|
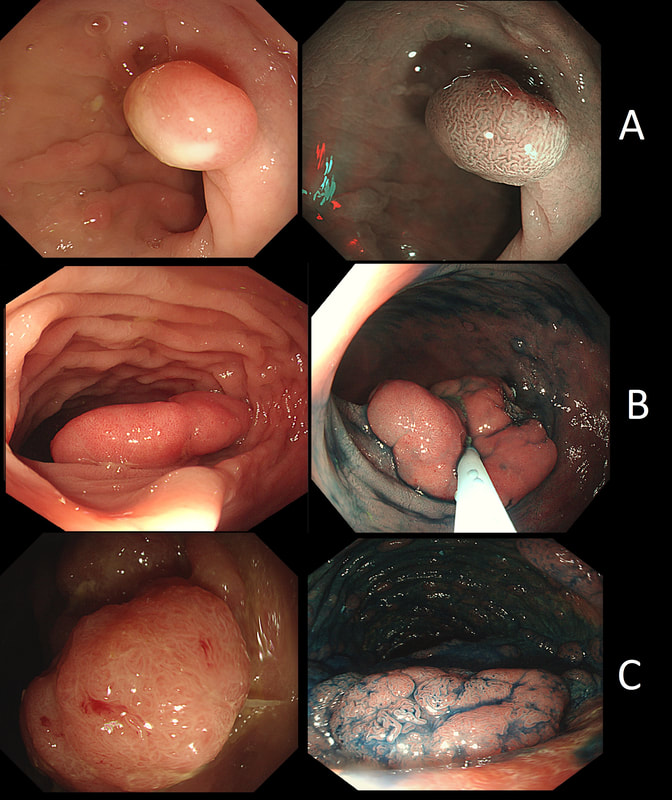
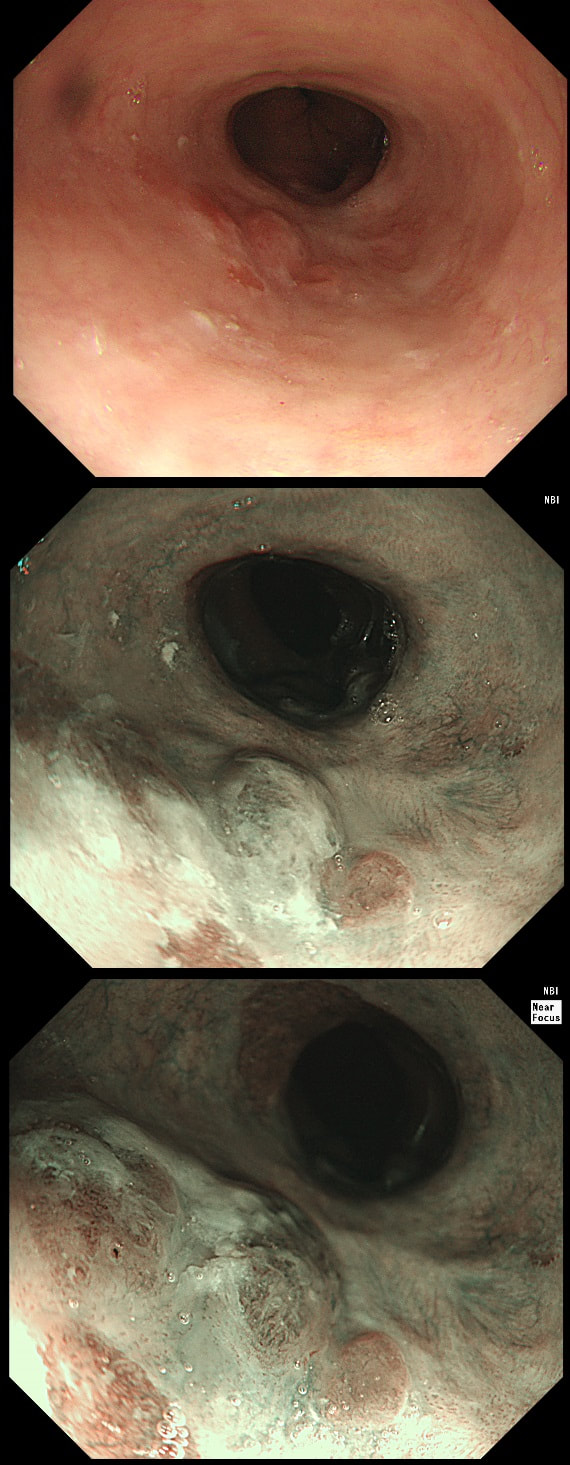
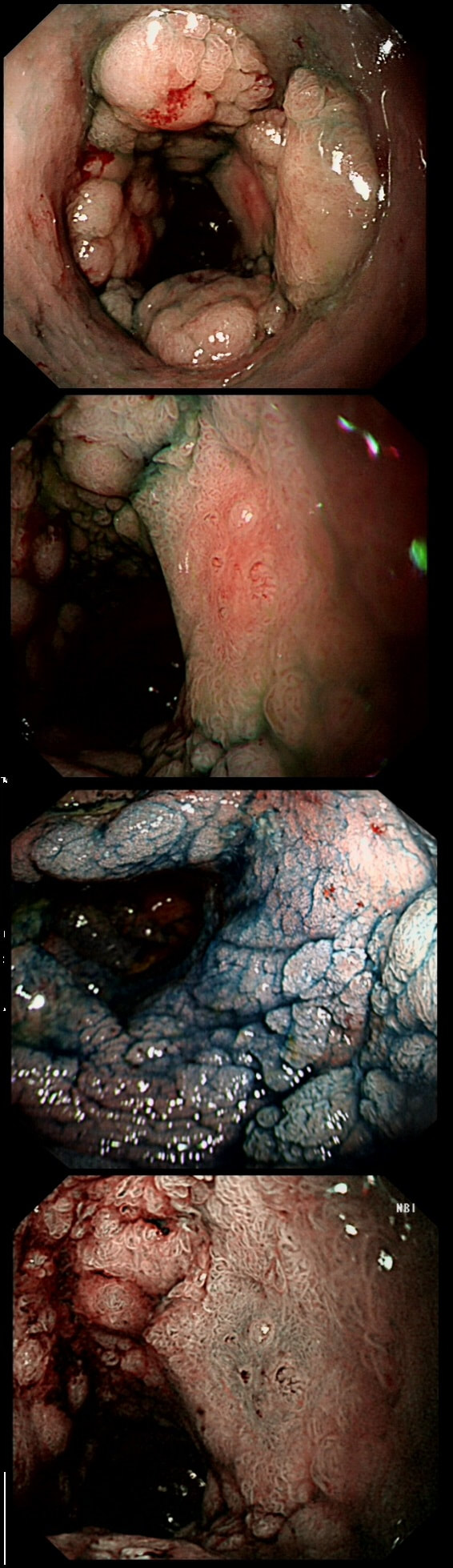
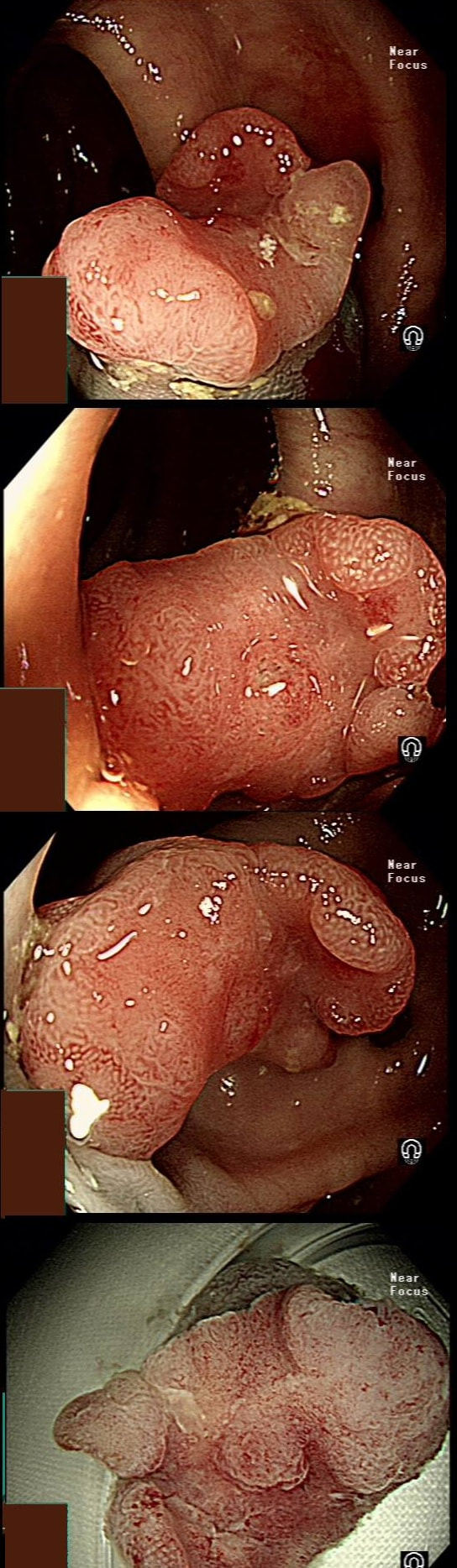
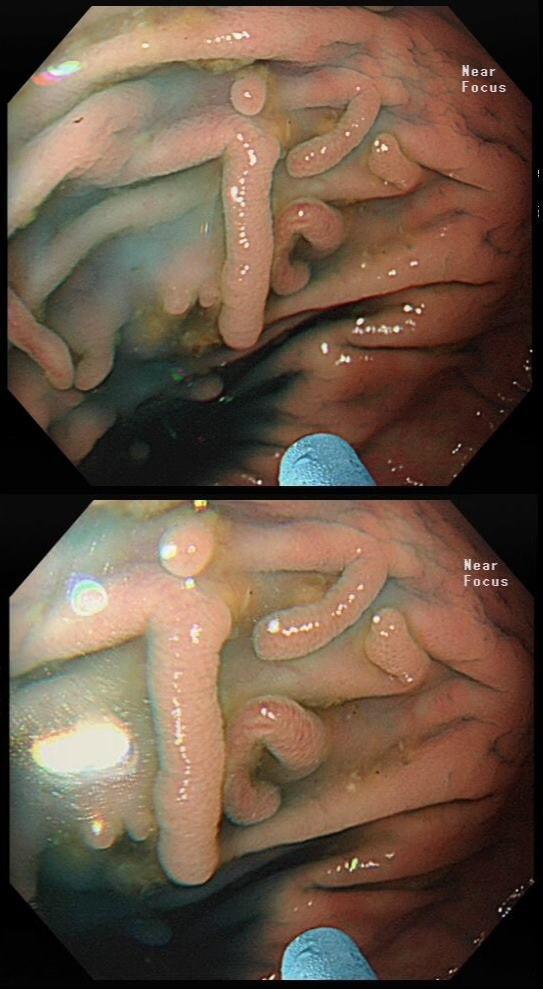
Here are three colonic polyps. One is a TA+LGD, another a TA+HGD and the final is a TVA+HGD
WHICH POLYP IS THE TVA?
