|
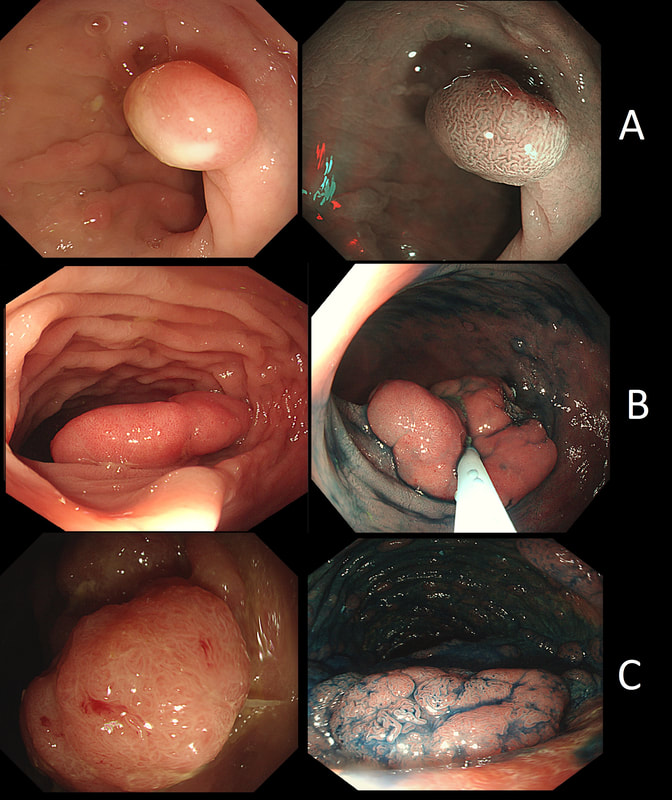
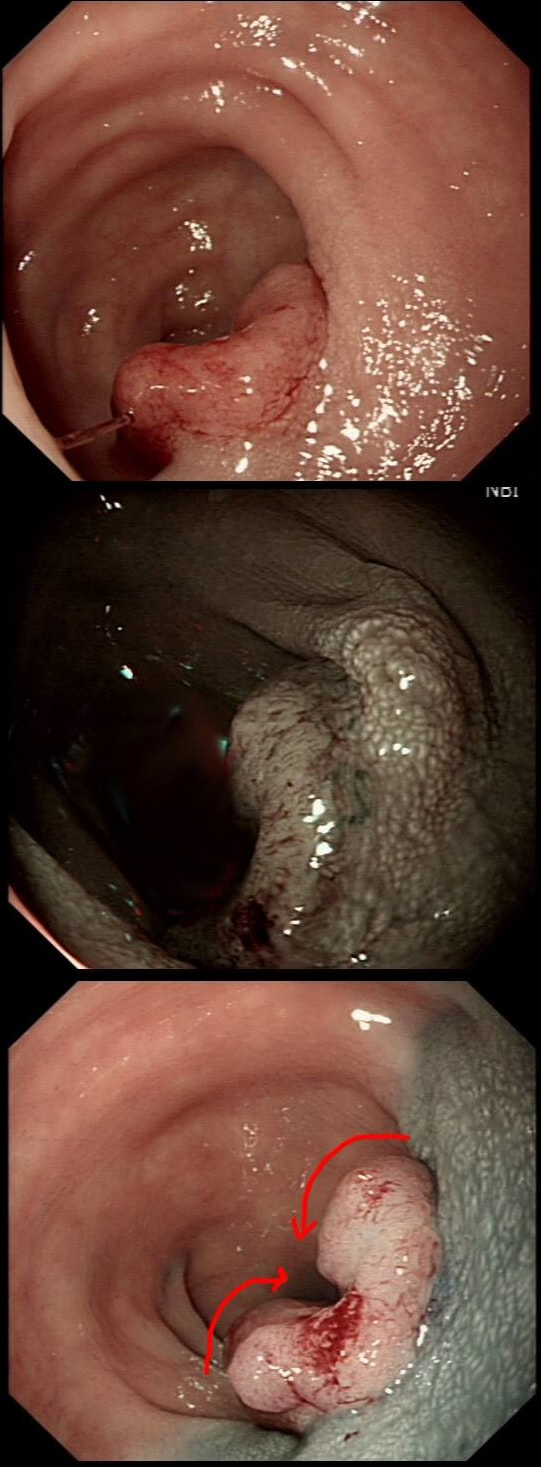
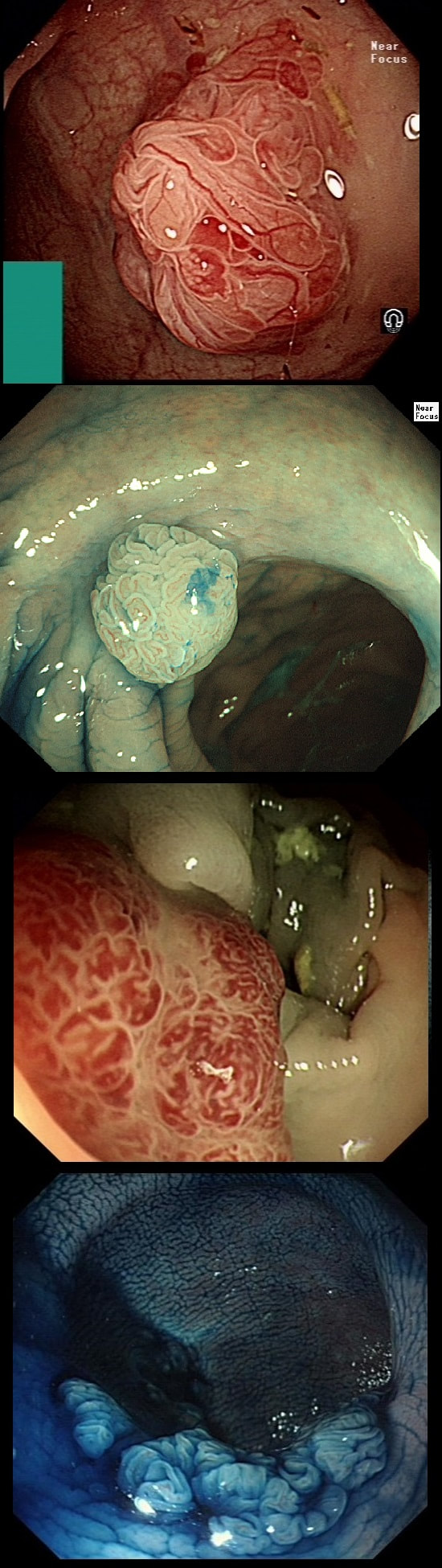
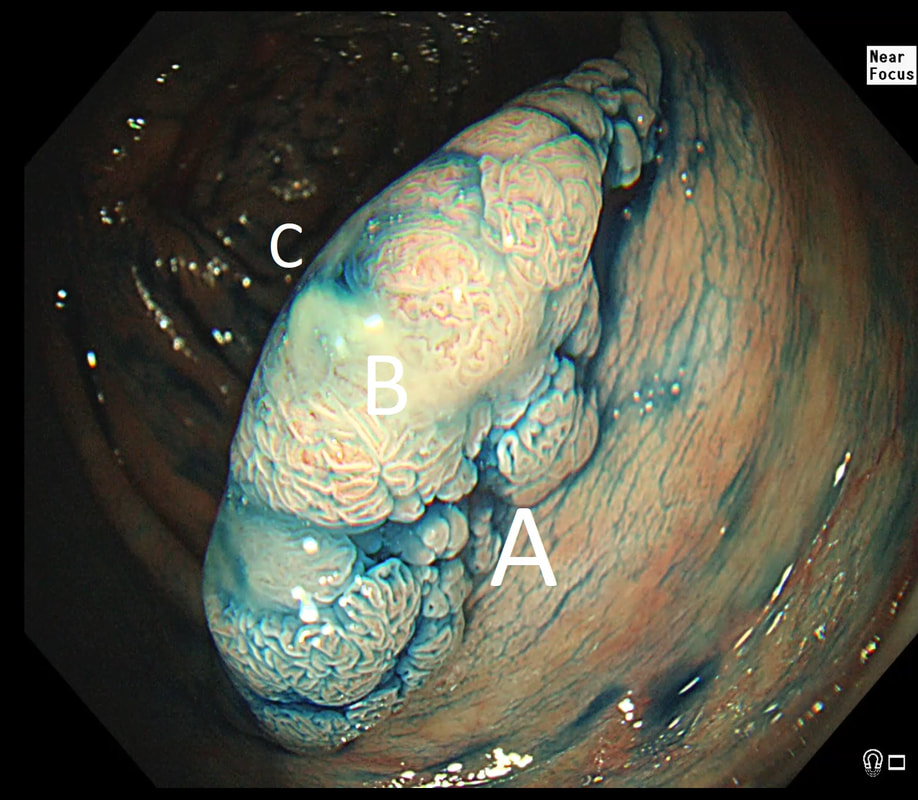
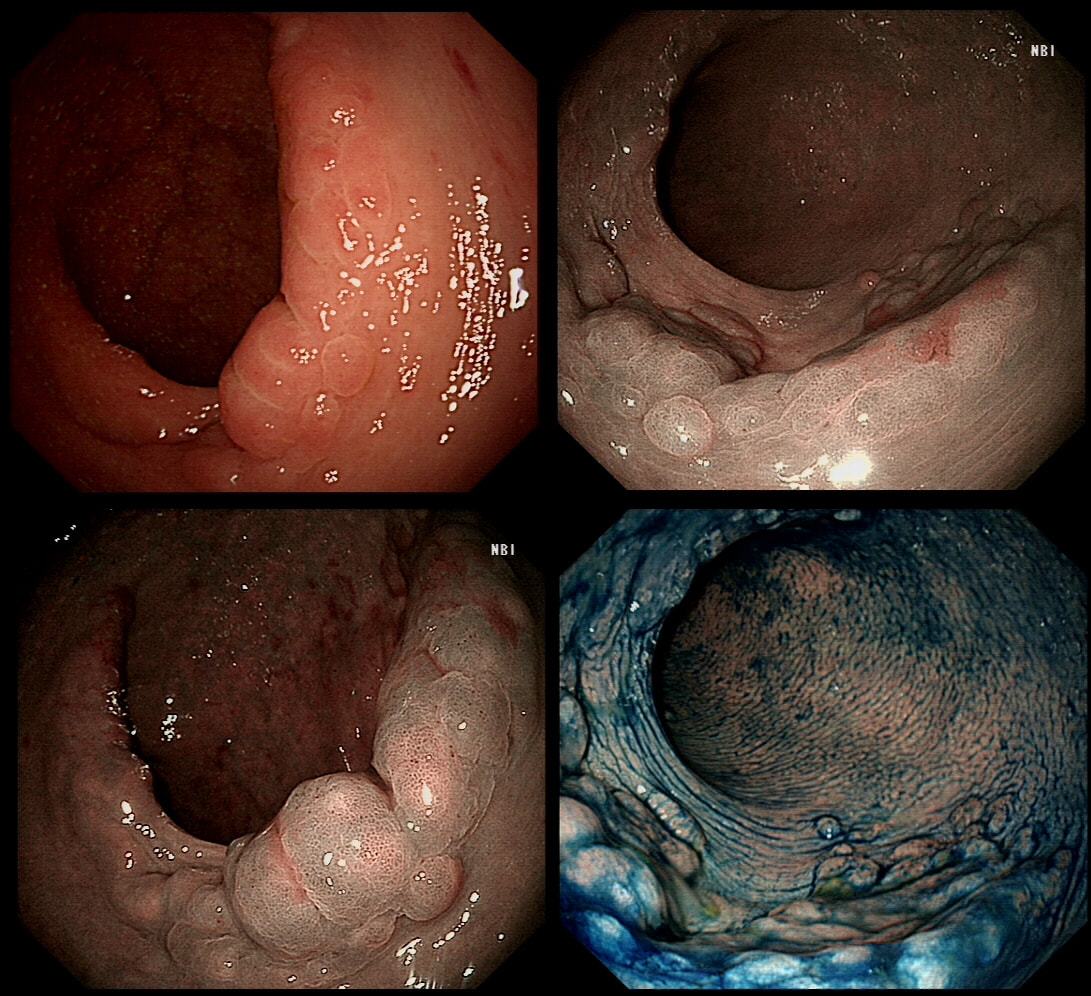
Here are three colonic polyps. One is a TA+LGD, another a TA+HGD and the final is a TVA+HGD
WHICH POLYP IS THE TVA?
