|
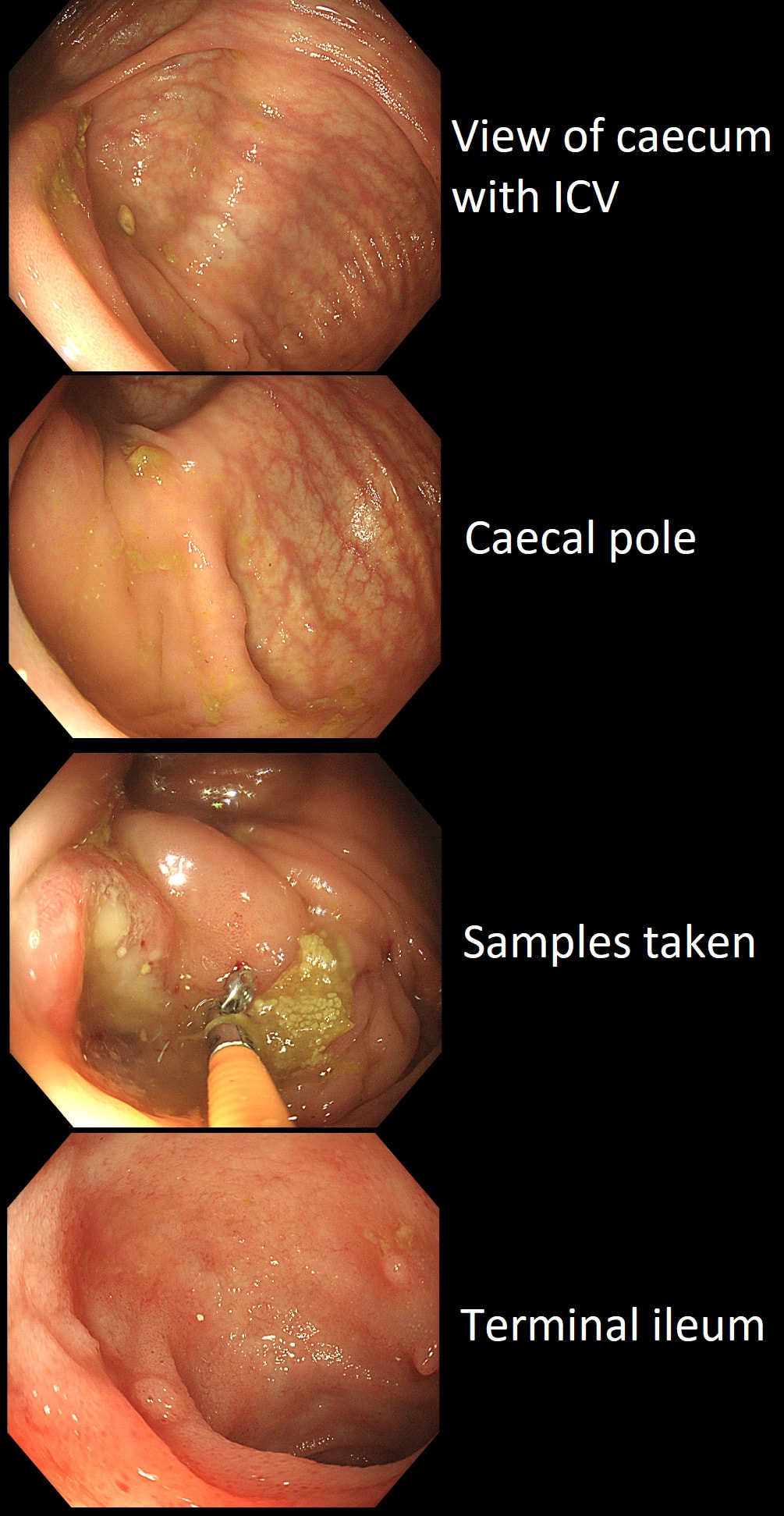
This patient underwent a CT angiogram for chest pain. The angiogram was unremarkable but the radiologist mentions an 'irregularity at the caecal pole with hyperenhancement and mild adjacent fat stranding' and recommend colonoscopy. After the examination, your patient asks you if you have found anything?