a) A
Nope, this one has slit-like crypts typical of a tubular adenoma (TA). In fact, most small polyps are TA's. Presumably there is a progression from TA→TVA→Cancer apart from depressed lesions (IIc lesions) which go directly from being TA's to cancer.
b) B
It's a little difficult to see the crypt pattern without a close-up. However, you don't need to! Look at the growth morphology. This is a LST-NG and they are ALWAYS TA's!
c) C
Yes! Gyrate crypts best seen after indigocarmine dye spray (I think).
explanation
Most 'laterally spreading TVA's are of course LST-G's. However, this doesn't have the usual cobblestoned appearance of a 'laterally spreading tumour of the granular type'. It's rather 'chunky' in fact. That's because its harbouring HGD! A rare beast indeed as almost all laterally spreading TVA's only harbour LGD.
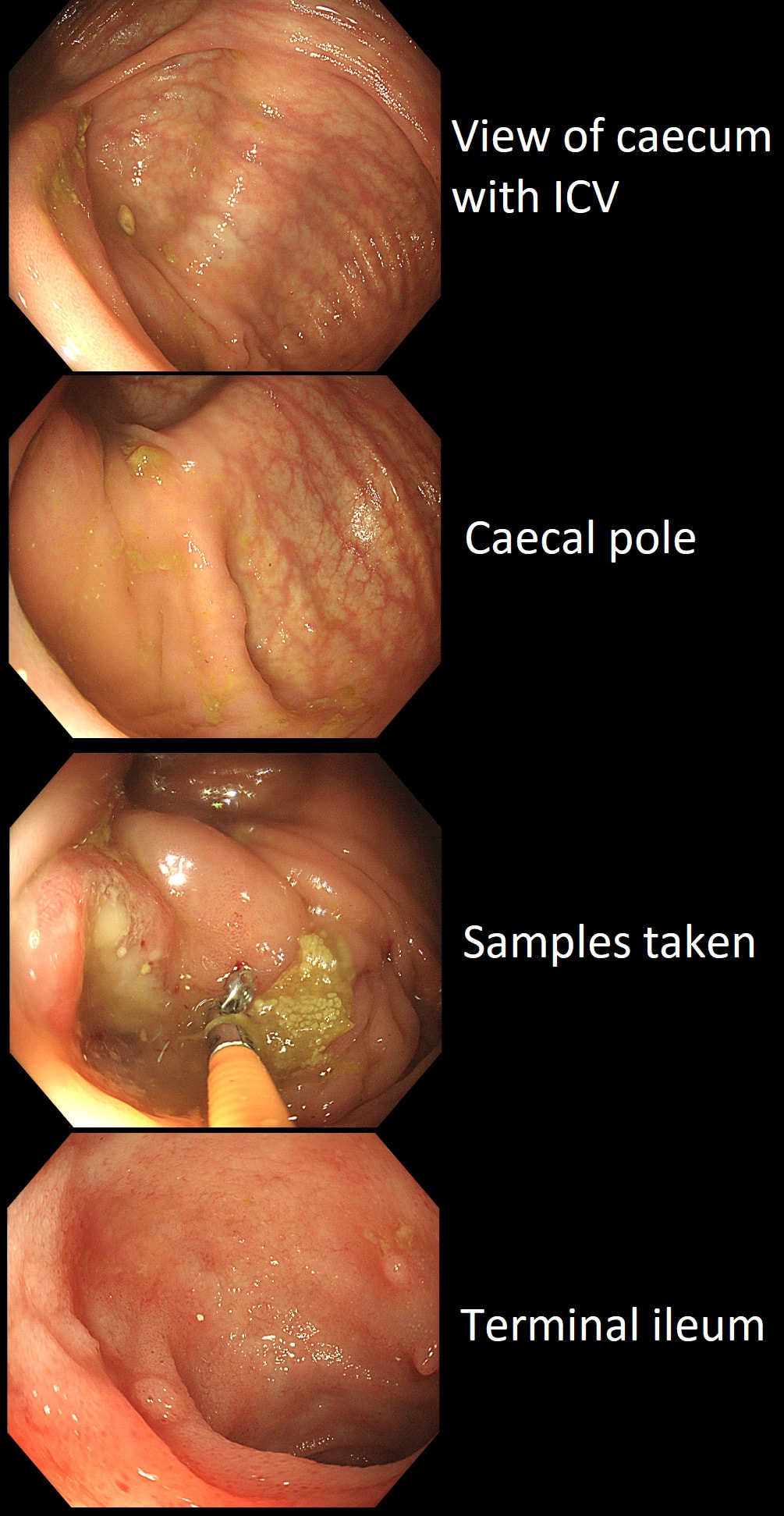
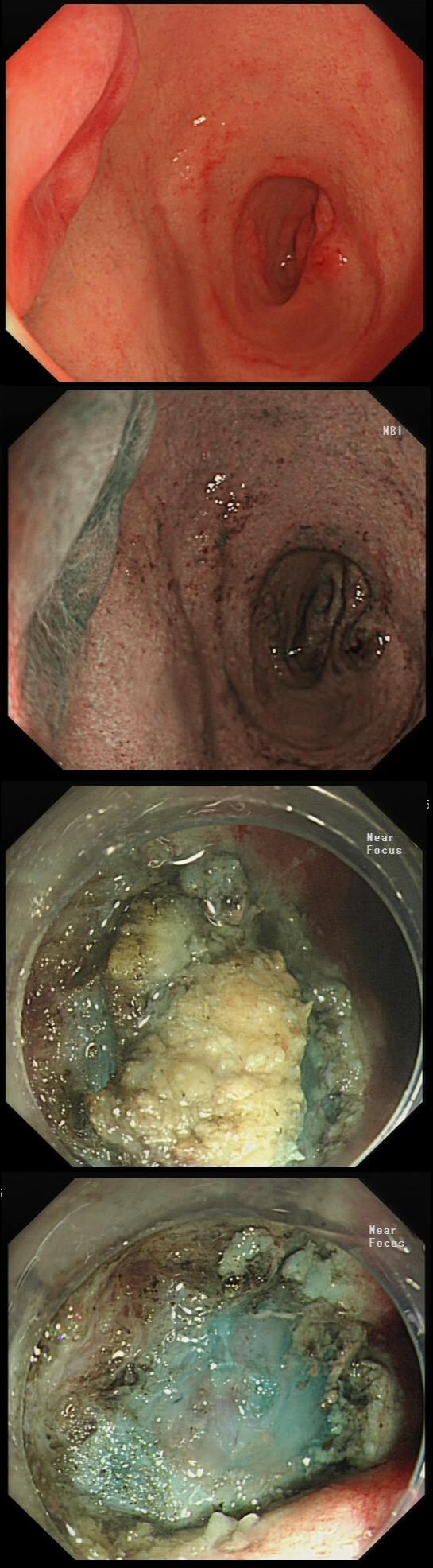
This patient underwent a CT angiogram for chest pain. The angiogram was unremarkable but the radiologist mentions an 'irregularity at the caecal pole with hyperenhancement and mild adjacent fat stranding' and recommend colonoscopy. After the examination, your patient asks you if you have found anything?
WHAT WILL YOU TELL YOUR PATIENT
a) Reassure the patient
That would be unwise!
b) Tell him to wait for the histology
Strictly speaking 'not wrong' but it's not the best option to choose
c) Tell him that we will organise an MRI scan next
MRI's are requested for rectal cancers
d) Tell him that we will organise an EUS next
Life is too short for EUS!
e) Tell him that we will organise a 'staging' chest/abdomen/pelvic CT
This is precisely what you should do!
explanation
Did you notice the lesion next to the biopsy forceps? At initial glance into the caecum, there is nothing to see. However, this lesion was about 20mm in diameter and with that rolled edge. It's clearly malignant and you need to request staging CT's. Histology did confirm a mucinous adenocarcinoma and CT sized it at 4.7cm. Far larger than initially thought! As it was involving the serosal surface, it was staged as T4, N1 (due to several small nearby nodes).