a) A
Nope, this one has slit-like crypts typical of a tubular adenoma (TA). In fact, most small polyps are TA's. Presumably there is a progression from TA→TVA→Cancer apart from depressed lesions (IIc lesions) which go directly from being TA's to cancer.
b) B
It's a little difficult to see the crypt pattern without a close-up. However, you don't need to! Look at the growth morphology. This is a LST-NG and they are ALWAYS TA's!
c) C
Yes! Gyrate crypts best seen after indigocarmine dye spray (I think).
explanation
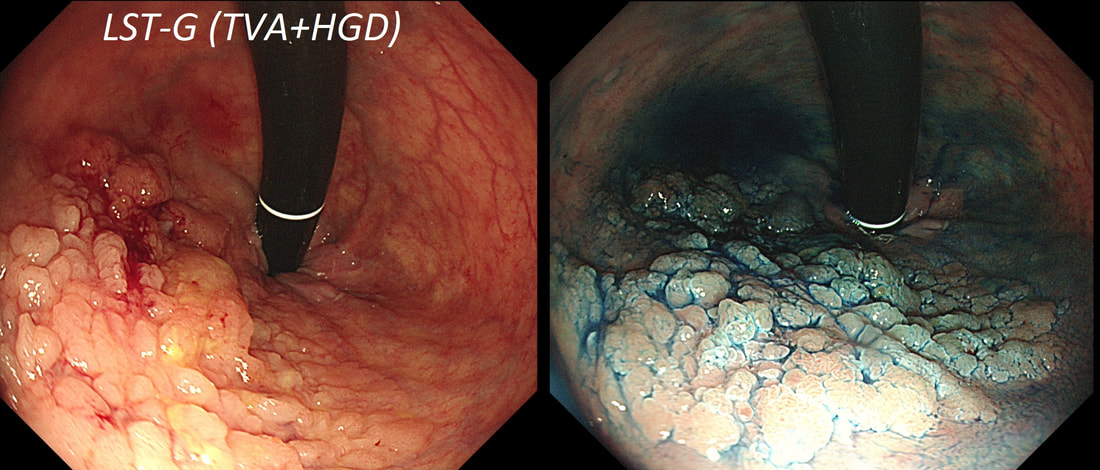
Most 'laterally spreading TVA's are of course LST-G's. However, this doesn't have the usual cobblestoned appearance of a 'laterally spreading tumour of the granular type'. It's rather 'chunky' in fact. That's because its harbouring HGD! A rare beast indeed as almost all laterally spreading TVA's only harbour LGD.
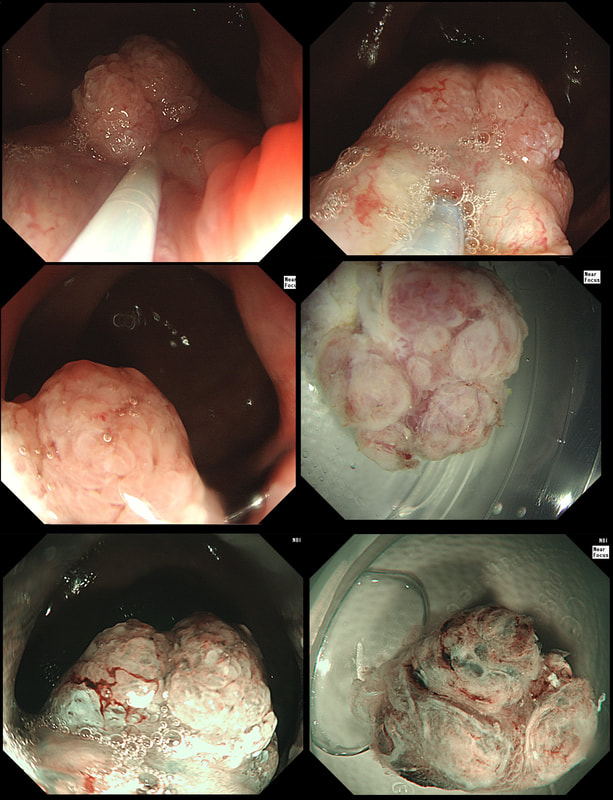
This scary looking polyp was found in the sigmoid in a patient on a polyp surveillance programme. WHAT IS THE LIKELY DIAGNOSIS?
a) Mucosal prolapse
Yes! You can't see any typical gland openings at the tip of this polyp. It's a mucosal prolapse polyp with a serrated lesion at the tip.
b) Adenomatous polyp
It's a battered polyp but without any recognisable crypt openings at the tip and therefore NOT adenomatous.
c) Malignant polyp
Well it looks scary and without an organised crypt pattern. All hallmarks of a malignant polyp. However, this lacks crypts because it's an innocent mucosal prolapse polyp!
Explanation
You've seen a few examples now of the 'mucosal prolapsing conditions' which include solitary rectal ulcer syndrome and inflammatory cloacogenic polyps. First described in 1985 [GIE 1985;31:196–9] they are usually said to be rare. However, experienced endoscopists know that these are common in the sigmoid. I've never seen a mucosal prolapse polyp outside of the sigmoid and would actually not make this diagnosis elsewhere in the colon. Histology is usually said to be the way to "confirm the diagnosis and rule out cancer". However, for me it's an endoscopic diagnosis. But I do understand that these lesions do look alarming. For this reason, I sometimes take it upon myself to resect the tip of the lesion, proving to all those of little faith, that the lesion is absolutely innocent. However, I'm not sure that my surgical colleauges are always reassured by the histology which usually rambles on about 'distorted elongated branched crypts, with fibromuscular obliteration of lamina propria with lots of intramucosal haemorrhages and a splayed and hypertrophied muscularis mucosae". However, the bottom line of that pathology report will read; 'No dysplasia'.