WHAT WILL YOU TELL YOUR PATIENT
a) Reassure the patient
That would be unwise!
b) Tell him to wait for the histology
Strictly speaking 'not wrong' but it's not the best option to choose
c) Tell him that we will organise an MRI scan next
MRI's are requested for rectal cancers
d) Tell him that we will organise an EUS next
Life is too short for EUS!
e) Tell him that we will organise a 'staging' chest/abdomen/pelvic CT
This is precisely what you should do!
explanation
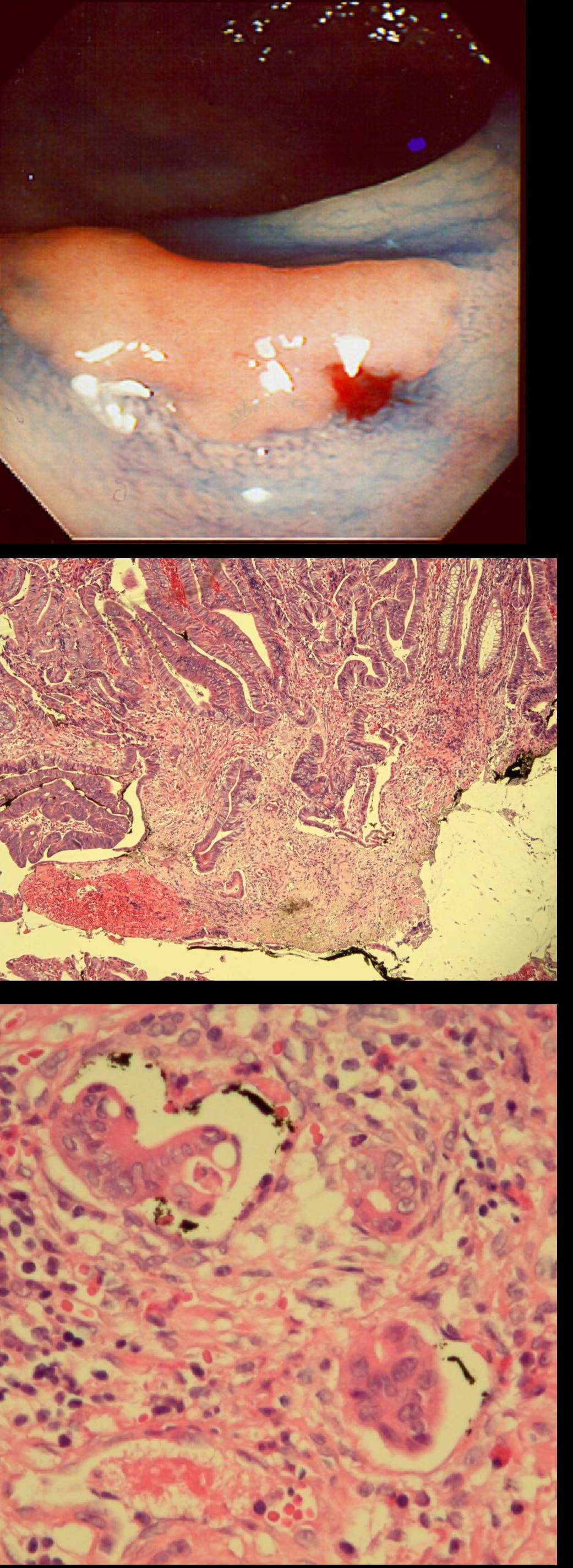
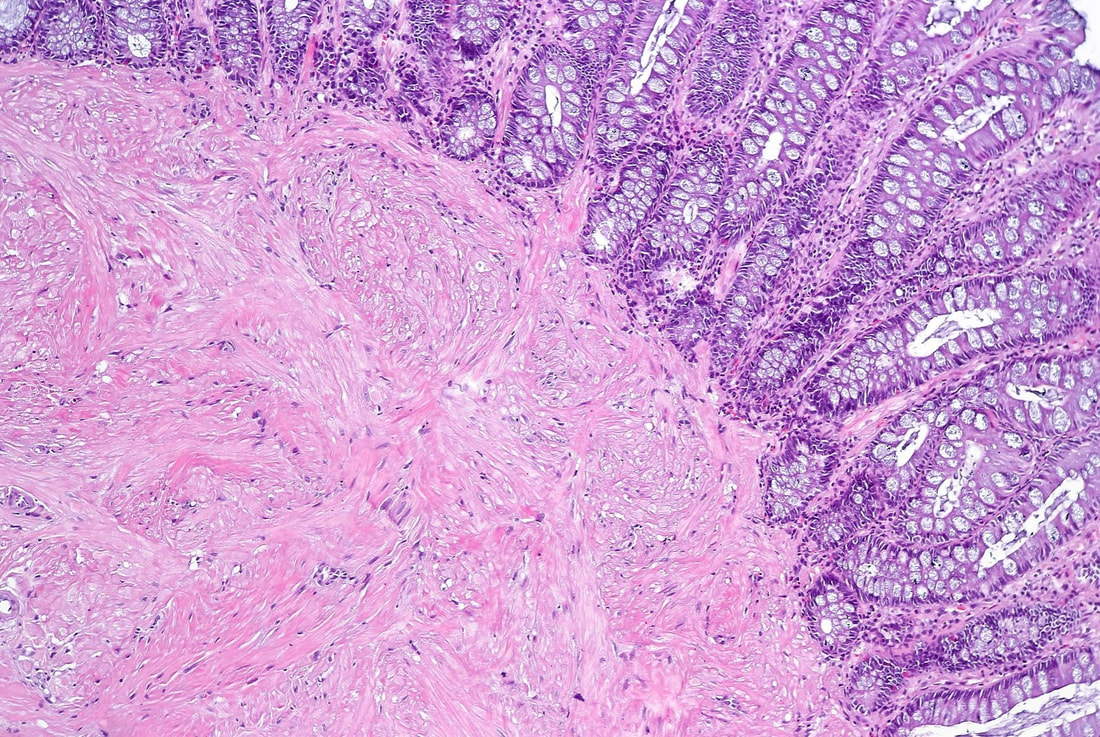
Did you notice the lesion next to the biopsy forceps? At initial glance into the caecum, there is nothing to see. However, this lesion was about 20mm in diameter and with that rolled edge. It's clearly malignant and you need to request staging CT's. Histology did confirm a mucinous adenocarcinoma and CT sized it at 4.7cm. Far larger than initially thought! As it was involving the serosal surface, it was staged as T4, N1 (due to several small nearby nodes).
Mucinous colonic cancers are unusual, accounting for about a little more than 10% of CRC's. They are usually situated in the proximal colon. This is not the only reason why they are easy to miss. At the early stages they have an infiltrative, ulcerative growth pattern which easily hides behind bubbles or a pool. These are small but evil little things which are easy to miss and grow fast. By the way, 'signet ring adenocarcinoma' (where the mucus is INSIDE the cell rather than OUTSIDE of the cells) is another sub-type of adenocarcinoma which may be part of the same spectrum. Mucinous and signet ring adenocarcinoma, share similar molecular features such as MSI-H, CpG island methylator phenotype-high (CIMP-H), and frequent BRAF V600E mutations. Of course microsatellite instability is linked with Lynch syndrome but in this case immunohistochemistry stains revealed normal mismatch repair proteins MLH1, PMS2, MSH2 and MSH6.