Mucinous colonic cancers are unusual, accounting for about a little more than 10% of CRC's. They are usually situated in the proximal colon. This is not the only reason why they are easy to miss. At the early stages they have an infiltrative, ulcerative growth pattern which easily hides behind bubbles or a pool. These are small but evil little things which are easy to miss and grow fast. By the way, 'signet ring adenocarcinoma' (where the mucus is INSIDE the cell rather than OUTSIDE of the cells) is another sub-type of adenocarcinoma which may be part of the same spectrum. Mucinous and signet ring adenocarcinoma, share similar molecular features such as MSI-H, CpG island methylator phenotype-high (CIMP-H), and frequent BRAF V600E mutations. Of course microsatellite instability is linked with Lynch syndrome but in this case immunohistochemistry stains revealed normal mismatch repair proteins MLH1, PMS2, MSH2 and MSH6.
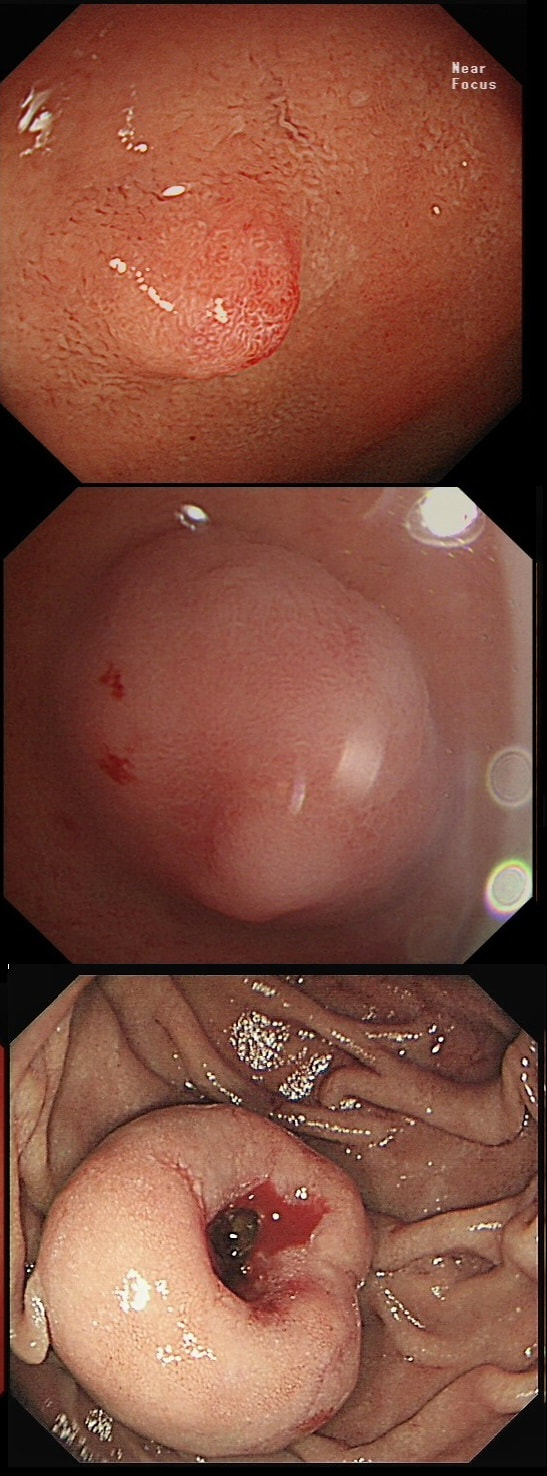
a) Reflux oesophagitis
An irregular squamo-columnar junction does suggest previous reflux oesophagitis but in this case there is more...
b) Non-dysplastic short segment Barrett's
Short-segment Barrett's is defined as Barrett's up to 3cm. In contrast, 'ultra-short Barrett's is less than 1cm. We sized the Barrett's as C0/M2 (according to the Prague classification). However, it doesn't look non-dysplastic!
c) Barrett's with intra-mucosal cancer
This would have been my own guess. There is no 'thickness' to the lesion and there is no ulceration, two features which would suggest that the lesion can't be removed endoscopically
d) Siewert II invasive cancer
Of course it may be invasive but that would be a little surprising. Endoscopically, this is IMca. Similarly HGD is unlikely as there is a lot of mucosal irregularity and you can see something has infiltrated below part of the squamous mucosa in the 2 o'clock position.
explanation
As you know, most of the time Barrett's neoplasia develops on the right-hand side and in the distal (rather than the proximal) Barrett's segment. In this case, in the 2 o'clock position there is a subtle mucosal irregularity. In fact, there appears to be something infiltrating below the squamous mucosa at this location. Biopsies had suggested IMca and a CT had been reassuring. Because the lesion is endoscopically resectable, we didn't bother with an EUS. The lesion was removed and confirmed as a poorly differentiated adenocarcinoma with signet ring morphology. On the other hand, all margins were clear and there was no LVI (lympho-vascular invasion). In the hierarchy of poor prognostic features, LVI and depth of invasion (>500µm) are the most important whilst poor differentiation is the least important. Nevertheless, I was a little worried when the patient declined surgery...
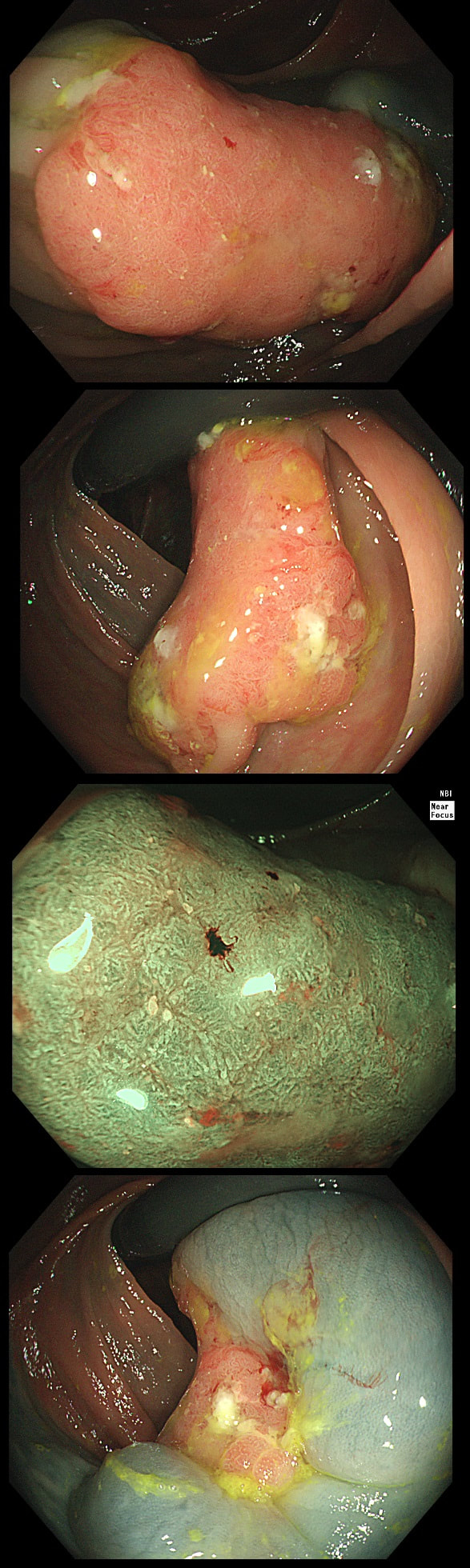
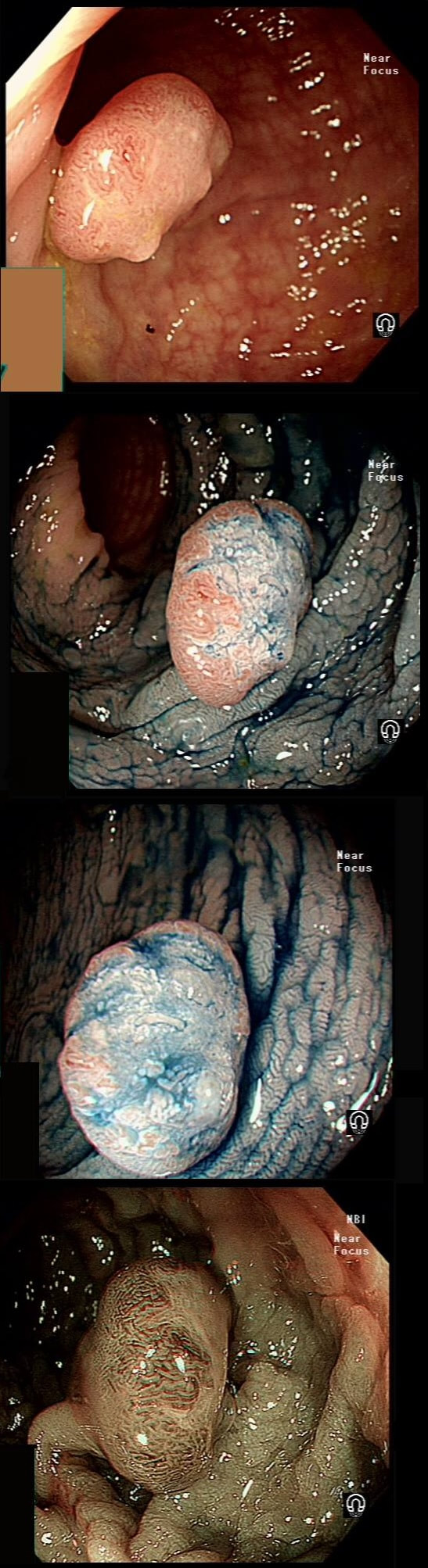
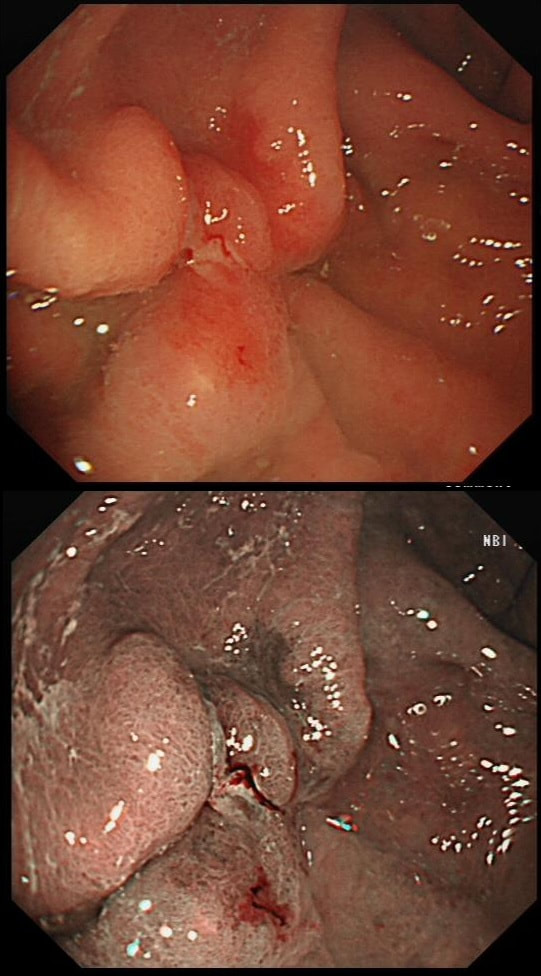
This lesion was found in the ascending colon and referred for removal.