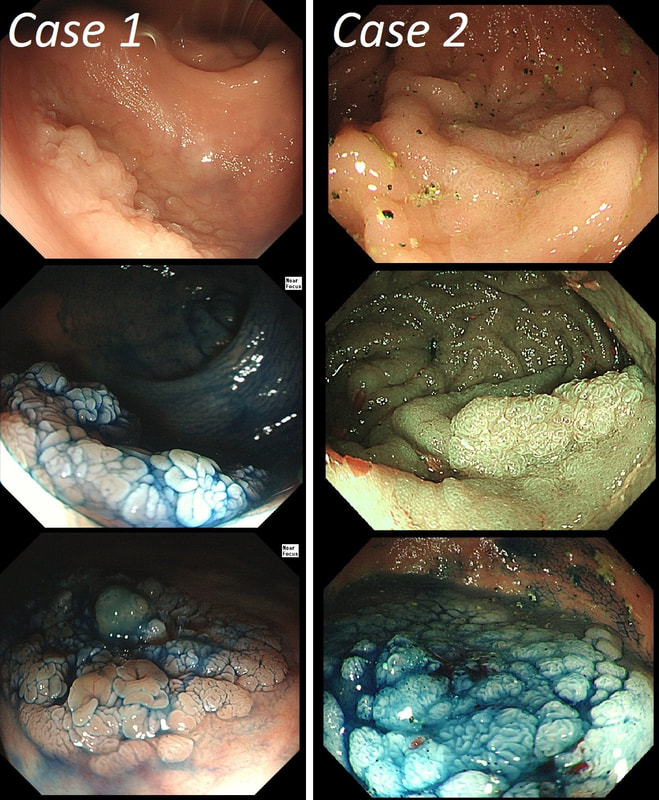
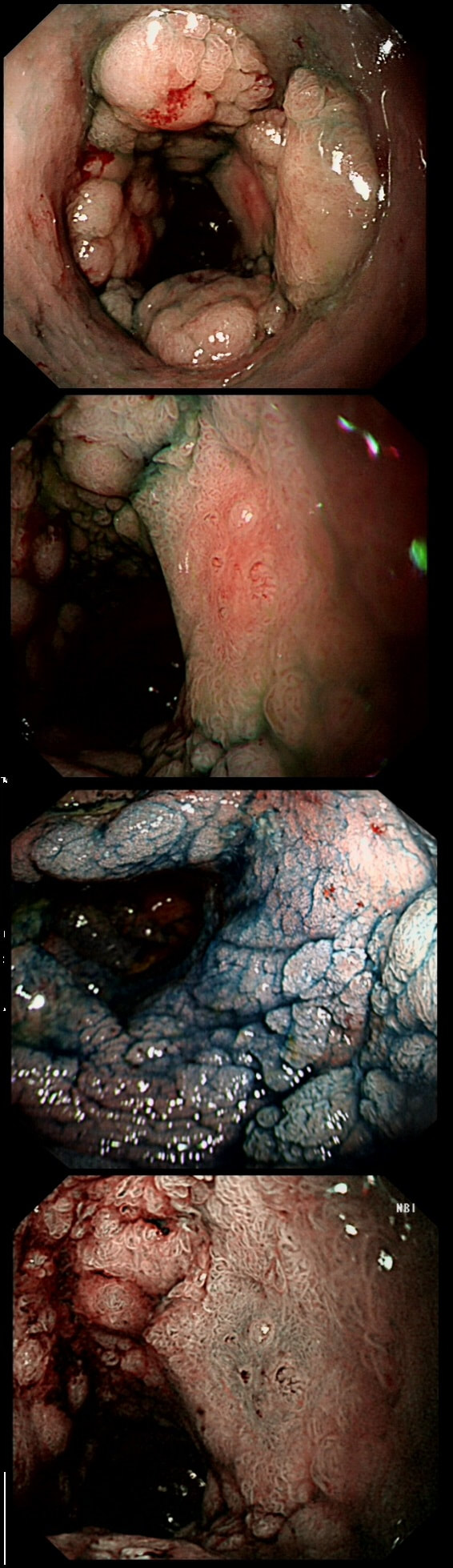
You may be surprised to hear that both these polyps (found in the caecum and rectum respectively) actually have the same histology!
WHAT IS THE LIKELY HISTOLOGY OF THESE TWO LESIONS?
a) SSL's
But it has gyrate crypts and perhaps villous in places??!
b) TSA's
Well done! TSA's are great cameleons !
c) TA's
Slit like or small round crypts? No !
d) TVA's
Reasonable guess as the growth morphology is that of a LST-G which are usually TVA's!
e) VA's
Right-hand image does look a little villous, perhaps
explanation
The surface appears villous in places and gyrate in other areas. The truth is that it is a bit of both - a 'Traditional Serrated Adenoma' (TSA), first described in 1990 by Longacre and Fenoglio-Preiser (Am J Surg Pathol 1990;14:524–37). TSA's are usually less recognisable and usually somewhat 'cerebriform' (like an exaggerated TVA).
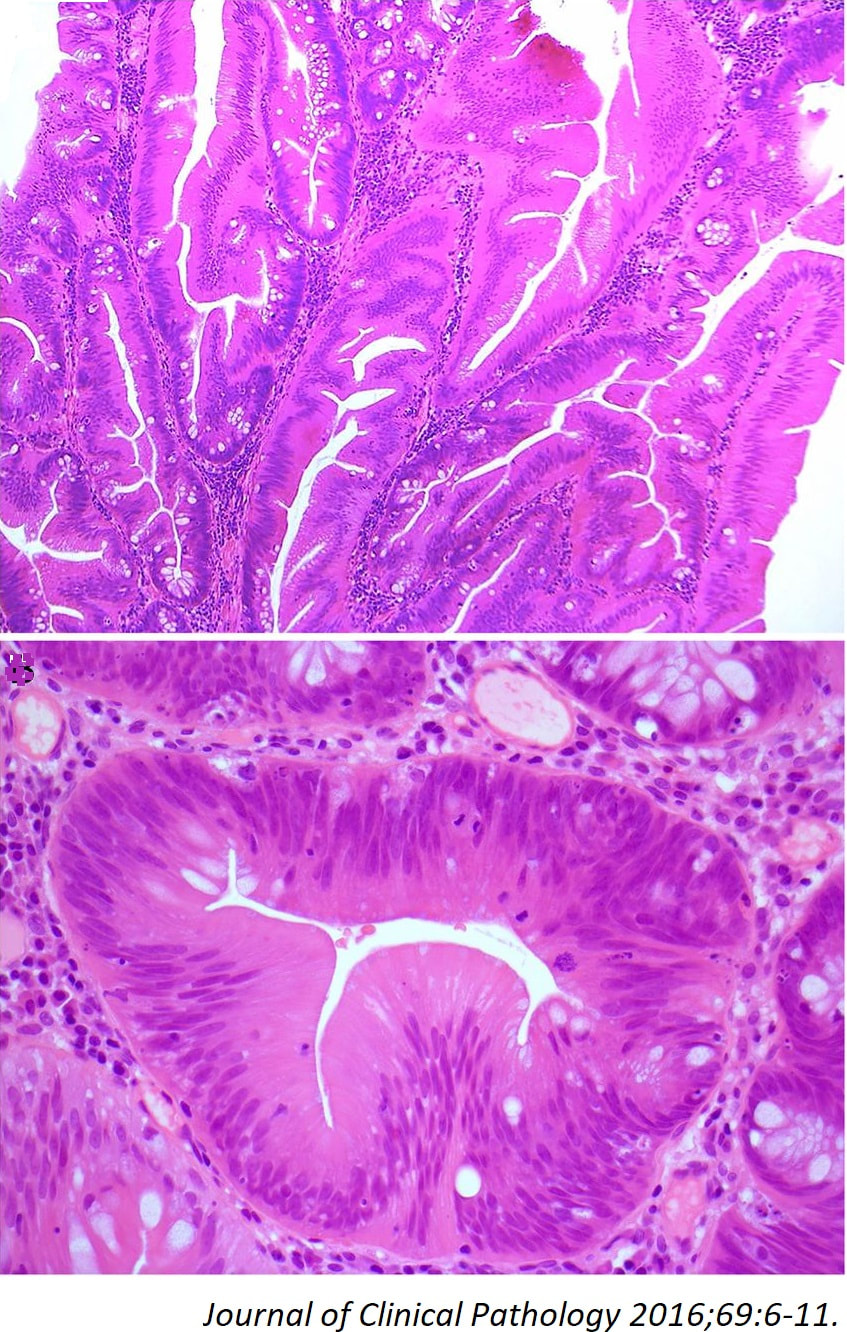
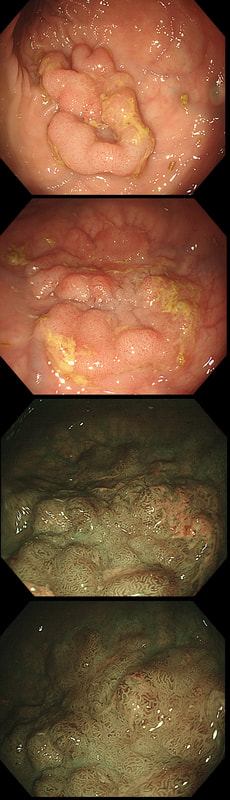
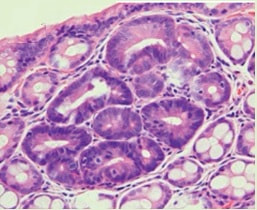
TSAs are the least common of the three serrated colonic polyps ('hyperplastic polyps', 'sessile serrated lesions' and TSA’s) and account for only about 5% of serrated polyps and less than 1-2% of all colonic polyps and are usually found in the distal colon. Pathologists rely on three typical findings when diagnosing TSA’s: deeply eosinophilic cells, flat top luminal serrations and numerous ectopic crypt foci (all demonstrated in the histology slide below from the Journal of Clinical Pathology 2016;69:6-11). Genetically, TSA's are more like adenomatous polyps harbouring low-level microsatellite instability (MSI-L) or microsatellite stable (MSS) serrated colorectal adenocarcinomas. They usually contain KRAS mutations rather than usual BRAF mutations which you find in the 'Serrated Pathway' to CRC associated with a high-level of microsatellite instability (MSI-H). Nevertheless, it’s thought that these precursor lesions give rise to serrated colorectal adenocarcinomas as part of the serrated (accelerated) pathway to cancer. This is an interesting looking colonic polyp WHAT IS THE LIKELY HISTOLOGY
a) mixed serrated / adenomatous polyp
And that is what it looks like!!!
b) traditional serrated adenoma
But there are TWO components to this polyp!
c) serrated polyp undergoing malignant conversion
Well something has happened but 'lesion' has beautiful crypts!