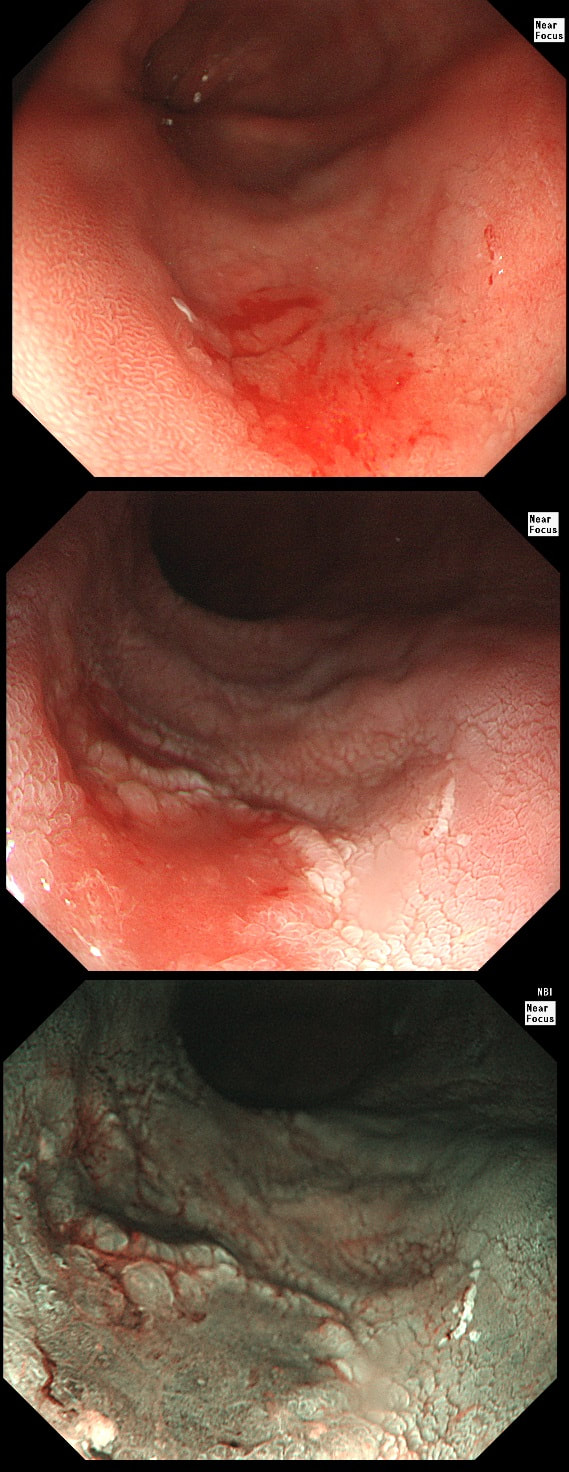
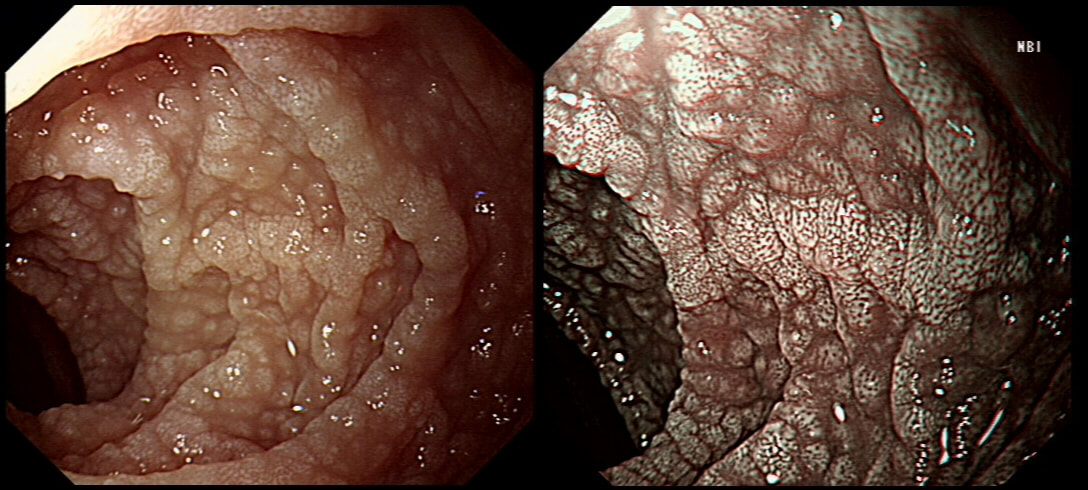
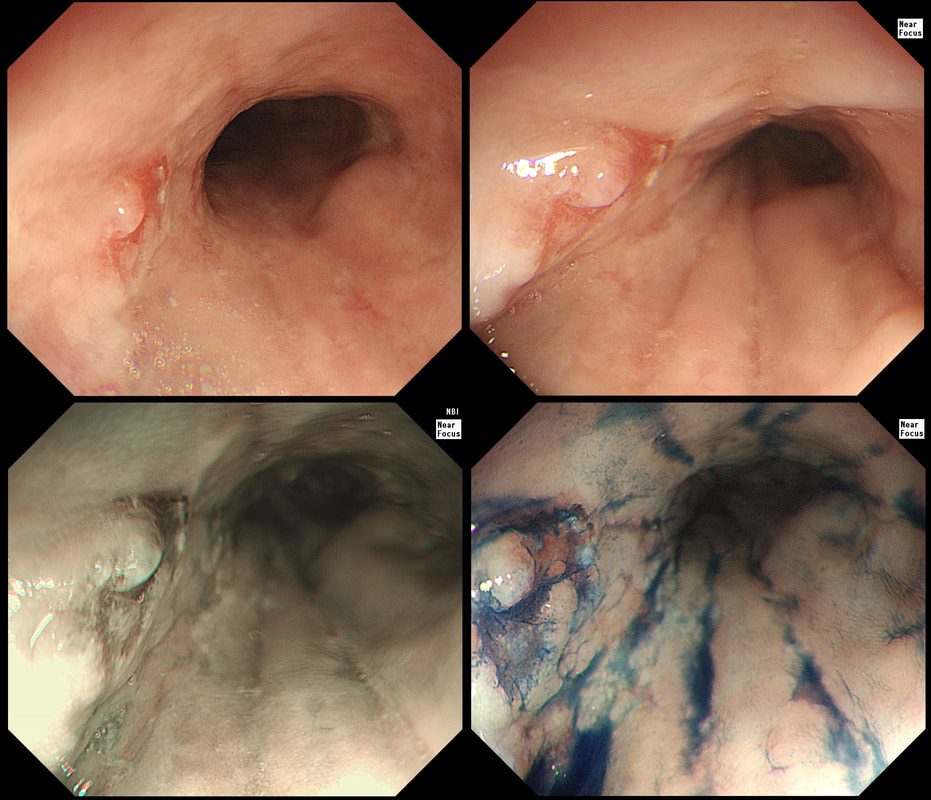
This 'lesion' was barely visible within in a Barrett's segment on white light. However, after acetic acid and with NBI it's more obvious.
WHAT IS THE LIKELY HISTOLOGY?
a) Barrett's LGD
Usually invisible or just a red, flat patch
b) HGD
Subtle change in crypt pattern but with little nodularity?
c) IMca
This was my own guess but it was wrong...
d) Invasive cancer
Yes, superficial invasion (100 microns) and poor differentiation!
explanation
I removed this lesion without worrying too much about the subtle 'depressed' growth pattern and the small, round crypts in the centre of the lesion. However, I was surprised to see the pathology report of a superficially invading adenocarcinoma, with poor differentiation to boot!!! This finding