WHAT IS THE LIKELY DIAGNOSIS?
a) HP
You must be joking!
b) SSL
You are not serious?
c) TA
Short slit-like crypts, well Yes but there is more to it!
d) TVA
Absolutely not!
e) Cancer
That non-lifting sign doesn't lie!
explanation
Must admit that I didn't like the look of this polyp. Sure, it does have an organised crypt pattern. I think that I can see short slits, making it a TA. This would fit with the fact that it probably is a LST-NG type of lesion (which are always TA's).
However, it also looks, 'chunky' and it's the 'thickness' of the lesion which made me suspicious. For this reason I was not surprised to find the non-lifting sign. As there is no lifting what-so-ever, I suspect that the lesion will turn out to be T2 at least.
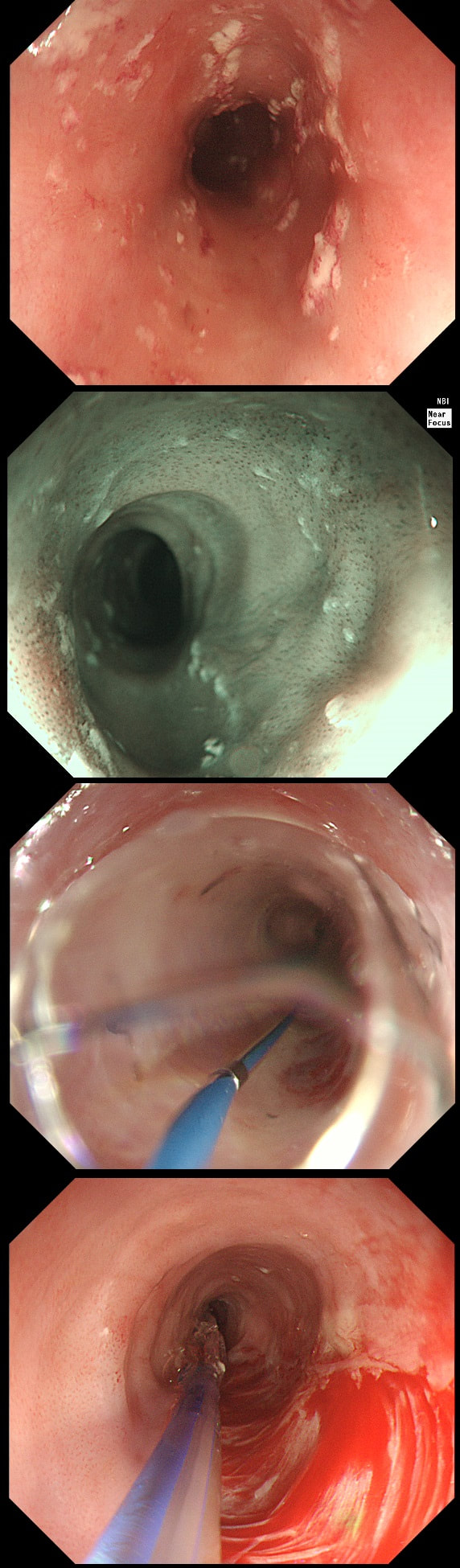
A somewhat scary dilatation.
WHAT IS THE LIKELY AETIOLOGY OF THE STRICTURE?
a) peptic stricture
There are no linear reflux ulcers!
b) eosinophilic oesophagitis
yes! Long and fusiform stricture and mucosa 'splits' in a frightening way
c) SCC
Those IPCL's do look a little prominent but not ectatic etc
d) adenocarcinoma
But the stricture is within the squamous part of the oesophagus!
explanation
Eosinophilic oesophagitis was first described in 1978 and is widely regarded as an allergic condition. Food allergy may play a significant role and the majority of sufferers have either personal or family history of other allergic conditions.
In contrast, Lymphocytic oesophagitis is much more recent, first described by Rubio et al in Sweden in 2006. As yet there is no consensus on how many lymphocytes are needed to make the diagnosis. Extrapolating from EoE, I take 2 samples from the proximal, middle and distal oesophagus respectively, in all patients with inexplicable dysphagia. LyC oesophagitis is still regarded as something of an entity "in search of a disease". For this reason it's difficult to know if the reported increasing prevalence is simply due to the pathologists being on the lookout and more oesophageal samples (correctly) being taken in patients with dysphagia. There is an association with old age, female gender, smoking, reflux disease and primary esophageal motility disorders. Clinically oesophageal rings, webs, nodularities, furrows and strictures have been described in both conditions. Personally, I think that they look subtly different! I suggest a LyC oesophagitis when there is subtle dilatation of the intrapapillary capillary loops within the squamous oesophagus. In my experience you don't usually see this in EoE. For this reason, a lymphocytic oesophagitis would have been a very reasonable guess in this case. Those IPCL's do look a little dilated don't they? Of course, strictures are supposedly far less common in LyC oesophagitis than in EoE.
Patient with Barrett's harbouring HGD has been treated with RFA. He has now returned for the second RFA session when this is found.
WHAT IS YOUR ENDOSCOPIC DIAGNOSIS?
a) Benign scarring
Doesn't look benign - Looks evil!
b) Local recurrence of non-dysplastic Barrett's
Barrett's are re-emerging in several places. Doesn't look benign!
c) Barrett's adenocarcinoma
Absolutely! Nodules are never allowed in Barrett's!
explanation
Actually, there is a re-emergence of several brown nodules below the squamous mucosa. Biopsies confirmed this as an invasive adenocarcinoma, re-emerging from below the 'neo-squamous mucosa'. Clearly, to try RFA again would be a mistake! The histology showed a 'poorly differentiated' cancer and we are recommending either surgery or chemo-radiotherapy next (CRT). Even if histology hadn't shown poor differentiation, this 'smells' like bad disease to me which we may well 'undertreat' endoscopically.
This is an interesting looking colonic polyp WHAT IS THE LIKELY HISTOLOGY
a) mixed serrated / adenomatous polyp
And that is what it looks like!!!
b) traditional serrated adenoma
But there are TWO components to this polyp!
c) serrated polyp undergoing malignant conversion
Well something has happened but 'lesion' has beautiful crypts!
explanation
Actually, the red bit of the polyp was composed of a tubulovillous adenoma and the rest was a serrated polyp! In other words, a mixed serrated/adenomatous polyp. Clearly the famous "three pathways to colorectal cancer": the APC mutation pathway (50%-70%); the mutator “Lynch syndrome” route (3%-5%); and the serrated pathway (30%-35%) can sometime get mixed up !!! WHAT IS THE MOST LIKELY DIAGNOSIS?
a) Portal hypertensive gastropathy
Doesn't really look like 'snake skin' does it?
b) HP associated gastritis
HPAG is a great mimicker but not this time
c) Gastric vascular ectasia
Absolutely, affecting the whole stomach!
d) Lymphocytic gastritis
LyC gastritis is ulcerative!
e) Linitis plastica
Another great mimicker but this is a vascular issue rather than mucosal infiltration!
explanation
Of course, this is a case of Gastric Vascular Ectasia. As the whole of the stomach is affected, I don't think that we can call it 'antral' ectasia. Gastric antral vascular ectasia “GAVE” (watermelon stomach) has been described with; • Cirrhosis • Atrophic gastritis/achlorhydria/hypergastrinaemia • Connective tissue diseases (especially systemic sclerosis and hypothyroidism) • Diabetes • Heart failure • Chronic renal failure It has been proposed that the underlying link between all the different conditions associated with GAVE may have autonomic dysfunction in common leading to prolapse of the antral mucosa through the pylorus. Must admit that I'm sceptical! A study suggested that a “punctuate pattern”, as seen in this clip, is typical of cirrhosis whilst a “striped type” was more common in non-cirrhotic cases. Indeed this patient had NASH cirrhosis. Portal hypertensive gastropathy probably has a different aetiology and is a different condition as gastric antral vascular ectasia can develop with a normal portal pressure. Patients with vascular ectasia of course usually present with iron deficiency anaemia. Patients with bleeding from GAVE were previously treated with antrectomy. Now, the first line treatment is “thermal therapy”. Octeotride and oestrogen-progesterone have also been used in small series but would now only be considered if endoscopic therapy fails. It is far easier to treat these lesions using the argon coagulator than the heater probe. Using a banding device may be quicker in patients who tolerate gastroscopies poorly. RFA is also effective but is VERY expensive. This patient attends for Barrett's surveillance WHAT IS YOUR DIAGNOSIS?