explanation
Actually, the red bit of the polyp was composed of a tubulovillous adenoma and the rest was a serrated polyp! In other words, a mixed serrated/adenomatous polyp. Clearly the famous "three pathways to colorectal cancer": the APC mutation pathway (50%-70%); the mutator “Lynch syndrome” route (3%-5%); and the serrated pathway (30%-35%) can sometime get mixed up !!! This lesion, situated in a very spastic sigmoid, has been referred for resection. WHAT IS THE LIKELY DIAGNOSIS?
a) lesion is not neoplastic
Yes, hard to believe but true!
b) lesion is adenomatous
Lesion is 'battered' but crypts don't look typically adenomatous
c) lesion is malignant
No way, lesion isn't angry red, chunky or devoid of crypts!
explanation
I couldn't stop myself! After yesterday's case of a rectal 'prolapse polyp, part of the 'Mucosal Prolapse Syndrome', I had to show an example of a sigmoid mucosal 'traction polyp' (my nomenclature). The mucosa at the apex of this sigmoid fold is traumatised and inflamed but not actually adenomatous! Histologically these lesions also appear somewhat bashed up. This is where pathologists may see 'pseudo-invasion' which is actually movement of crypts due to trauma and inflammation. The sigmoid colon is the most 'powerful' part of the colon developing the force needed to go to the toilet. Presumably this is the reason that diverticular disease first develop in the sigmoid. The force can also create these pseudo-polyps from patches of inflammation which I presume gets tugged along with each peristaltic wave. The end result is that this is the most difficult part of the colon to make head an tail of polyps. These are common lesions and if you are a 'therapeutic endoscopist', you will be refer these lesions. In these cases, I don't go overboard by placing a snare far down the 'pseudo-stalk'. If you did, you will find that it's taking a long time to cut through all that healthy sigmoid mucosal fold and you run the risk of a perforation (early or late). Instead, I just catch the tip of the fold and ask my assistant to close the snare as quickly as possible. Of course, you don't need to worry about a type of chunky central vessel which you may find in an adenomatous polyp. Analysis of a small piece of mucosal apex confirmed a normal mucosa. Hopefully this was enough for everyone to relax ... This lesion in the low rectum has been referred for endoscopic resection WHAT IS YOUR DIAGNOSIS?
a) Mucosal prolapse syndrome
Well done!
b) Sessile Serrated lesion
Looks just like it but it isn't!
c) Traditional Serrated Adenoma
That's a No!
d) Adenomatous polyp
Patient complained of constipation...
e) Malignant polyp
But it's not 'chunky', red and it has crypts ?!?
explanation
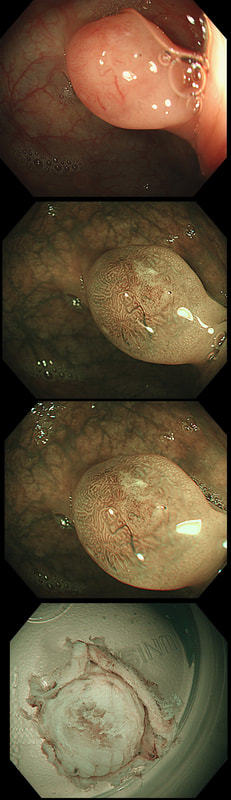
There is no distinct edge to the 'polyp' and the crypts look normal, although a little splayed out and larger than normal, reminiscent of a serrated polyp. In real life, I had the advantage of knowing that the patient had been sent for colonoscopy because of constipation. I low-risk indication of course. There is no distinct edge to the 'polyp' and the crypts look normal, just a little splayed out and larger than normal. This 'polyp' is actually part of the "Mucosal prolapse syndrome", first described in 1983 by Du Boulay et al [J Clin Pathol. 1983;36:1264–8]. Obviously, this syndrome includes solitary rectal ulcers, inflammatory cloacogenic polyps but also rectal 'prolapse polyps' such as this. In addition, the 'Mucosal Prolapse Syndrome' includes those prolapsing mucosal folds in the sigmoid and perhaps most surprisingly - GAVE ! In a case like the above, a couple of superficial biopsies will usually reveal the true nature of the lesion as it's easy for pathologists to spot the fibromuscular hyperplasia with overlying epithelial crypt distortion rather than dysplasia. This is a video clip of a small lesion removed from the sigmoid colon. WHAT IS THE MOST LIKELY HISTOLOGY?
a) TA+LGD
But it's a IIc lesion with IIIs crypts in the centre?!?
b) TA+HGD
Absolutely!
c) Invasive cancer
But there ARE small round crypts in the centre and it DOES lift!!!
explanation
You may call this a "flat elevated lesion with a central depression (IIa+IIc lesion) or simply a depressed lesion (IIc lesion). Frankly it doesn't matter because both a part of the same 'family' of evil little b.....ds. They are always TA's and the small, round crypts (Kudo type IIIs crypt pattern) tells you that the lesion harbours HGD. This is because as dysplasia progresses from low to high grade, crypts get smaller and more withered. Of course they eventually disappear altogether as the lesion develops into a cancer which no longer follows any 'instructions' to form organised crypts. However, the crypt pattern is still discernible in the centre AND the lesion lifts well. Both of these tells you that the lesion is likely to still be benign. Ultimately, the pathologists called it a TA+HGD. However, there was mucinous differentiation in the centre of the lesion. Could these little shits be the early stage of mucinous colonic cancers? Quite likely! Imagine how easily they are missed when hiding behind a fold or below a shallow puddle !!!
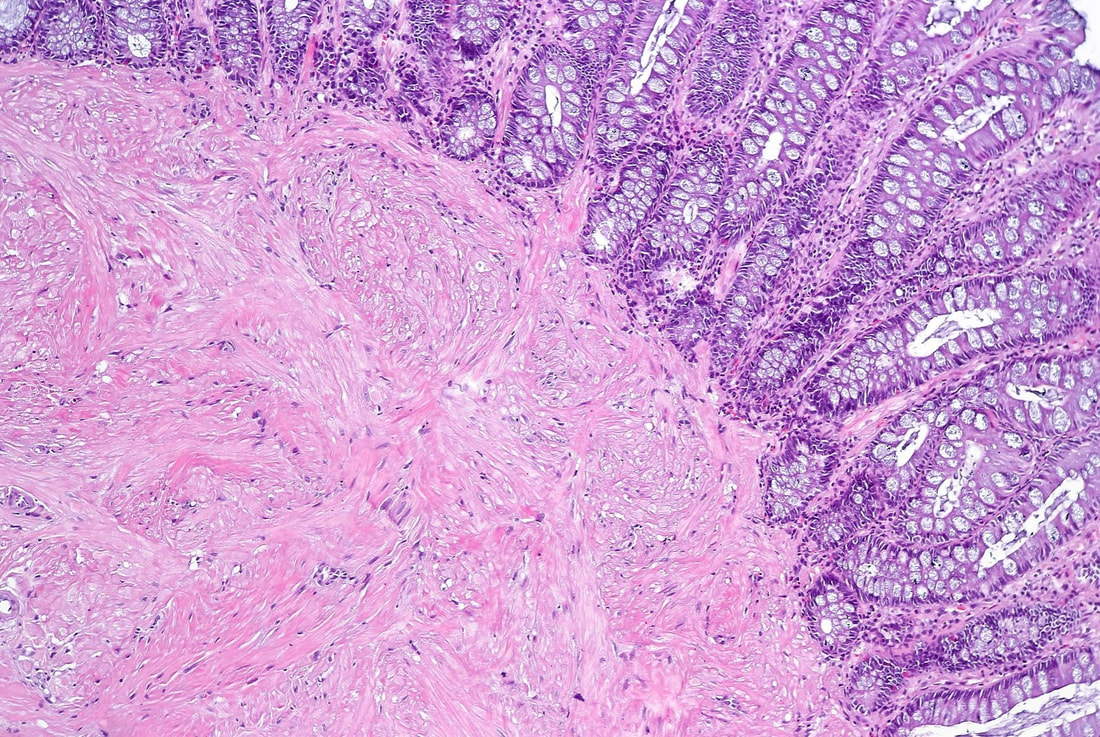
This colonic polyp was removed as a single fragment from a 60 year old lady. You can see the mucosal defect in the last image. The patient asks you what will happen next?
WHAT WILL YOU REPLY?
a) Can't tell, we will have to wait for the histology
You can do better!
b) In all likelihood there will be a site-check in 4 month or so
You are missing something!
d) You will probably need an operation
You've spotted the desmoplastic reaction!
explanation
The polyp looks very suspicious but did seem to lift and I therefore decided to go ahead and removed it using a stiff, large snare. It took a little longer than expected for the snare to cut through. Of course, the mucosal defect should be blue. In this case it's yellow! The polyp was malignant, invading about 1mm into the submucosa and you are looking at the 'desmoplastic' reaction generated by the cancer.
Apart from the sometimes deceptive 'non-lifting sign', there are two further signs that a lesion may be malignant. First, it may look smaller and smaller as you inject below the lesion (see example below). Another sign is that your blue sub-mucosal injection appears to lift the lesion until you retrovert and have a look at the other side. If you then find that it hasn't actually 'crossed the mid-line', there is fibrosis below the lesion preventing the fluid to disperse evenly. I was not entirely surprised to learn that the patient declined surgery. After all, he was 86 years old! He lived another 7 years and never developed any sign of bowel cancer. By the way, there is a theoretical risk of tumour seeding if the lesion is perforated during resection. However, when the perforation is done with a red-hot tool such as a knife or snare, the risk of seeding is surprisingly low. I have perforated a handful of cancers but have never had a case of late disseminated peritoneal disease. My Japanese colleagues (off the record), agree that the risk is there (some have seen it) but is low. If you decide to sample a suspicious looking polyp, you shouldn't use the same forceps to sample another lesion. This is because if cancer cells become lodged in the biopsy forceps, which are then used to sample another location and them become stuck in the biopsy, the histopathologist will diagnose cancer in TWO locations when in fact, there is only a single cancer.