makes the advice on 'further treatment' more complex. As you know, in both the upper and lower GI tract, the finding of 'lymphovascular invasion' (LV) is probably the most 'ominous sign' that a patient needs surgery (or chemo-radiotherapy in case of the oesophagus). Poor differentiation is 'bad', but less bad than LVI.
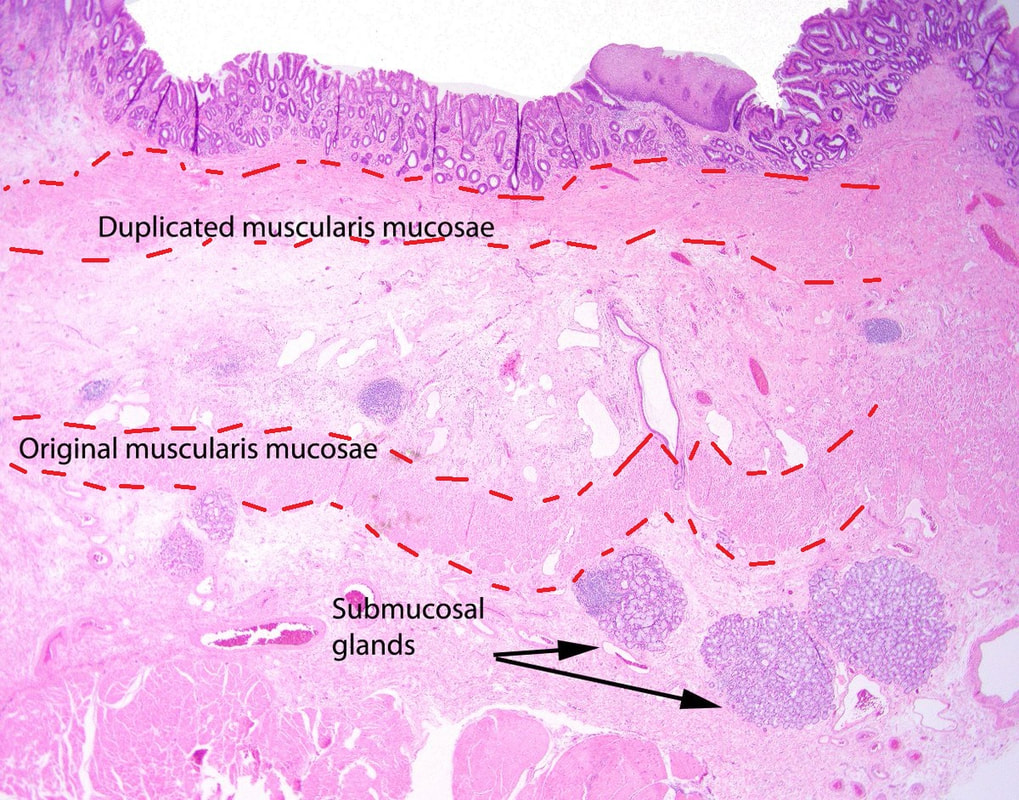
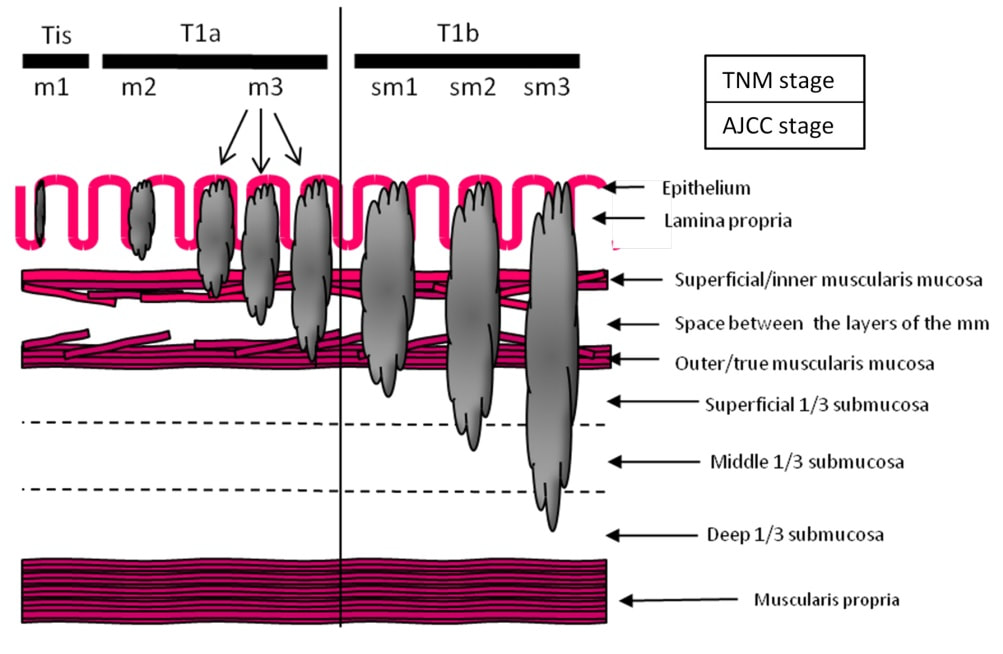
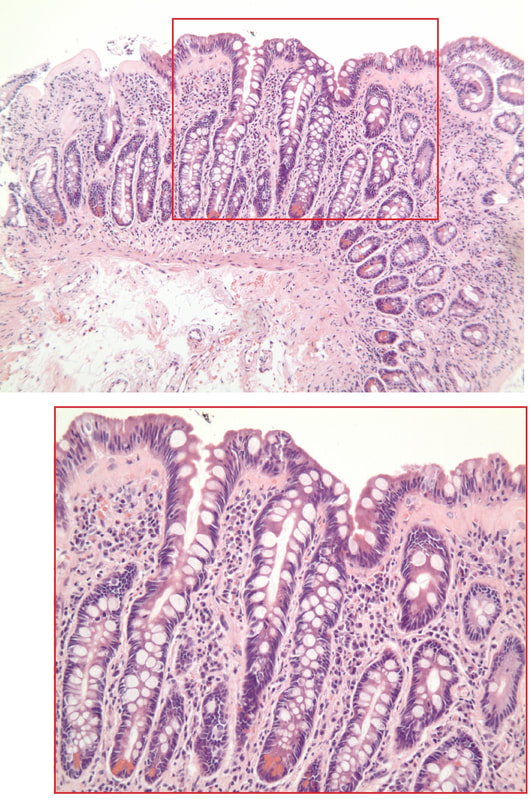
Depth of invasion is also important and in Barrett's you are 'allowed' invasion to about 500 microns below the muscularis mucosa. The corresponding 'safe margin' in SCC's is only 200microns. In this case the depth of invasion was only about 100 microns, leaving 'poor differentiation' as the only 'bad sign'. The patient wasn't a surgical candidate and refused CRT. This was 3 years ago and so far all is well! By