a) Barrett's without dysplasia
A nodule within Barrett's is NEVER 'normal'!
b) LGD
LGD is flat, not nodular!
c) HGD
Good guess! Although your guess actually proved to be wrong
d) IMca
Small crypts + nodule = correct!
e) invasive cancer
Nodule should have been larger and without those small crypt openings
explanation
My 'rules of thumb' when assessing Barrett's is as follows: 1) crypts look different in a distinct patch but the area is flat = LGD 2) areas of superficial ulcers within the Barrett's = widespread LGD 3) crypts look different and there is a distinct nodule = HGD 4) crypts are tiny small and there is a nodule = IMca 5) there are no crypts and there is larger nodule = invasive cancer Of course this isn't fool proof ! After all, it's very difficult to distinguish HGD from IMca even on histology! However, it gives you a starting point on how to assess Barrett's and what you should enter on that Histopathology request form. Remember that your pathologists need your help! This lesion was found at gastroscopy WHAT IS YOUR ENDOSCOPIC DIAGNOSIS?
a) Siewert I SCC
Nope!
b) Siewert I adenocarcinoma
Lesion isn't centred 1-5cm above the GOJ
c) Siewert II adenocarcinoma
Yes! Lesion staged as T2,N0 on PET-CT
d) Siewert III adenocarcinoma
Lesion isn't centred 2-5cm below the GOJ
explanation
This lesion is clearly malignant with a rolled edge and surrounding mucosal ulceration. It was confirmed as a Siewert II adenocarcinoma, T2,N0. Cancers at this location are becoming more common. They are easy to miss, particularly if you don't slow down as you traverse the gastro-oesophageal junction (GOJ) and/or retrovert at a distance. On retrovertion you need to pull the scope back up so that you can have a close view of the cardia. Siewert et.al. developed the classification as follows:
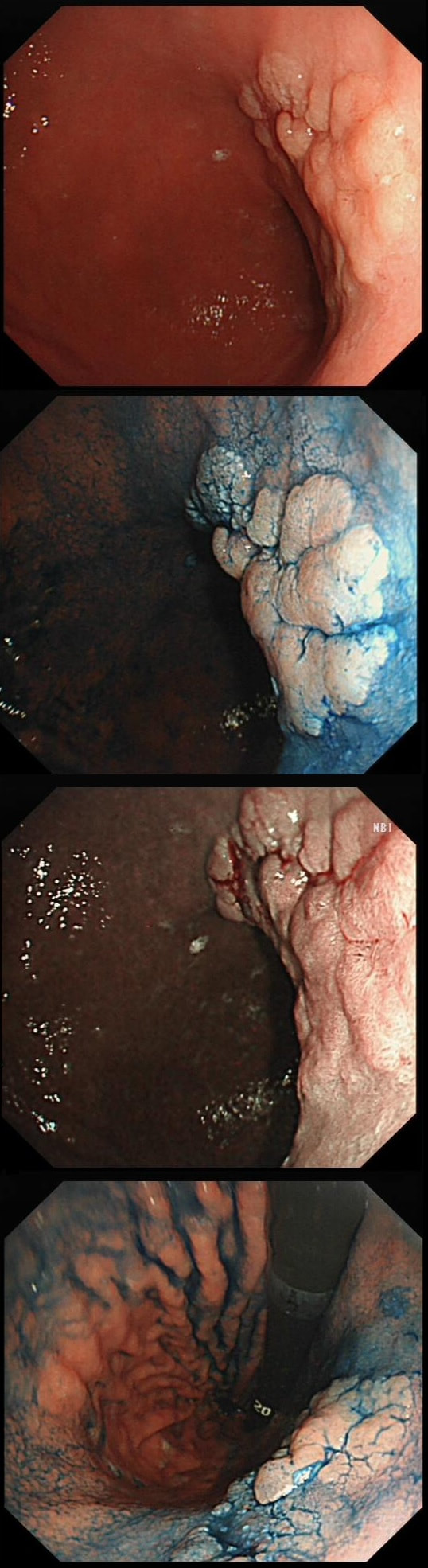
Of course it can be difficult to determine where the epicentre of a cancer is, particularly on imaging. At endoscopy you are in a unique position to accurately record the correct Siewert type of the cancer. It makes a difference because the Siewert type has implication for treatment! Siewert I lesions are treated with oesophagectomy and gastrectomy as these lesions usually metastasise to nodes in the mediastinum. Siewert II are 'true' junctional cancers and mainly metastasise to nodes in the abdominal nodes but in around 15-20% of cases, there are mediastinal nodes. For this reason, patients with Siewert II lesions are only offered gastroectomy (without oesophagectomy) IF there is no mediastinal lymphadenopathy. Arguably, any borderline mediastinal nodes should probably be sampled before or during surgery before a final decision is made not to clear the mediastinal nodes with the resection specimen. Patients with Siewert III cancers are usually offered total gastrectomy plus a distal esophagectomy (to get clear resection margins, a so called R0 resection ) as these lesions spread to peritoneal nodes. This is a video clip of a small lesion removed from the sigmoid colon. WHAT IS THE MOST LIKELY HISTOLOGY?
a) TA+LGD
But it's a IIc lesion with IIIs crypts in the centre?!?
b) TA+HGD
Absolutely!
c) Invasive cancer
But there ARE small round crypts in the centre and it DOES lift!!!
explanation
You may call this a "flat elevated lesion with a central depression (IIa+IIc lesion) or simply a depressed lesion (IIc lesion). Frankly it doesn't matter because both a part of the same 'family' of evil little b.....ds. They are always TA's and the small, round crypts (Kudo type IIIs crypt pattern) tells you that the lesion harbours HGD. This is because as dysplasia progresses from low to high grade, crypts get smaller and more withered. Of course they eventually disappear altogether as the lesion develops into a cancer which no longer follows any 'instructions' to form organised crypts. However, the crypt pattern is still discernible in the centre AND the lesion lifts well. Both of these tells you that the lesion is likely to still be benign. Ultimately, the pathologists called it a TA+HGD. However, there was mucinous differentiation in the centre of the lesion. Could these little shits be the early stage of mucinous colonic cancers? Quite likely! Imagine how easily they are missed when hiding behind a fold or below a shallow puddle !!! WHERE IS THE BARRETT'S CANCER?
a) 12 O'clock
Eagle eye!
b) 3 O'clock
But the round crypt openings say otherwise!
c) 6 O'clock
Nope!
d) 9 O'clock
Where there is hardly any Barrett's?!
explanation
Of course the lesion is situated at 12 O'clock. There you can see a subtle mucosal nodularity with an irregular vessel pattern. It was removed by 'suck within the cap' EMR and confirmed as an IMca. This lesion is being removed from the distal oesophagus WHAT IS THE LIKELY HISTOLOGY?
a) Hyperplastic polyp
You must be joking!
b) Adenomatous polyp
A gastric adenomatous polyp would surely now be malignant
c) Early malignant polyp
But it's arising from a malignant flat component!!!
d) Advanced gastric cancer
Absolutely!
explanation
Of course this is all very odd. Clearly this is an advanced cancer at the GOJ. What business do I have in 'attacking' this endoscopically?! Actually, the elderly patient had completed a course of chemoradiotherapy (CRT) for a T2, N0 junctional adenocarcinoma some 30 months previously. Now he has developed dysphagia and a CT confirmed a 2cm polyp at the gastro-oesophageal junction. Histology had shown 'at least IMca' and he was referred for consideration of an endoscopic resection. Clearly this lesion can't be cured endoscopically. In fact, the elderly patient is not a candidate for surgery and therefore there is no cure at all. However, I was thinking that as the cancer is mainly polypoidal, perhaps if the nodule could be removed, his swallowing will improve and he will not need a stent and could be offered brachytherapy. Clearly this was all 'speculative' but I'm glad to say that 6 months later, the patient still has not developed any dysphagia and his now starting brachytherapy. No doubt a better outcome than could be offered by a stent? This is a solitary gastric polyp WHAT IS YOUR ENDOSCOPIC DIAGNOSIS?
a) Hyperplastic polyp
Spot on!
b) Hamartomatous polyp
A bold guess but wrong!
c) Cystic fundic polyp
Too red and should be several of them!
d) Adenomatous polyp
But there is no defined crypt pattern?!
e) Malignant polyp
May be a 5% chance that you are right!
explanation
A missing piece of information, which I perhaps should have provided, was the H.pylori status of this patient! This lesion was arising from a H.pylori associated gastritis.