This patient with ulcerative colitis has developed a polyp in the transverse colon. The lesion has now been sent for an endoscopic resection.
WHAT WOULD YOU DO?
a) Abort!
Smart!
b) Attack!
You are creating a problem of your own making!
explanation
Some would say; "if you can remove the lesion in that colitic colon, then 'do it'! The problem is that nothing may appear "irresectable" giving plenty of time, determination and poor judgement.
Many studies looking at outcomes of polypectomy in UC, excluded polyps >1-2cm or flat polyps. Other studies have included polyps arising outside of the colitic field or only have a short follow up period of a few years. Actually, most are coming to believe that when dysplasia develops in the colitic colon, it's not a 'random' case of bad luck. It can be the result of a long process of progressive DNA damage! At some stage we will be able to have a look at the state of the stem cell DNA in patients with conditions such as Barrett's, Colitis and atrophic gastritis. I think that we are in for a surprise ! In addition, did you spot the small focus of invasive cancer in the 2-3 O'clock position? Surprisingly, this was only T1 disease!
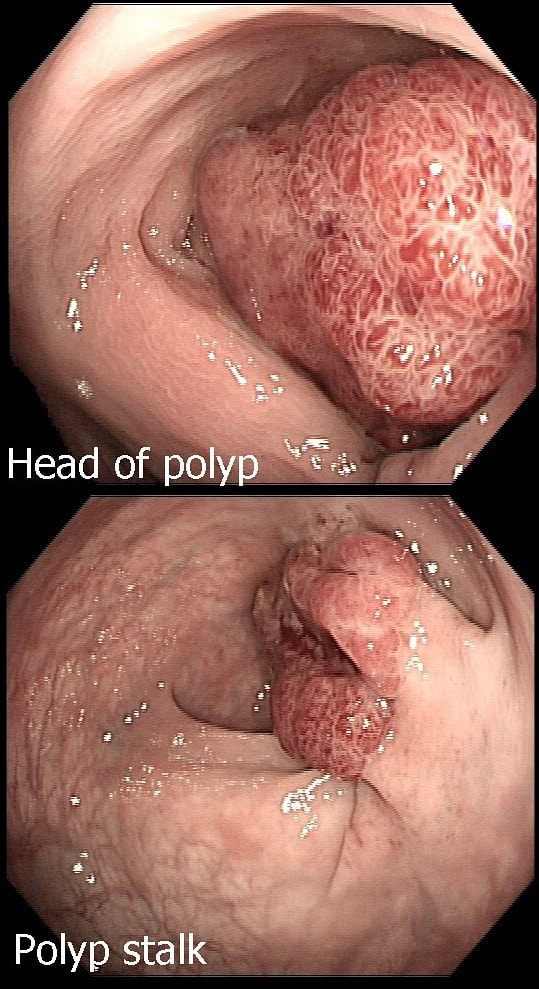
This polyp on a short stalk was removed from the colon
WHAT IS THE LIKELY HISTOLOGY?
a) TA
At the edge, the crypts are clearly sli-like but how about the centre?
b) TVA
Doesn't look like gyrate crypts!
c) VA
Doesn't look like a sea anenome!
d) TSA
TSA's do have crypts but this lesion doesn't in the centre!
e) Early cancer
Was my own firm diagnosis!
explanation
The head of the lesion is clearly of concern. There are 'horns' on it! Of course, the devil has horns but as it was arising from a stumpy stalk. I went ahead and removed it. Somewhat surprising our pathologists called the lesion TA+HGD!
Admittedly, there was disagreement between our histopathologists and 2 out of 5 believed that it was an invasive cancer. Endoscopically, the lesion is clearly malignant and at the very least it's an intramucosal cancer (IMca). Of course, our UK histopathologists are unable to make the diagnosis of Intramucosal cancer in the colorectum because this is not a diagnosis recognised by the 'Vienna classification'. Elsewhere in the GI tract, intramucosal cancer is a diagnosis which our pathologists are 'free' to make. It makes no sense whatsoever to me !
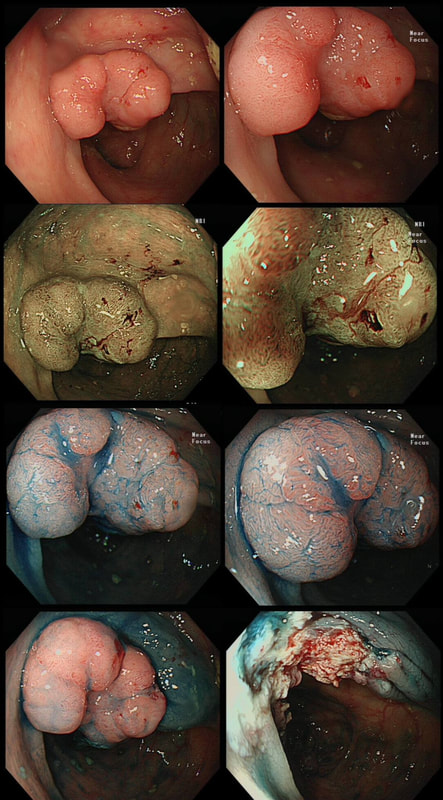
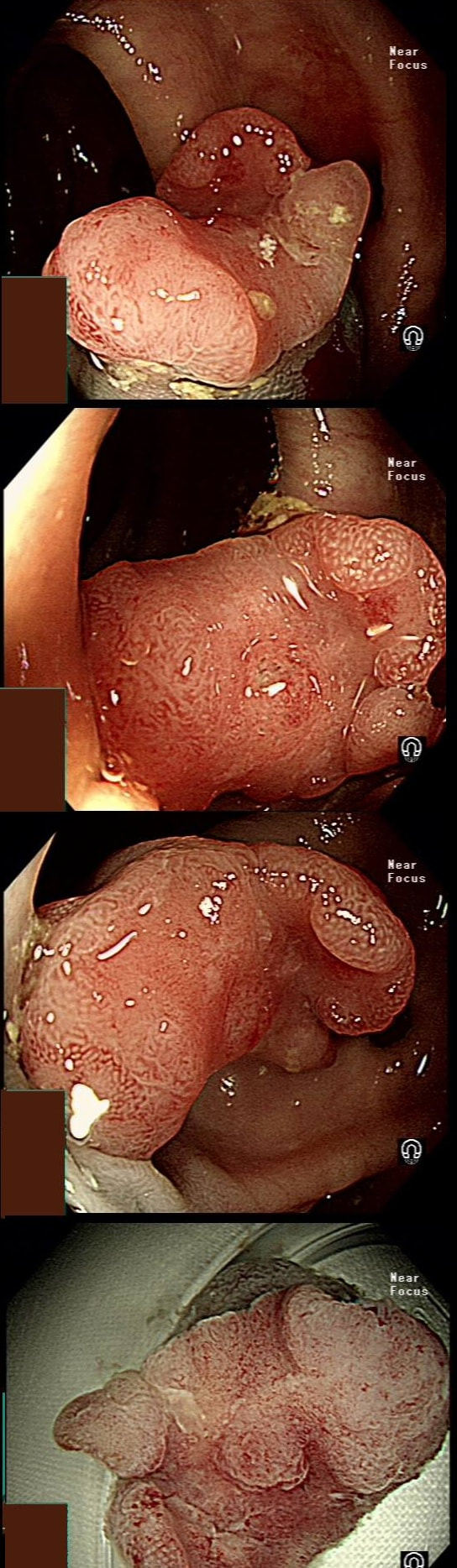
This is an odd looking sigmoid polyp which is seen arising from a broad stalk.
WHAT IS YOUR ENDOSCOPIC DIAGNOSIS?
■ Tubular adenoma (TA)
But the crypts don't look slit-like do they?
■ Tubulovillous adenoma (TVA)
Was my first thought but it doesn't look quite right...
■ Villous adenoma (VA)
If your bx forceps disappears into the polyp it's a VA
■ Traditional Serrated adenoma (TSA)
You'd be excused if you got this wrong - these are rare beasts!
explanation
My initial endoscopic diagnosis was that of a TVA. However, it looked a little "exaggerated". Actually it turned out to be a "traditional serrated adenoma" (TSA).
