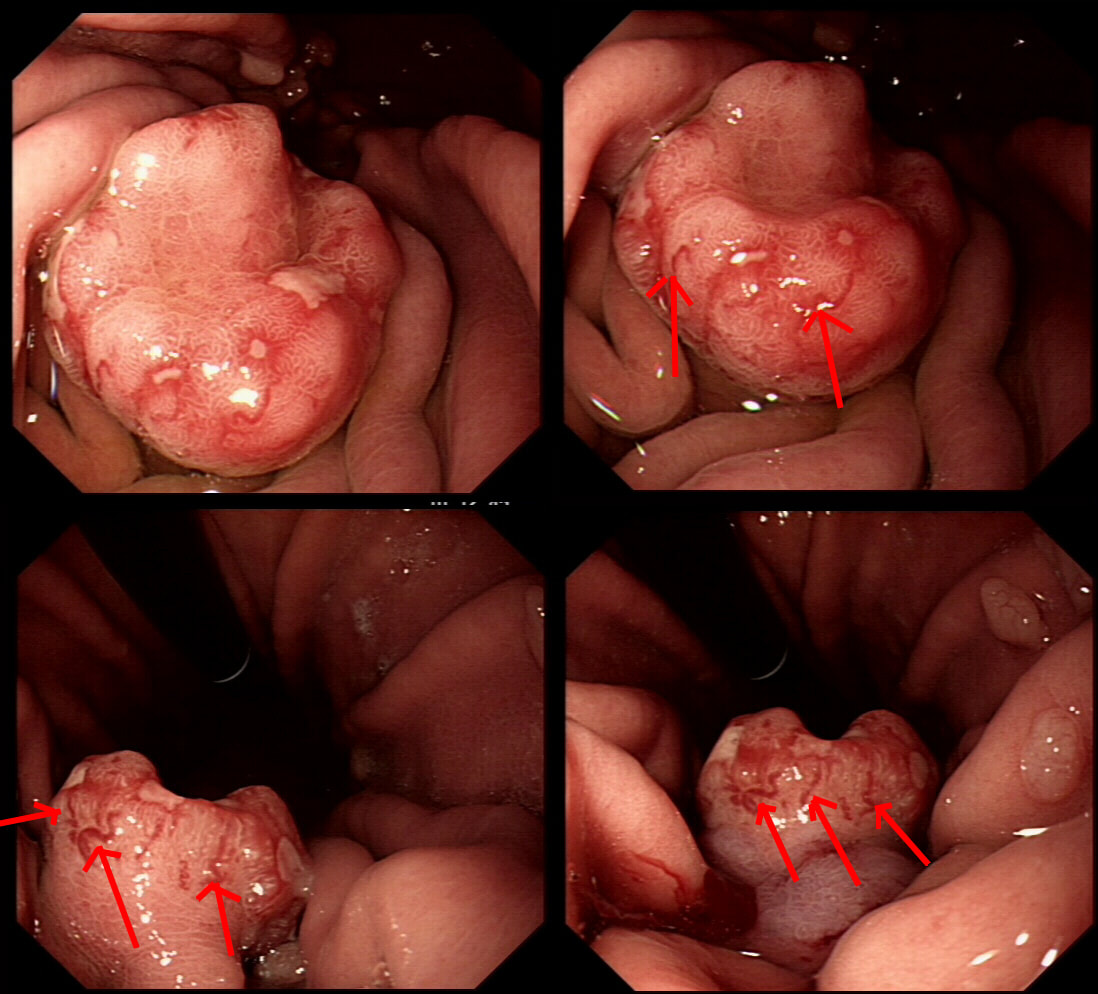
Previous sampling of this gastric nodule has confirmed HGD and an EMR has been organised.
WHAT IS THE MOST APPROPRIATE MANAGEMENT?
■ Take further set of Samples!
No, because it's unlikely to change anything!
■ Organise staging CT and EUS first!
YES! This may well be invasive cancer!
■ Go ahead and resect it!
INCORRECT!
explanation
The correct approach would be to slow down a little! Pathologists would always like more samples but actually, surface samples may not be a true reflection of what is nestling deep within this lesion! This lesion is a little over 3cm in diameter and it would be very surprising if it wasn't an invasive cancer after all. Furthermore, there is a suspicious looking red, flat patch close to the shaft of the endoscope in the bottom row image on the left.