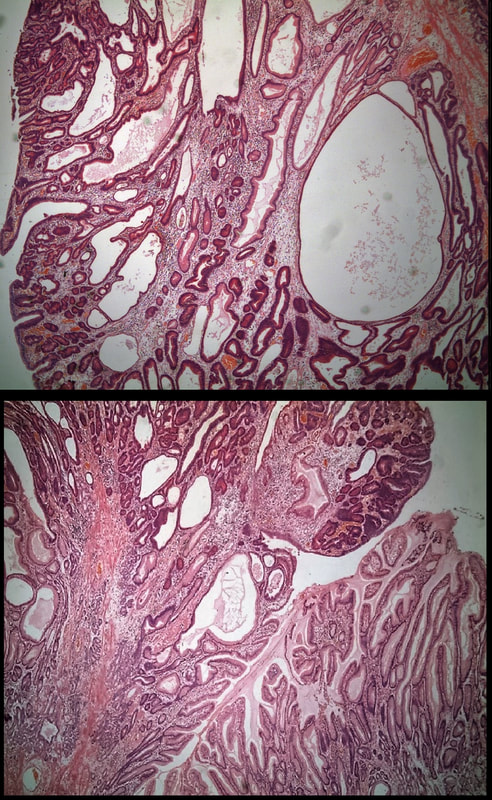
Most polyps found in a stomach with Helicobacter pylori associated gastritis are hyperplastic (inflammatory). They appear angrily red (because they have lots of capillaries) and often with white fibrin caps, making them look a little like mushrooms. A small proportion of hyperplastic looking polyps are actually malignant. In my experience, these 'stealth cancers' are most common close to the gastric cardia and are always solitary. Conversely, multiple inflammatory polyps in the antrum and gastric body are almost certainly benign. Of course, this is a 'solitary' hyperplastic polyp, close to the fundus. Perhaps not unexpectedly it harboured some (low grade) dysplasia (histology below). The dysplasia is in the crypts lined by deep purple/blue cells rather than the normal light pink cells. The contrast is probably best seen in the second histological slide where the dysplastic part is towards the top of the slide and the non-dysplastic is at the bottom half. If the H.pylori test is negative, and particularly if the patient is taking a PPI, the polyp is more likely to be a 'Fundic gland polyp' (cystic fundic polyp) which are full of cystic spaces and therefore look a little translucent like frog-spawn. Rarely polyps are hamartomatous as in Peutz-Jeghers polyps and the polyps arising in patients with 'Cronkhite-Canada syndrome or in Juvenile polyposis. In many cases these lesions have an odd but distinct surface crypt pattern or, alternatively look again look translucent like frogspawn. It's rather difficult to explain the appearance of a hamartomatous polyp and for this reason, I have attached some images below (after the histology slides). Another polyp, most likely to arise in an atrophic gastritis with patches of intestinal metaplasia, is an adenomatous polyp of the 'intestinal type'. To remind you, there are at least 4 types of gastric adenomas which all have an organised and regular surface crypt pattern:
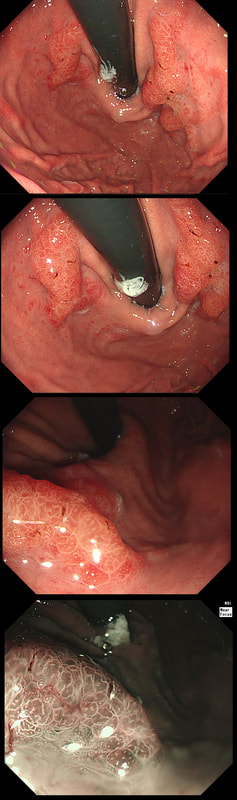
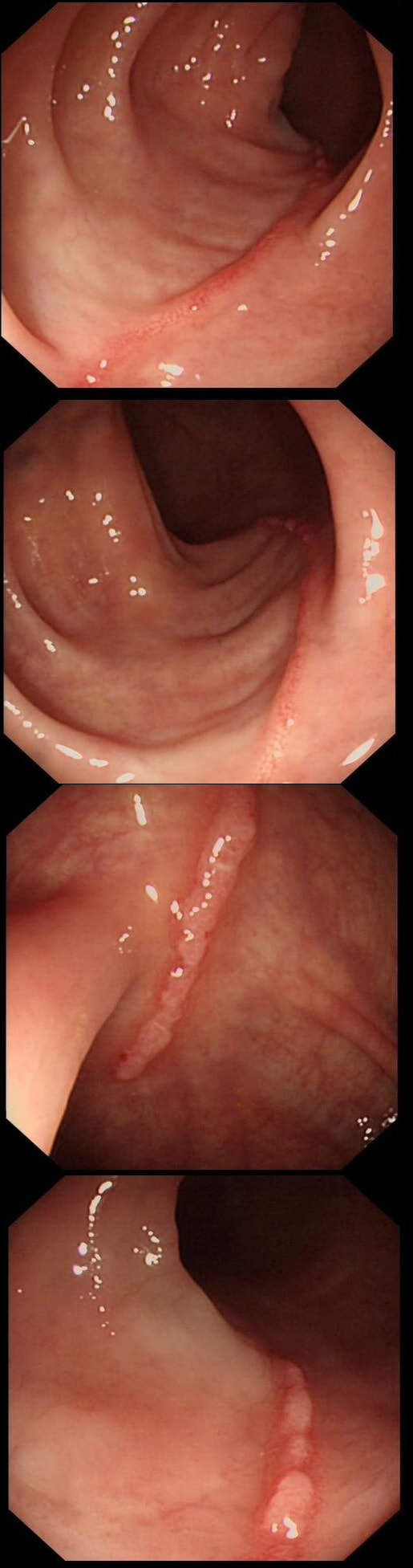
These linear lesions were found in a 50 yr old patient undergoing colonoscopy to investigate her loose stool.
WHAT IS THE LIKELY DIAGNOSIS?
a) Crohn's disease
Does give linear ulcers but should be larger and deeper!
b) Ischaemic colitis
Also gives linear ulceration, often on the ante-mesenteric border.
c) Collagenous colitis
Absolutely!
d) Ulcerative colitis
Doesn't give linear ulceration and surrounding mucosa wouldn't be normal
e) Lymphocytic colitis
Perhaps one could develop into the other?
explanation
Actually, this patient had collagenous colitis! I suspect that the acute injury are linear tears in the mucosa which then granulate as in the images above. Finally, you are left with linear scars as in the image below. Pure speculation but seems logical ! At colonoscopy, the mucosa is often unremarkable but there may also be mild, patchy erythema or linear cracks. Histology reveals the typical thickening of the subepithelial collagen layer from the normal 3-6 μm to more than 10 μm as well as lymphocytic infiltration of the epithelium and the lamina propria. Rectal biopsies are not sufficient to make the diagnosis as normally the collagen layer is particularly thin here. Samples from the rectum and sigmoid confirms the diagnosis is more than 90% of cases. Interestingly, patients with eosinophilic oesophagitis may also develop some fibrosis in the lamina propria which also 'cracks' in a spectacular way when a dilatation is carried out.
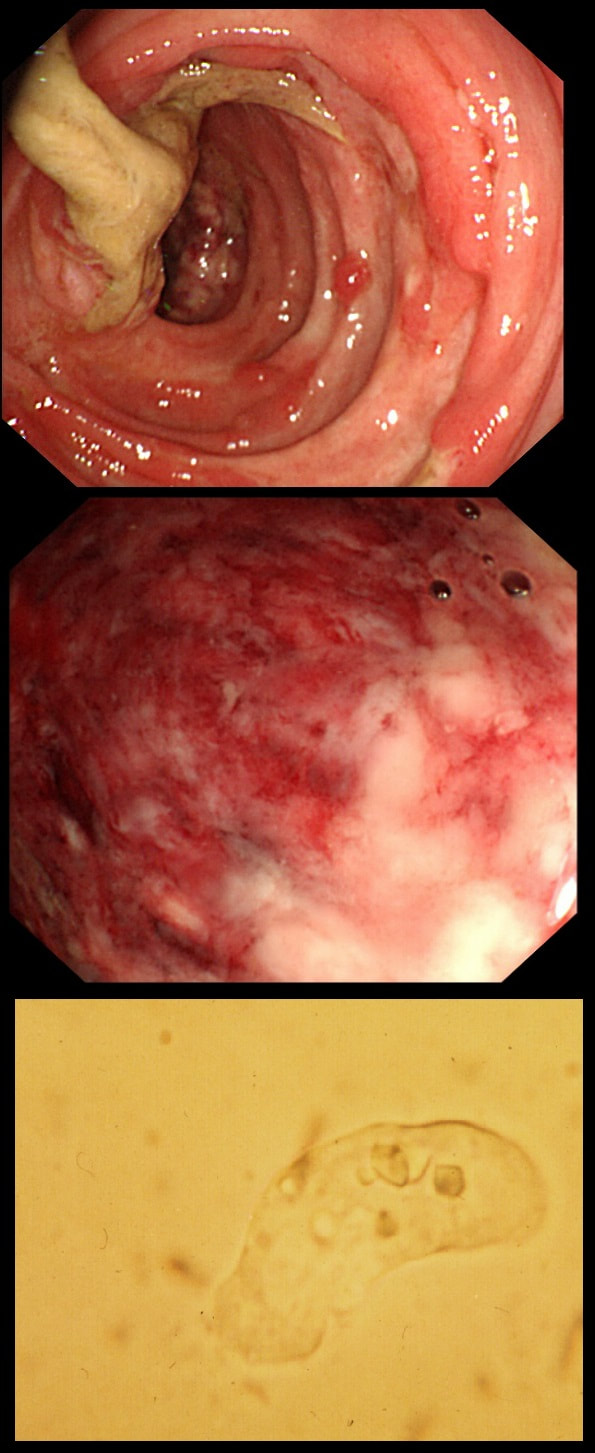
This patient developed bloody stool whilst on holiday in Africa. They continue on return home and he undergo a flexible sigmoidoscopy. Of course, stool samples are obtained (last image).
WHAT IS THE PATHOGEN?
a) Clostridium difficile
Can be seen moving down a microscope
b) Salmonella enterica
Can give bloody diarrhoea but its not it!
c) Entamoeba histiolytica
Rasberry stool, to be examined warm
d) Campylobacter jejuni
May also give bloody stool but that's not it
e) Vibrio cholerae
Hmm, rice-water stool...
explanation
Let me give you some more clues! Classically, there are 'flask-shaped' ulcers found on resectional histology in which the mucosal surface ulcer is rather narrow with a wider, necrotic submucosal component in which the amoeba multiply. Stools have been described as like "Raspberry jam" and when examined whilst still warm, something can be seen moving. Of course PCR is the best way of confirming the diagnosis.
This patient had amoebic dysentery! Mild cases can resolve without metronidazole.