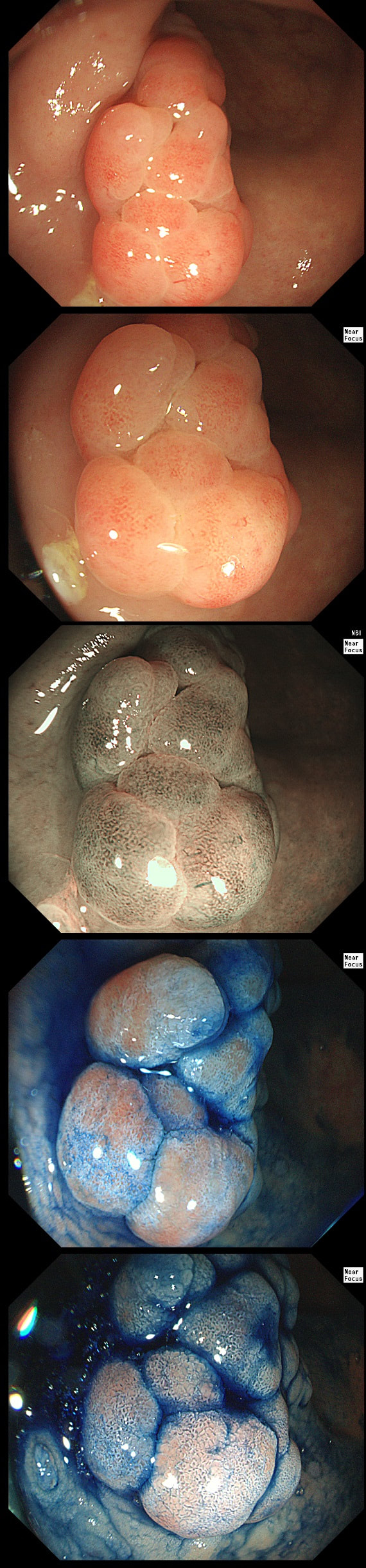
TSA's are rare and mysterious polyps. They only accounting for about 1% of colorectal polyps and are most common in the sigmoid and rectum. Endoscopically they can appear as “exaggerated” tubulo-villous adenomas or as villous adenomas. In the image below, I've put four examples of TSA's together to illustrate how differently these can appear ! It has been proposed that because one-third of "serrated cancers" are found in the distal colon and perhaps these cancers arise from TSA’s! However, genetically, TSA's are more like adenomatous polyps harbouring low-level microsatellite instability (MSI-L). They usually contain KRAS mutations rather than the BRAF mutations which you find in the 'Sessile Serrated Lesions'. For this reason, it's perhaps unlikely that they contribute to the 'Serrated Pathway' to cancer. By the way, a recent paper has highlighted that patients with TSA's are also at greater risk of 'high risk polyps', elsewhere in the colon.
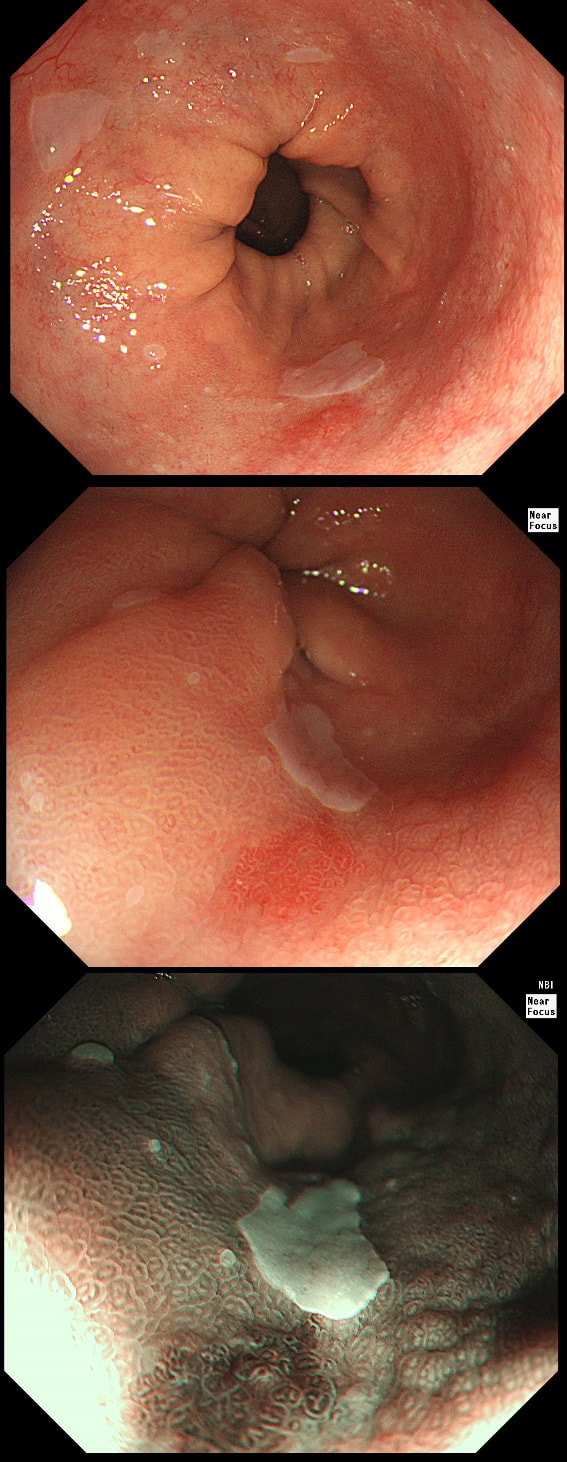
During a Barrett's surveillance OGD, I notice this small red spot at 6 O'clock.
WHAT IS THE LIKELY HISTOLOGY?
■ Reflux ulcer
I don't think that you get this within Barrett's without dysplasia!
■ LGD
Clear crypt pattern, not that different from surrounding mucosa? Good guess!
■ HGD
Quite possible and would have been my guess IF there was some nodularity associated
■ Early cancer
Impossible as lesion has an organised crypt pattern!
explanation
Previous surveillance samples, 3 years earlier had not revealed any dysplasia. However, that red spot shouldn't be there! A reflux ulcer is unlikely as the Barrett's is there because it protects the food pipe from acid. Of course it does this by developing crypts with Goblet cells which secretes protective mucus (of course the pathologists call this process 'metaplasia'). Early texts usually stated that ulceration is common within Barrett's. In my experience, ulceration within Barrett's only happens when dysplasia develops. With the arrival of dysplasia, you begin to see a disappearance of the Goblet cells and of course when the Goblet cells start to dwindle, so does the protective mucus.
In the bottom zoom image you can see that there is a distinct surface crypt pattern at the site of the red spot. Therefore the cells can't be too disorganised, making cancer unlikely. Actually this turned out to be a spot of LGD. I would have guessed HGD/IMca (pathologists struggle to tell the difference) if there was a visible nodule at the site of the red spot. Isn't it interesting that even at the very earliest stage of dysplasia, the unstable cells signal the need for more oxygen to nearby vessels?
This lesion was found in the ascending colon.
HOW WOULD YOU DESCRIBE IT?
■ Flat elevated lesion (IIa)
Only lesions 10mm or smaller are referred to as 'IIa lesions'
■ Flat elevated lesion with a central depression (IIa+IIc)
But that depression dissappear when lumen is inflated!
■ Laterally spreading tumour of the non-granular type (LST-NG)
But the surface is 'cobbled' not smooth!
■ Laterally spreading tumour with a central depression (LST-D)
LST-D's are always TA's (and therefore have a smooth surface)
explanation
Lets just state something from the start! A flat elevated polyp is of course a IIa type of lesion BUT when larger than 10mm in diameter is no longer referred to as a IIa lesion. Now, it's a 'Laterally Spreading Tumour'!
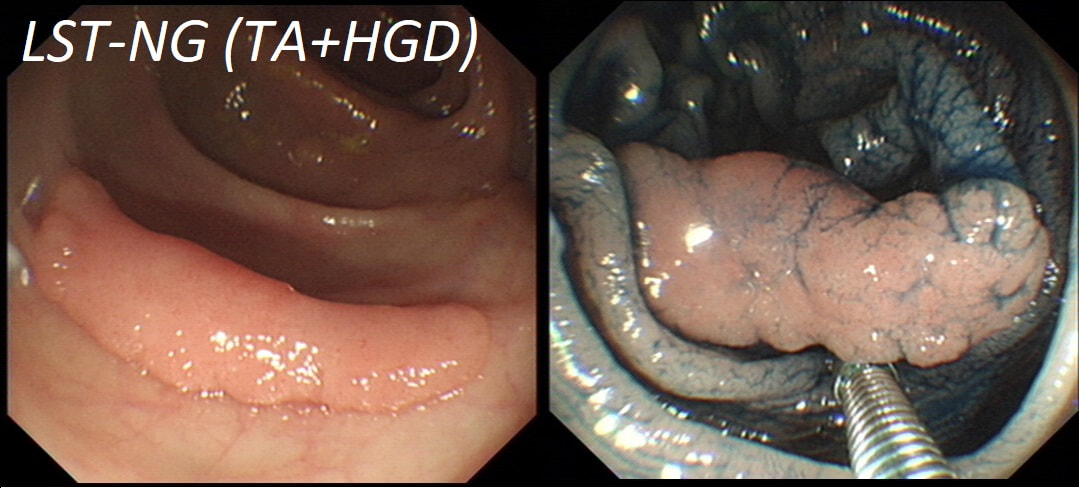
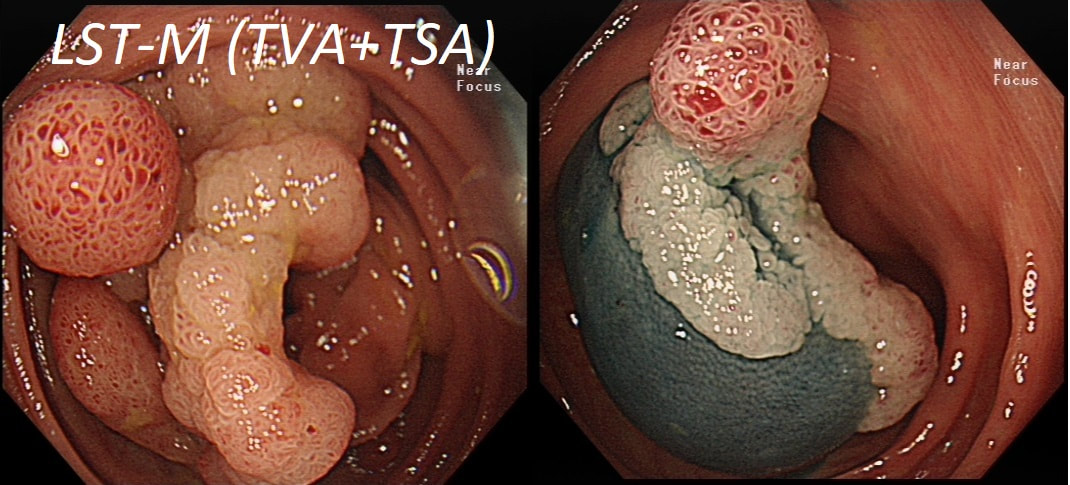
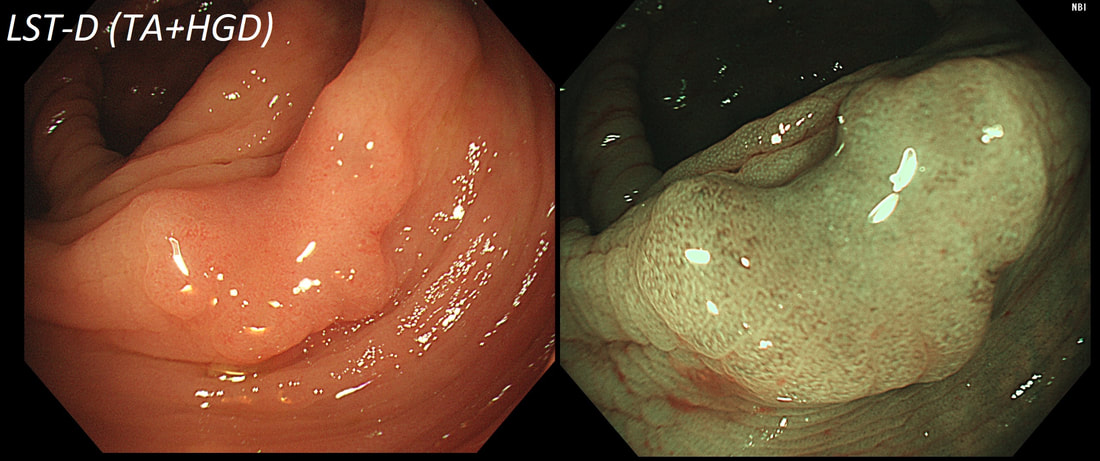
There are 4 types as follows:
In this case the crypts are gyrate, (type IV crypts), making it a TVA. Laterally Spreading Tumours with a gyrate crypt pattern are all TVA's and almost never harbouring more than LGD. OK but it looks depressed in the centre doesn't it? No, it isn't depressed in the centre! It's simply they way the large lesion has folded itself. This is something of a 'trick question' as I deliberately didn't inflate the colonic lumen fully in the first image. When the lumen is inflated, you can see the true shape of the lesion; a LST-G ! A meta-analysis in 2013 by Voorham et al. [Voorham QJM. AM J Gastro 2013;108(7):1042-] concluded that pedunculated polyps were more likely to harbour KRAS mutation and APC mutations than flat lesions, and that flat lesions were more likely to harbour BRAF mutations. Depressed lesions and LST-NG’s were particularly unlikely to contain KRAS. Below are some examples of LST's
This lesion was found in the rectum of a patient undergoing colonoscopy because of constipation
WHAT IS THE LIKELY DIAGNOSIS?
■ Polyp from mucosal prolapse
Clever!
■ Serrated polyp
Crypts look a little serrated but the rest doesn't
■ Adenomatous polyp
Polyp doesn't look 'neoplastic'
■ Malignant polyp
Now, this doesn't look malignant!
explanation
Well perhaps the crypts look a little like serrated crypt openings but somehow the rest of the polyp doesn't look like a typical serrated lesion. Where is the covering mucus?! Furthermore, the polyp definitely doesn't look adenomatous or malignant!
Actually this has arisen as part of the 'Mucosal prolapse syndrome' which is the umbrella term for entities such as; solitary rectal ulcer syndrome and inflammatory cloacogenic polyps. Patients are often constipated or have difficulty with defaecation with straining on the toilet or undergo the sigmoidoscopy because of tenesmus, altered bowel habits or incontinence. Surprisingly, some patients don't have any straining-related complaints! Most pathologists would recognise the typical mild fibrosis, thickening of the muscularis mucosae and crypt irregularity (dilated, diamond shape crypts). The surface epithelium show regenerative changes. These polyps are found in a rectal pouch WHAT IS THE UNDERLYING DIAGNOSIS?
■ Serrated polyposis syndrome
Polyps is SPS are flat (usually)
■ FAP
Looks like a classic example of rectal surveillance in FAP but polyps do look odd don't they?
■ Juvenile polyposis
Yes! Pt had already developed a CRC and after a subtotal colectomy offered rectal surveillance
explanation