the way, the histopathologists do have a more difficult job than you perhaps imagine, measuring the depth of invasion in Barrett's cancer. This is because they often see several bands of muscularis mucosa, so called 'duplication of the muscularis mucosa'. Elsewhere in the GI tract, the muscularis mucosa is a single band of smooth muscle. They measure the depth of invasion from the top of the muscularis mucosa down the the deepest point of invasion. However, if there are several bands of muscularis mucosa, which one do you measure from?!? Below is an example to illustrate the dilemma.
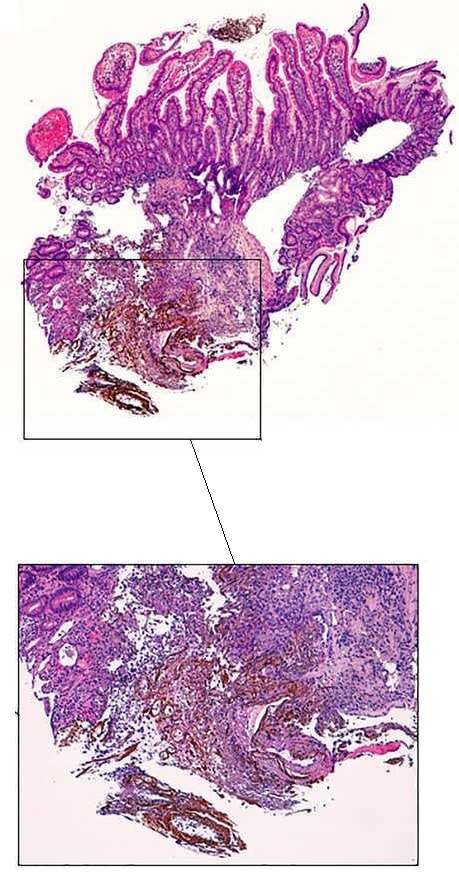
A polyp found in the descending colon and removed as a single fragment (H&E attached)
WHAT IS THE DIAGNOSIS
a) Tubular adenoma
Yes but you are only partly correct!
b) Tubulovillous adenoma
No way, this is a LST-NG!!!
c) Villous adenoma
Absolutely not - there are no villi!!!
d) Sessile serrated lesion
Looks like it but histology doesn't!
e) Malignant polyp
Full marks!
explanation
This is a LST-NG type of lesion (laterally spreading tumour of the non-granular type). They are always TA's (tubular adenomas) and often (but usually not), harbour HGD or cancer. I guess that we can't really be sure about the crypt pattern as this is a non-magnified image. However, looking at the histology slide with narrow crypts, I expect that the crypt pattern is probably IIIs (small round crypts) which goes with TA+HGD. Must admit that I was surprised to find invasive cancer and LVI (lymphovascular invasion) in a small lesion such as this! The last image shows clusters of malignant cells within lymphatics. Of all the 'markers' to suggest that the patient needs surgery, LVI is the most important!