Ultimately, this patient underwent a gastrectomy which confirmed that the large nodule was indeed an invasive adenocarcinoma invading as deep as the muscle propria layer (T1b). In addition, there was multifocal LVI and a nearby involved node (N1). That red patch turned out to be a spot of HGD. Because EUS found a suspicious node, a gastrectomy was the most appropriate therapy. Of course, we don't normally organise a CT and EUS for endoscopically resectable lesions which are not thought to be malignant. Of course biopsies have indicated that this is HGD only. However, the lesion is chunky and its unthinkable that it's HGD only. More surface biopsies may or may not reveal the true nature of the lesion. And frankly it doesn't really matter because endoscopically this is likely to be an invasive cancer. It's a dysplastic nodule about 3cm in diameter and probably close to 2cm in height !!! Of course, the pathologists always want more samples but with a lesion of this size, of borderline resectability, a CT and EUS is more reassuring than further samples. The EUS revealed a chunky node and the patient had an appropriate gastrectomy. How about a a test-lift? Unfortunately, test-lifts are difficult to interpret in the stomach and is likely to be 'borderline' committing you to 'having a go' trying to resect the lesion endoscopically. Of course, there is nothing wrong with an 'attempt at EMR/ESD' but the point is that first you would like to have the reassurance of a normal CT and EUS. Of course, you would probably take another set of samples rather than just looking at the lesion but whatever the pathologists say, it will not trump the endoscopic diagnosis of; ' invasive cancer'. But why not just 'lob it off' for a definite diagnosis as well as staging? The reason is that a ESD resection of a cancer is very difficult. This is because the semi-translucent lifting plane disappears due to the desmoplastic reaction and you end up cutting blindly through opaque beige coloured tissue. Perforating during the resection of a cancer is potentially devastating for the patient as it can upstage the lesion to 'disseminated disease'. As the stakes are very high in cancer, it would be inappropriate to subject the patient to a significant risk with only a slim chance of benefitting.
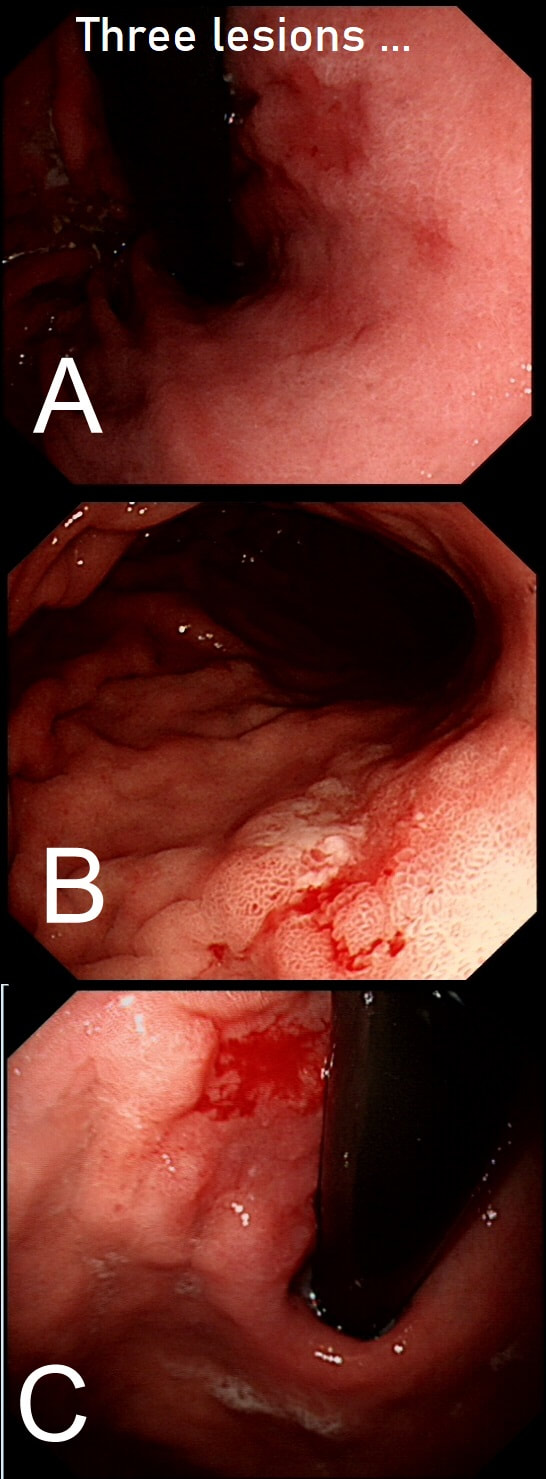
Three gastric lesions (A,B and C) in three separate patients all undergoing a gastroscopy because of iron deficiency anaemia
WHAT IS YOUR ENDOSCOPIC DIAGNOSIS?