These polyps have a funny crypt pattern, but THERE IS A PATTERN! Accordingly, they are likely to be 'hamartomatous' (a non-sensical histological term which basically means overgrowth of normal tissue). Of course, this patient has Juvenile Polyposis syndrome. Although the polyps are 'hamartomatous', as in Peutz-Jeghers syndrome, there is a stark difference. The polyps in JP often turn dysplastic and are presumably the origin of this patients adenocarcinomas. In contrast, the polyps in PJS are extremely rarely reported to harvest dysplasia (I've never seen a dysplastic polyp in PJS). By the way, the WHO criteria for diagnosis of juvenile polyposis syndrome are one of either:
This polyp was found in the transverse colon of a 60 year old man. WHAT IS THE LIKELY HISTOLOGY OF THE LESION?
■ Serrated polyp
Crypt openings look a little serrated but nothing else does!
■ Adenomatous polyp
This is not an adenomatous crypt pattern!
■ Hamartomatous polyp
When in doubt, it's hamartomatous !
■ Malignant polyp
The crypt pattern is too organised and regular for cancer!
explanation
Sadly, an intimate knowledge of the Kudo crypt patterns doesn't help you here! Adenomatous polyps should be covered with one of the following;
In this case, the closest match is of a serrated polyp, which have wide open crypts with a somewhat jagged outline. However, apart from the crypt openings, there is nothing on this which looks like a serrated polyp! If you can't see a crypt pattern, the lesion is likely to be malignant. Conversely, if the lesion does have crypts but still doesn't look familiar, its either a hamartomatous polyp or, perhaps less likely, a 'Traditional Serrated Adenoma'. This lesion turned out to be a hamartomatous polyp. Why a 60 year old man would grow a hamartomatous colonic polyp remains a mystery ! This is beautiful polyp, perched on a fold, was found at the junction between the caecum and the ascending colon. The video gives you a better idea of the size and extent of the lesion. WHERE WOULD YOU PLACE THE NEEDLE FOR THE BEST 'LIFT' ?
■ On the side facing you
A convenient choice but lift will not be optimal
■ Into the apex of the polyp
That would be my choice!
■ Just behind the lesion
Better than into the fold facing you
explanation
I have no qualms about injecting straight into the middle of lesion provided that I'm sure that it's benign. The lovely gyrate pattern of this polyp tells you that it's a TVA, likely to harbour no more than LGD. Injecting into the fold facing you is likely not to raise the 'blind side' of the polyp which extends down the back of the fold and onto the 'flat' beyond. Conversely, injecting into the back of the lesion would lift the back end but probably not the front. My choice was to inject into the apex (see video below) which resulted in a lovely lift. But, why not inject in two places? Because you should try to avoid injecting into more than one place (if it can be avoided). If you have made more than one hole in the epithelium you may will find that your injection leaks out through the previous hole and that the elevation is less effective. Of course when removing large, flat lesions multiple injection sites can't be avoide, unless you are removing the lesion by ESD of course.
This small polyp was removed from the transverse colon. H&E histology is attached
WHAT IS THE DIAGNOSIS?
■ Serrated polyp
Crypt pattern looks more adenomatous!
■ Adenomatous polyp
Slit-like crypts but that thick capillary doesn't fit!
■ Leiomyoma
You're good at histology!
■ Neuroendocrine tumour
That was my guess too but I was wrong!
■ Small CRC
This lesion is submucosal!
explanation
I was quite sure that this was a small NET. It's that vessel crawling up it's side which convinced me. The reason that I enclose the histology is because it's not a neuroendocrine tumour! Histology shows interlacing bundles of spindle-shaped, smooth muscle cells. with bland-looking nuclei probably arising from the muscularis mucosa just below the epithelium.
It's a leiomyoma, which are very uncommon in the colon but of course very common in the oesophagus. Endoscopically, it's usually not difficult to tell the two apart. NET's often have a dip in the centre and/or sizeable vessels crawling up their sides. Colonic leiomyomas are usually covered with entirely normal looking epithelium. With immunohistochemistry it's easy to tell the two apart. A GIST (thought to arise from the 'interstitial cells of Cajar') usually stain with c-kit (CD117) while a leiomyoma is (thought to originate from smooth muscle cells) stain positive for smooth muscle actin or desmin, but not for c-kit. Of course leiomyomas are subepithelial lesions, covered with a normal mucosa. The differential diagnosis of submucosal swellings in the colon include:
This sessile polyp was found in the caecum, close to the appendiceal orrifice WHAT IS YOUR DIAGNOSIS?
■ Mixed adenomatous & serrated polyp
I don't see a mixture of crypt patterns!
■ SSL
Absolutely!
■ TSA
No, TSA's look like a cross between TVA & VA!
■ Adenomatous polyp
Perhaps the crypts look a little slit-like but it's covered with mucus...
explanation
Well, perhaps the crypt openings look a little slit-like but the lesion is partially covered with that mucus typical of an SSL. By the way, couldn't it be a hyperplastic polyp? I think that pathologists have given up even attempting to tell the two apart! My own rule of thumb is that anything which is ≥10mm, I call an SSL and remove. By the way, SSL's usually lift very well. In this particular case, the lift is a little sub-standard for an SSL, presumably because it's situated very close to the appendix orifice which anchors it down. A word of caution! I've had several 'post polypectomy syndrome' cases after removing large SSL's. In these cases, the lift was excellent and I asked for the LARGE snare to remove the lesion en-bloc. However, in both cases, I found that the snare was taking quite a long time to cut through. Perhaps because of that fatty reaction in the stroma below which Neil Shepherd talked about in the Podcast. To avoid any risk of the 'post polypectomy syndrome', you should ask your assistant to close the snare as quickly and hard as possible. Don't worry about bleeding. These lesions are never supplied by any significant vessels. Oh yes! And place clips !!! This 80 yr old man was first referred for a CT to investigate his PR bleeding and mucus. The CT reported that the rectum appeared thickened and a Flex Sig is organised WHAT IS YOUR DIAGNOSIS?