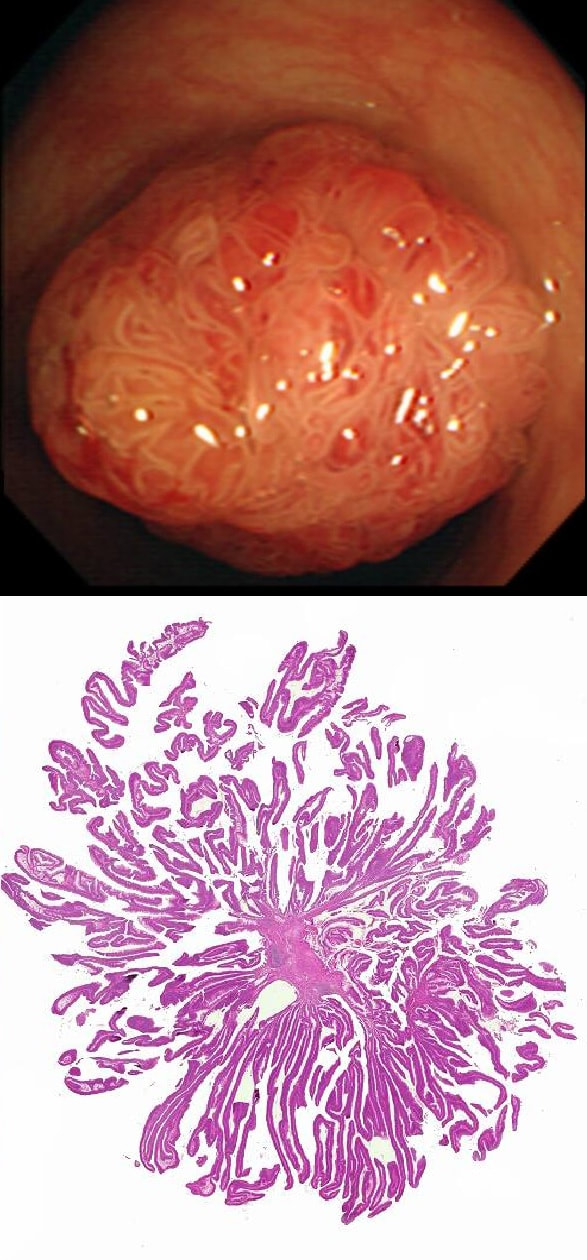
This polyp is pretty when viewed by the endoscopy but stunning below the microscope!
WHAT IS THE HISTOLOGY?
a) Tubular adenoma (TA)
This polyp is prettier than that!
b) Tubulovillous adenoma (TVA)
Endoscopically it could be but histology says otherwise!
c) Villous adenoma (VA)
Absolutely!
d) Traditional Serrated Adenoma (TSA)
Endoscopically Yes, but histologically No!
e) Sessile Serrated Lesion (SSL)
Would be unusal for an SSL to be 'subpedunculated'
explanation
Endoscopically it can be difficult to recognise a villous adenoma. Unless you submerge the lesion to see the villi rise up like the arms of a sea anemone. Apparently, they are quite difficult to 'process' by the pathology lab as well. They are fragile and those beautiful villi, easily become damaged.
Of course, villous adenomas are regarded as 'high risk lesions'. The other high risk findings often used as surrogate markers for cancer in guidelines are: having 3 or more adenomas, polyps 10mm or larger, and polyps harbouring high-grade dysplasia. However, if you have to make a choice of the two strongest predictors of future cancer risk, a study in Gastroenterology found that it would be; finding a polyp 2cm or larger and resecting a polyp found to harbour HGD [Gastroenterology 2019;158(4);875-83].
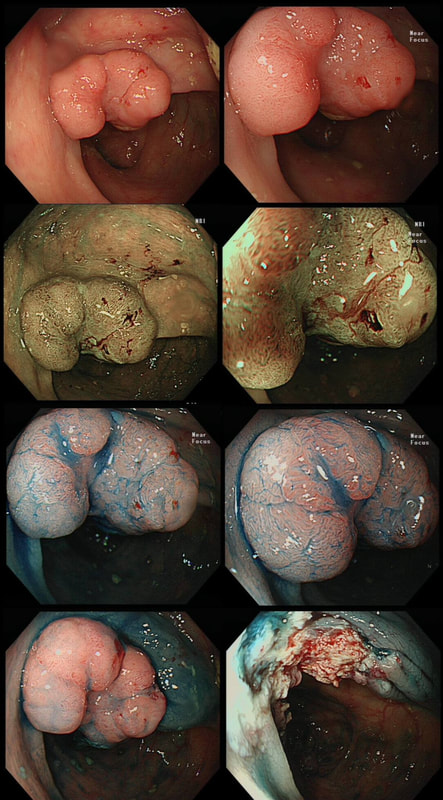
This colonic polyp was removed as a single fragment from a 60 year old lady. You can see the mucosal defect in the last image. The patient asks you what will happen next?
WHAT WILL YOU REPLY?
a) Can't tell, we will have to wait for the histology
You can do better!
b) In all likelihood there will be a site-check in 4 month or so
You are missing something!
d) You will probably need an operation
You've spotted the desmoplastic reaction!
explanation
The polyp looks very suspicious but did seem to lift and I therefore decided to go ahead and removed it using a stiff, large snare. It took a little longer than expected for the snare to cut through. Of course, the mucosal defect should be blue. In this case it's yellow! The polyp was malignant, invading about 1mm into the submucosa and you are looking at the 'desmoplastic' reaction generated by the cancer.
Apart from the sometimes deceptive 'non-lifting sign', there are two further signs that a lesion may be malignant. First, it may look smaller and smaller as you inject below the lesion (see example below). Another sign is that your blue sub-mucosal injection appears to lift the lesion until you retrovert and have a look at the other side. If you then find that it hasn't actually 'crossed the mid-line', there is fibrosis below the lesion preventing the fluid to disperse evenly. I was not entirely surprised to learn that the patient declined surgery. After all, he was 86 years old! He lived another 7 years and never developed any sign of bowel cancer. By the way, there is a theoretical risk of tumour seeding if the lesion is perforated during resection. However, when the perforation is done with a red-hot tool such as a knife or snare, the risk of seeding is surprisingly low. I have perforated a handful of cancers but have never had a case of late disseminated peritoneal disease. My Japanese colleagues (off the record), agree that the risk is there (some have seen it) but is low. If you decide to sample a suspicious looking polyp, you shouldn't use the same forceps to sample another lesion. This is because if cancer cells become lodged in the biopsy forceps, which are then used to sample another location and them become stuck in the biopsy, the histopathologist will diagnose cancer in TWO locations when in fact, there is only a single cancer.
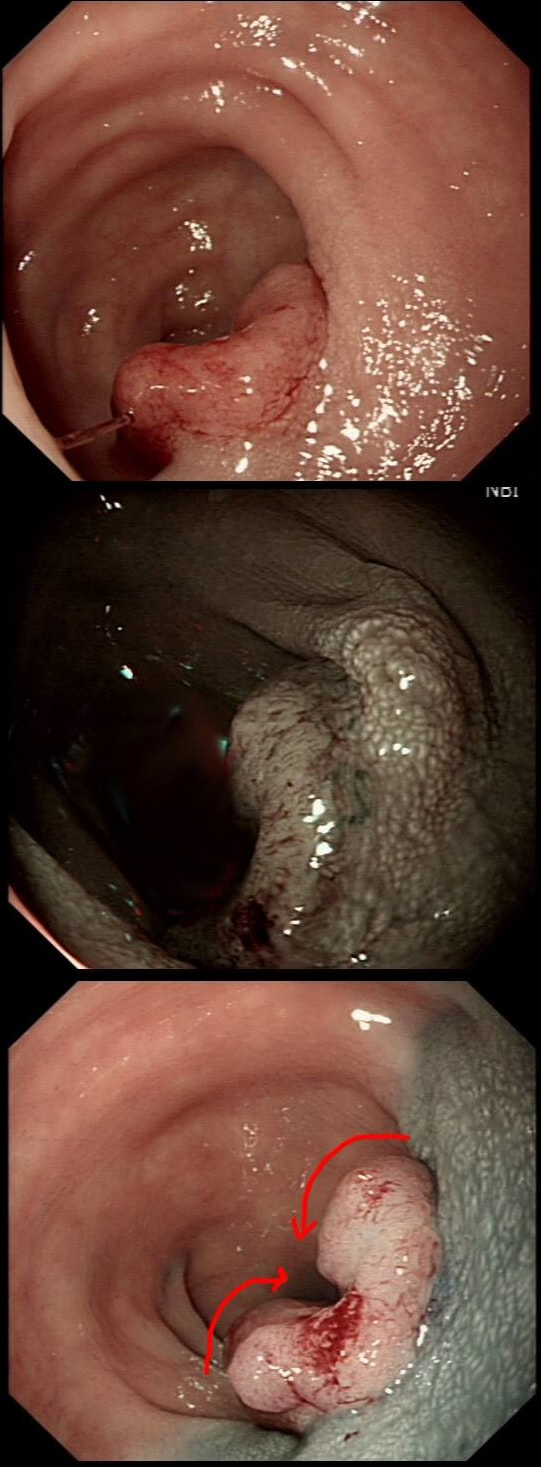
This patient with ulcerative colitis has developed a polyp in the transverse colon. The lesion has now been sent for an endoscopic resection.
WHAT WOULD YOU DO?
a) Abort!
Smart!
b) Attack!
You are creating a problem of your own making!
explanation
Some would say; "if you can remove the lesion in that colitic colon, then 'do it'! The problem is that nothing may appear "irresectable" giving plenty of time, determination and poor judgement.