■ BENIGN GU's
INCORRECT!
■ GASTRIC ADENOMAS
INCORRECT
■ EGC's
CORRECT!
EXPLANATION
These are all intramucosal gastric cancers. The typical scenario of an early gastric cancer is:
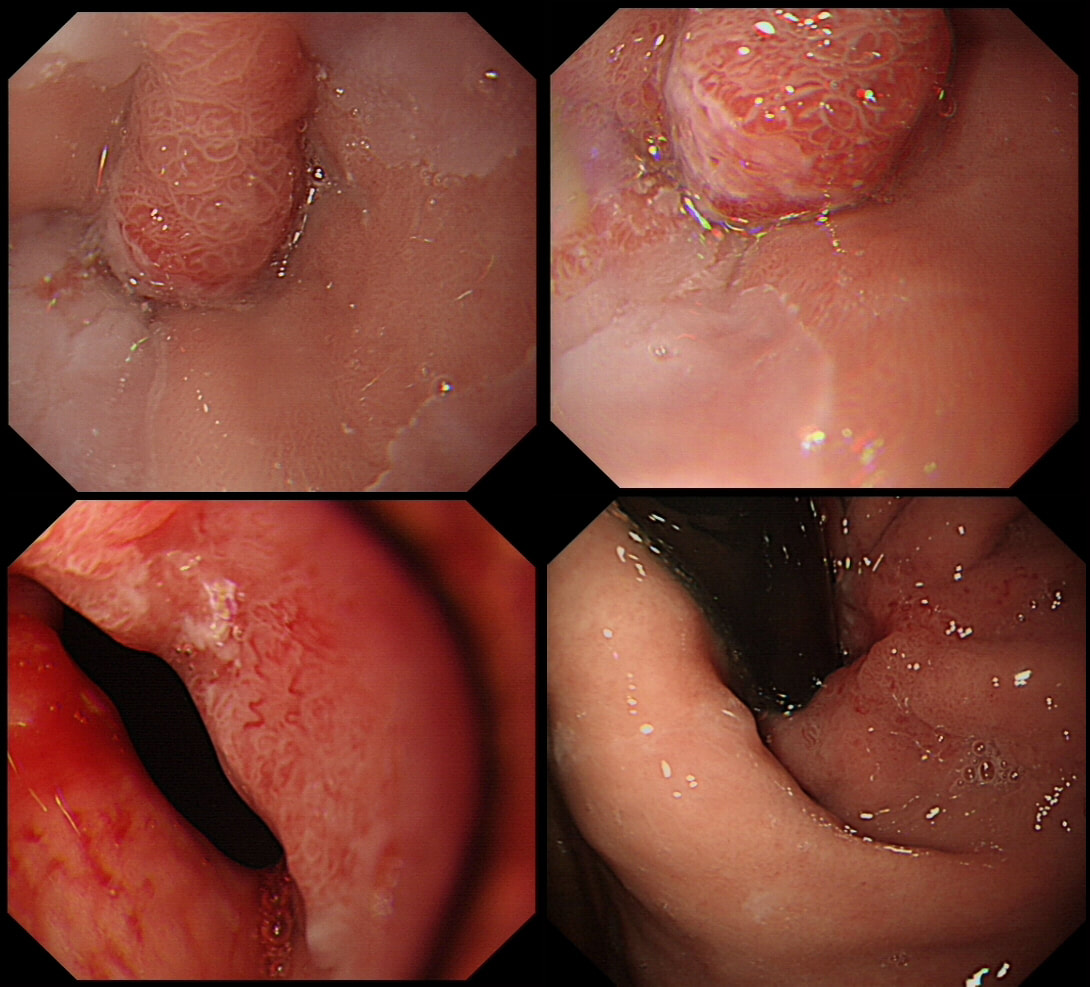
Lesion found at the gastro-oesophageal junction
SHOULD I ATTACK OR ABORT?