■ Active proctitis with inflammatory polyp
That was my diagnosis and I was half correct!
■ Active proctitis with adenomatous polyp
Very good call!!! Better than mine if fact!
■ Circumferential polyp with dominant nodule
Apologies for the indigo carmine dye which has confused you!
■ Circumferential polyp with malignant change
The blue dye is confusing and the proctitis does look weird in places
explanation
Yes, it does seem odd to refer for a CT rather than a Flex Sig? Perhaps this was because the patient was 80 yrs old or because of our endoscopy waiting list. Anyway, I was half correct. I diagnosed an active colitis and histology confirmed a: transmucosal inflammation with crypt abscess and focal epithelial loss (tiny, tiny ulcers). We all know that active colitis is characterised by an dull erythema, bleeding and erosions/ulcers. The best endoscopic scoring system is currently the Ulcerative Colitis Endoscopic Index of Severity (UCEIS) which scores these three parameters on a scale of 0 to 3. There are other scoring systems of course such as the Mayo score, the Baron score, The Rachmilewitz Endoscopic Index, The Sutherland Index, The Matts Score, The Blackstone Index and the most recent addission, The Ulcerative Colitis Colonoscopic Index of Severity! Of course this tells you than none of them is the last word on the topic. For a good review of all endoscopic issues relating to colitis, please check this article out; Annese V. European evidence based consensus for endoscopy in inflammatory bowel disease, J Crohn's Colitis 2013;7(12);982-1018 Anyway, I find it odd that other common features of an active colitis are not included in any of these scoring systems. This includes mucosal oedema, a villous epithelial surface (as seen in this video) and a mucopurulent discharge (which I regard as feature of a severe attack of colitis). These three features are prominent in this video and if not recognised as features of an active colitis, can be mistaken for a neoplastic process. I thought that the nodule would be inflammatory but it turned out to be a 'TSA' (traditional serrated adenoma), the most rare and elusive of all colorectal polyps. Well I blame the inflammation and that thin mucopurulent layer obscuring the crypt pattern. This polyp just distally to the caecum has been referred after an initial set of samples have indicated that it's a benign TVA harbouring no more than LGD. An unfortunate, fixed loop at the hepatic flexure means that I can't retrovert behind this polyp. In fact, it's difficult to get much closer to the lesion than this. WHAT STATEMENT DO YOU AGREE WITH?
■ The resection may be difficult but it can be done
I agree with that!
■ The location of the lesion and difficult colonic configuration probably makes a resection impossible
Of course the patient may have to travel to a nearby centre with the neccessary expertise!
■ All this is irrelevant as the polyp looks suspicious and would be best removed surgically
I disagree, it does look benign! But will of course have a 15% risk of harbouring cancer...
explanation
Actually, the lesion does look very benign. It wobbles about freely perched on top of the first fold distally to the caecum. Furthermore, it has a reassuring gyrate crypt pattern of a TVA. Naturally, any polyp of this size will have a 15% risk of actually containing an unexpected focus of cancer within. Because our therapeutic waiting list is in mess in the aftermath of Covid, I do encourage everyone to sample the sessile lesion even though you are planning to refer it for resection. Such sampling on the surface of large sessile lesions doesn't induce any fibrosis and provides some (limited of course) reassurance that the lesion isn't malignant. In this case, the position is very challenging but you can get close enough to stand a good chance of removing the lesion. That's actually what I did and I've just uploaded a video of the resection together with my face-to-camera annotation to highlight the main points. Ultimately, histology confirmed a TVA harbouring no more than LGD This subcentimetere polyp was discovered in the rectum of a 60 year old woman undergoing a bowel cancer screening colonoscopy following a positive FIT. Considering the endoscopic appearance and the degree of lifting, WHAT WOULD YOU DO?
■ This is a benign adenoma but it's too close to a large vein for removal
Don't worry about the vein!
■ This is a benign adenoma which I would remove
Doesn't look benign!!
■ This is a likely early cancer which I would remove
Yes it looks evil but should you attack?
■ This is an invasive cancer which I would only sample
Can't argue with that!
explanation
That vein looks intimidating but don't be intimidated! It's not a 'show stopper'. it will move out of the way. However, that polyp does look nasty as there is no definite crypt pattern in the centre, just a swirly surface. However, the lifting is good, suggestive that we stand a good chance of clearing the lesion endoscopically ...
And at this point you can either go ahead and remove the likely early cancer. The advantage is that you get the correct diagnosis whilst biopsies are often inconclusive. The reason why surface biopsies are often unable to confirm invasive cancer is because your pathologists rely on seeing invasion of malignant tissue below the muscular mucosa. The underlying muscularis mucosa is usually NOT included in surface biopsies. However, this is a good example of the potential disadvantages of going ahead to resect the lesion. As you'll see in the video below, I removed it. Histology reported that the lesion was indeed an early rectal carcinoma, 6mm in diameter and with clear margins. However, the deep margin was 'only' clear by 200 microns which of course our pathologists would call a 'positive deep margin'. A 1mm (that is 1000 micron) is the 'accepted' definition of a 'clear surgical margin'. Secondly, there was of course a small focus of LVI. There is usually something to keep you awake at night after removing an early cancer. The disadvantage is obviously that you don't know what to tell the patient!!! - Is my cancer gone or not? - I'm sorry but I'm not sure! - Couldn't you keep a close eye on me with regular colonoscopies and CT's? Well, we could but there is absolutely no evidence that this will save your life IF you still have cancer cells inside of you... Fortunately, there is a study under way in the Netherlands to answer this very question! At the moment, we know that LVI is 'bad' and that the normal next step would be to offer the patient surgery.
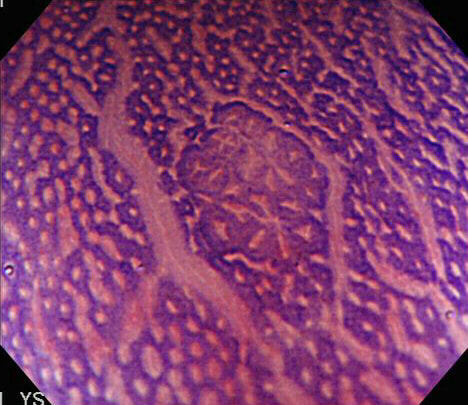
With a magnifying colonoscope and dye, another world is revealed.
WHAT IS SHOWN IN THIS PHOTOGRAPH?
■ Crypt distortion
No, these crypts are MUTATED!
■ Abberant crypt focus
You know your stuff!
■ Small flat adenoma
Histologically, you could be correct!
EXPLANATION
The link between ACF and proximal adenomas, I see as evidence of a colonic “field effect” in which exposure to carcinogenic 'drivers' (dietary, lifestyle, disease and genetic) all work together to prime the colon for the development of cancer. It's for this reason, that endoscopically resecting DALM's (I arbitrarily define DALM's as flat polyps within the colitic field of a patient with >6 yrs of colitis), then telling patients that they now have nothing to worry about, is naïve ...
I found this in the low rectum of an elderly lady undergoing a flexible sigmoidoscopy because of rectal bleeding. A quick check with the patient confirmed my suspicions.
WHAT IS THE DIAGNOSIS?
■ Rectal prolapse
The patient would know about that!
■ Solitary rectal ulcer syndrome
A good guess and patient would know about constipation and straining but actually not the correct answer
■ Likely serrated polyp syndrome
Hmm, what question would you ask patient? FH of CRC perhaps?
■ Likely FAP
Findings of a rectal pouch in FAP? No, that's not it!
explanation
On first glance this looks like a circumferential serrated polyp in the rectum. Of course, this would be somewhat odd and furthermore, unlikely that the patient would know anything about this.
Actually, on direct questioning she did know about her prolapse! Another common endoscopic finding is an erythematous and oedematous patch, close to the anal margin.
A very odd looking polyp at the anal verge. I removed it but still couldn't really make out what it was!?
WHAT IS YOUR BEST GUESS AT THE MOST LIKELY DIAGNOSIS?
■ Inflammatory polyp
Good guess as there is no crypt pattern!
■ Adenomatous polyp
It has the colour of an adenomatous polyp but no crypt pattern...
■ Small adenocarcinoma
Certainly ugly enough but small carcinomas are usually more button-shaped
■ Small SCC
Arising from close to the anal margin but nearby squamous mucosa is unremarkable...
explanation
This is rare stuff! I was clueless although reassured that the thing was soft and didn't feel malignant when squeezed between my fingers.
It proved to be a (rare) 'cloacogenic polyp'! You'd be excused if you have never heard of this (I hadn't). Apparently these were first described as recently as 1981! The same year that I was born ☺! Essentially they are inflammatory polyps arising from the transitional zone of the anorectal junction. They can be much larger than this and even be multiple and can occur at any age. Patients are usually asymptomatic but could (naturally) present with some blood on the toilet paper (this patients complaint). Histology is similar to that of solitary rectal ulcers with inflammation and fibromuscular hypertrophy. For this reason it is perhaps not surprising that they are linked with constipation and straining. Rare associations include small nearby cancers and human papillomavirus infection. |
Categories
All
|