Many studies looking at outcomes of polypectomy in UC, excluded polyps >1-2cm or flat polyps. Other studies have included polyps arising outside of the colitic field or only have a short follow up period of a few years. Actually, most are coming to believe that when dysplasia develops in the colitic colon, it's not a 'random' case of bad luck. It can be the result of a long process of progressive DNA damage! At some stage we will be able to have a look at the state of the stem cell DNA in patients with conditions such as Barrett's, Colitis and atrophic gastritis. I think that we are in for a surprise ! In addition, did you spot the small focus of invasive cancer in the 2-3 O'clock position? Surprisingly, this was only T1 disease!
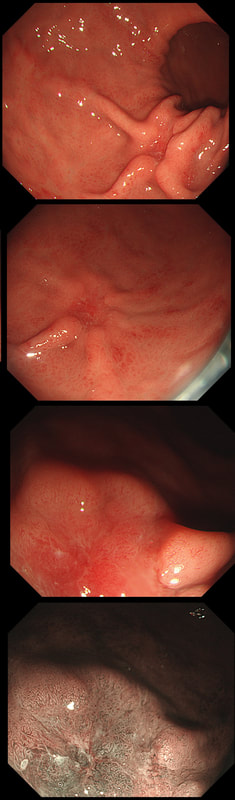
A polyp found in the transverse colon
WHAT IS YOUR ENDOSCOPIC DIAGNOSIS?
a) Tubular adenoma
You are half correct!
b) Tubulovillous adenoma
On the left side, the crypt's are slit-like rather than gyrate
c) Villous adenoma
VA's are soft like sea anenome !
d) Serrated polyp
No, the crypts are not large, round openings!
e) Malignant polyp
Yes, there are no crypts on the right-hand side !
explanation
Did you spot that cancer has developed on the right side of this tubular adenoma (TA). On the left side, the crypts are slit-like whilst on the right hand side, the crypt pattern has been destroyed (Kudo type V crypt pattern). The Haggitt stage was I and I took particular care in removing the polyp with a large margin. The cancer was only 4mm in diameter. Perhaps the smallest adenocarcinoma of the colon you will ever see ?
This polyp found on the lesser gastric curve.
WHAT IS THE MOST LIKELY DIAGNOSIS?
a) Hyperplastic polyp
HP polyps are usually subpedunculated and angry red
b) Intestinal metaplasia
IM is also 'pale' but not 'flat elevated'
c) Gastric adenoma
Would be my first guess!
d) Early invasive cancer
This is a plaque without a depression or dominant nodule to suggest invasive cancer
explanation
Of course, this could have been rather easy to miss! An insignificant pale, plaque-like lesion but with distinct borders best seen after indigo carmine dye spray (as usual). Endoscopically this is what a gastric adenoma looks like and it was removed endoscopically. Somewhat surprisingly, histology reported a small spot of IMca and therefore its at actually an early cancer - not invasive though!
Most invasive, gastric EGC's are of course shallow depressions as in the example below. In fact the lesion below was invading into the middle of submucosa and was confirmed as poorly differentiated. Afterwards the patient underwent a distal gastrectomy where no further cancer was found. 'Poor differentiation' is the least reliable and lymphovascular invasion (LVI) is the most reliable predictor of nearby lymphnode micro-metastases.
This polyp on a short stalk was removed from the colon
WHAT IS THE LIKELY HISTOLOGY?
a) TA
At the edge, the crypts are clearly sli-like but how about the centre?
b) TVA
Doesn't look like gyrate crypts!
c) VA
Doesn't look like a sea anenome!
d) TSA
TSA's do have crypts but this lesion doesn't in the centre!
e) Early cancer
Was my own firm diagnosis!
explanation
The head of the lesion is clearly of concern. There are 'horns' on it! Of course, the devil has horns but as it was arising from a stumpy stalk. I went ahead and removed it. Somewhat surprising our pathologists called the lesion TA+HGD!
Admittedly, there was disagreement between our histopathologists and 2 out of 5 believed that it was an invasive cancer. Endoscopically, the lesion is clearly malignant and at the very least it's an intramucosal cancer (IMca). Of course, our UK histopathologists are unable to make the diagnosis of Intramucosal cancer in the colorectum because this is not a diagnosis recognised by the 'Vienna classification'. Elsewhere in the GI tract, intramucosal cancer is a diagnosis which our pathologists are 'free' to make. It makes no sense whatsoever to me !
This lesion was referred for resection just beyond the pyloric ring. It seemed to lift well but rather to my surprise, the snare cut through the base of the lesion (bottom image on the left). It took some further lift and a stiff snare to scrape the lesion of the muscle propria layer.
WHAT IS THE LIKELY HISTOLOGY?
■ Adenoma
Adenomas are usually not umbilicated ...
■ Brunners gland hyperplasia
I don't bother to remove those and they are not umbilicated!
■ Gastric heterotopia
Is usually found at this location but never require EMR
■ NET
Of course it is - it's umbilicated !!!
■ Early malignancy
Would be VERY rare in the duodenal cap!
explanation
The lesion is umbilicated... As you probably remember, umbilicated lesions in the duodenum are usually NET's. For some reason, they look different to NET's found elsewhere in the gut, where thin vessels are usually seen crawling up their sides. In the photograph below, you can see that at the very earliest stage (first image), there is no central indentation. However, very soon a subtle central dip develops (second image) which at the advanced stage (when I was tasked with removing the lesion), it has a deep central pit which was intermittently bleeding.
This proved to be a WHO grade I NET (neuroendocrine tumour, with a mitotic rate <2%). These lesions always seem to be tethered down close to the muscle propria and can be difficult to remove without taking a chunk of muscle propria with it. Duodenal NET's should of course be discussed at the 'neuroendocrine MDT' and usually end up having: 1) Measurement of plasma chromogranin A (pCgA) levels. CgA is produced by all cells derived from the neural crest and high levels are found particularly in patients with metastatic disease. 2) A ‘gut hormone screen’ (measurement of gastrin, glucagon, vasointestinal peptide, somatostatin and pancreatic polypeptide levels). 3) Finally, an ‘octreotide scan’ (somatostatin receptor scintigraphy) or, even better, a PET/CT scan using peptides that bind to somatostatin receptors (68Gallium-DOTA-TOC/NOC/TATE).
This was found on the anterior gastric wall in an elderly patient with iron deficiency anaemia
WHAT IS THE MOST LIKELY DIAGNOSIS?
■ Gastric ulcer scar
Quite possible but you would take samples surely?
■ Gastric erosion
But there is a nodule in the centre?
■ Benign gastric ulcer
But there is no 'ulcer' in the centre?!
■ Early gastric cancer
Surely, the most likely diagnosis?
explanation
Gastric folds are being pulled into this 'lesion'. Of course most EGC's are depressed or flat elevated with a central depression. This is not easy to classify into the Paris classification system. I guess that you can argue that its' an elevated lesion with a central depression? Anyway, at the centre of the lesion there is a nodule! Not an ulcer! It's most likely an EGC. In fact, the lesion was resected endoscopically and proved to be poorly differentiated invasive cancer with a positive deep margin ! As the patient was 84 yrs old, no surgery was offered. That was 6 years ago and the patient remains well with only an unremarkable scar in the stomach to remind him ! Wonderful !
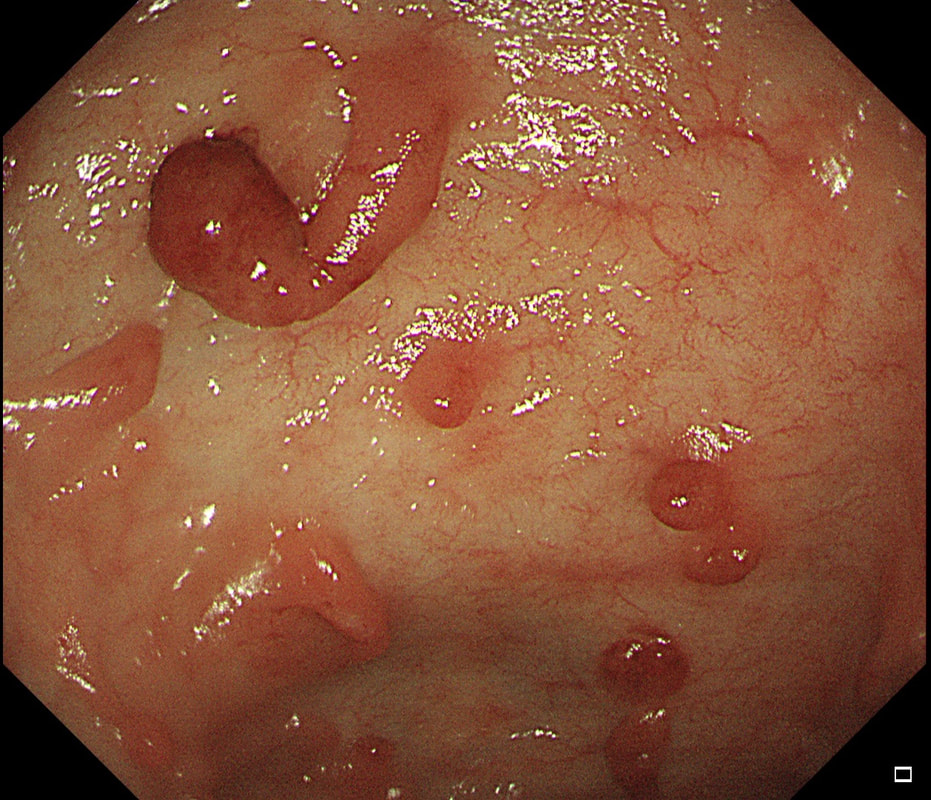
These peculiar colonic lesions were found in the colon of an asymptomatic patient.
WHAT ARE THEY?
■ Inflammatory polyps
Absolutely!
■ Adenomatous polyps
These are too long and peculiar looking to be adenomatous!
■ Hamartomatous polyps
No way!
|
Categories
All
|