■ ATTACK!
INCORRECT. SOMETHING HORRIBLE IS LURKING BELOW!
■ ABORT
CORRECT, THERE IS A HORRIBLE LOOKING ULCERATED CANCER SEEN AT CARDIA
explanation
From above this looks like a reactive nodule, perhaps secondary to reflux. Naturally, one couldn't rely on a visual inspection and an EMR seemed indicated. However, on retrovertion (bottom right image), the true nature of the lesion is revealed. There is a depressed, indurated area just at the GOJ (the close-up image at the bottom on the left show some irregular, malignant looking vessels). I diagnosed a type III early gastric cancer (Sievert's type III guess) probably involving the muscle propria (T2 disease) and organised an EUS. Surprisingly, the subsequent EUS upstaged the lesion to T3,N0,M0). I find it tricky to "stage" lesions at the cardia. Even small lesions can be far more advanced that expected.
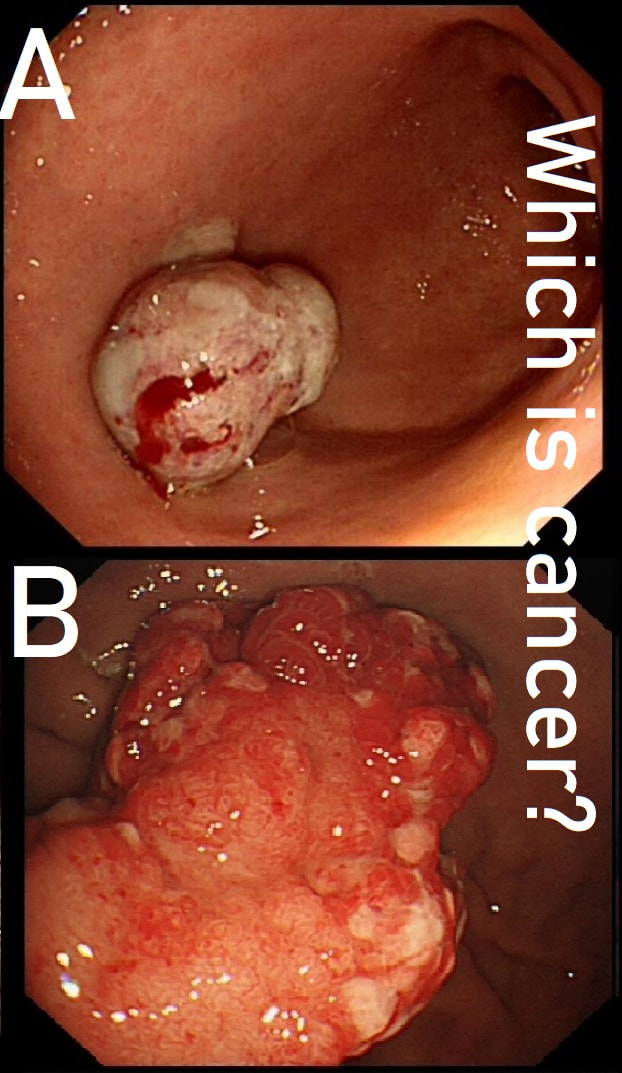
These two antral polyps look very different. That's because one is a polypoid cancer and the other is an innocent hyperplastic polyp.
WHICH IS WHICH?
■ A is malignant
Yes! A was poorly differentiated gastric carcinoma!
■ B is malignant
Surprisingly, B is actually the innocent hyperplastic polyp
explanation
Most (73%) thought that the hyperplastic polyp (polyp B) was the malignant one. Although large, the angry red colour and white spots are both strongly suggestive of a hyperplastic polyp.
Interestingly, in both cases, the patient presented with anaemia. Of course, when the patient undergoes the endoscopy because of anaemia, any gastric polyp should be viewed with suspicion. Although the lack of surface crypts was disconcerting, as polyp A did not seem to be firmly attached, it was removed as a single fragment (of course). Histology confirmed that the lateral and deep margins were clear and there was no lymphovascular invasion (LVI). In the subsequent 'cancer meeting', it was pointed out that 'poor differentiation' may be considered an indication for surgery. This would indeed be the case elsewhere in the GI tract. However, in the stomach, poor differentiation 'on its own' (clear margins and no LVI) is 'allowed' provided that margins are clear, there is no invasion into the deepest layer of the submucosa and, most importantly, there is no LVI. Emerging data highlights LVI as THE MOST IMPORTANT predictive factor anywhere in the GI tract. The original ‘Japanese Standard Gastric EMR criteria’ gave the go-ahead for resection of well differentiated cancers up to 2cm provided that there was no surface ulceration. Nowadays, most of us would apply the 'Japanese Extended criteria’ which allows us to attack well differentiated EGC's up to 3cm in size even if there is some superficial ulceration of the surface. Pertinent to this case, the extended criteria also allows us to attempt to resect poorly differentiated cancers up to 2cm provided that there is no surface ulceration.
This was found at the gastric cardia in a previously well 75 yr old woman. Biopsies have reported 'at least intramucosal cancer'.
Fortunately, both EUS and CT were reassuring WHAT IS NOW THE MOST APPROPRIATE ACTION?
b) Recommend surgery
INCORRECT!
c) Recommend chemo-radiotherapy
INCORRECT!
Explanation
This proved to be a difficult question as 90% of our FB group actually thought that surgery would be necessary. This is because cardia cancer is difficult to stage both endoscopically and by EUS. This IIa+IIc lesion (i.e. flat elevated lesion with a central depression) does indeed look concerning for invasion into the deepest part of the submucosal space (sm3 invasion). By the way, test-lifts are difficult to interpret in the stomach and are better done as 'test-resections'. Naturally, if you find that the submucosal plane is obliterated by the pale, fibrous tissue of 'desmoplasia', you'll have to bail out.
Although the chances of a cure may be a little slim, a total gastrectomy at the age of 75 is a lifechanging decision (and may be a life-ending decision) and should only be considered if absolutely necessary. The 'half-way house' of radiotherapy was not on offer. This is because the stomach usually don't cope well to 'full dose' radiotherapy (i.e. given with a curative intent). In the end, the lesion was removed by ESD and histology confirmed as mainly HGD and IMca but with a central invasive focus extending into the middle of the submucosal space (sm2 invasion). Fortunately, the differentiation was good and most importantly there was no LVI !!! This was 7 years ago and the patient remains well, aged 82 yrs ! I was asked to remove this sigmoid lesion. Previous samples have revealed a TA+HGD. The dark colouration is due to a previous ink tattoo, placed rather too close to the lesion itself for my liking. SHOULD I ABORT OR ATTACK?
ABORT!
Correct!
ATTACK!
WRONG!
EXPLANATION
When this question was first published on our FB group, >95% correctly answered; Abort ! The complete absence of a crypt pattern means that this lesion should not be endoscopically resectable. The Japanese would say that there is evidence of 'massive invasion' (invasion beyond 2mm below the muscularis mucosa). Of course, I confirmed my impression with a 'test-lift', which of course failed. Subsequently, surgery confirmed that the lesion was T3 (invading through the muscle propria layer) but luckily no nodes were involved. The take-home message? If a 'test lift' had been done a the first examination, we could have saved a lot of time. A test-lift will not scupper a future endoscopic resection! This high rectal lesion was treated with chemoradiotherapy 3 months ago (left image). On re-assessment of the site, there is some erythema remaining. WHAT IS THE LIKELY NEXT STEP? a) APC ablation b) EMR c) ESD d) Further radiotherapy e) Surgery explanation
The initial staging of this rectal cancer was T2, N0. It took many years for Prof Angelita Habr-Gama (a famous colorectal surgeon from Sao Paulo, Brazil) to convince her fellow surgeons that neo-adjuvant chemoradiation for stage 0 rectal cancer could be as curative as surgery but without the need for a subsequent AP resection and a stoma. She produced a landmark paper; “Operative versus nonoperative treatment for stage 0 distal rectal cancer following chemoradiation therapy: long-term results,” [Ann Surg 2004;240(4):711-7] which proved the case. This particular patient had received chemoradiotherapy and the result has been good but NOT complete. Samples taken from that red area confirmed residual cancer. As it happens, the patient was not a surgical candidate and a brachytherapy boost was instead given. This was of course something of a trick question as both D and E are 'correct' next steps. |
Categories
All
|