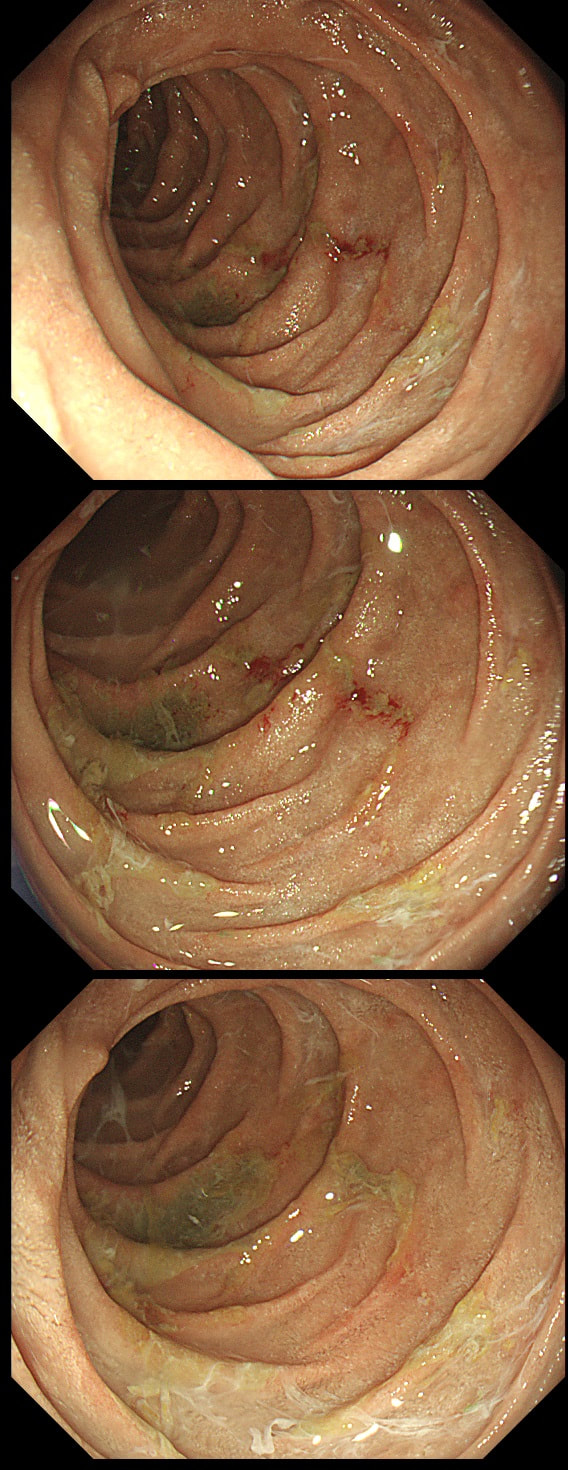
This patient was undergoing a gastroscopy for iron deficiency anaemia. A small ulcer is found in D2.
WHAT IS THE LIKELY AETIOLOGY?
■ Peptic ulcer
Statistically probably most likely diagnosis
■ Pill ulcer
Yes, but what pill?
■ Adenoma
Often do appear depressed here (IIc lesions) but not in this case
■ Early cancer
Early duodenal cancer usually look like sessile adenomas or, less commonly like indurated ulcerated lesions
explanation
If it wasn't for the somewhat odd, elongated appearance of the ulcer, it wouldn't seem odd. However, the only clue to the aetiology is that yellowish appearance. You may be familiar with 'iron pill gastritis' but can you get iron pill duodenitis? Yes you can! It has even been reported in the hypopharynx and the oesophagus. Those dark spots on the histology slides are crystalline iron deposits are easy to spot for the pathologists.
Of course, ferrous sulphate (Fe2+) tablets is the non-toxic form of iron (naturally). However, the tablets can produce mucosal inflammation if the iron becomes oxidised to Ferric iron (Fe3+). You may remember from medical school that most dietary nonheme iron is in the toxic, oxidized form of ferric iron (Fe3+). To absorb iron it first needs to be reduced, in the presence of acid, to ferrous iron (Fe2+). After this, the iron is easily transported across the enterocytes via a divalent metal-ion transporter 1 (DMT1) protein. Here is a link to an open access article if you want to brush up on iron absorption. Naturally, the treatment is to change to the liquid form of iron supplement which is much less toxic to the mucosa than solid iron tablets.
This small polyp was removed from the transverse colon. H&E histology is attached
WHAT IS THE DIAGNOSIS?
■ Serrated polyp
Crypt pattern looks more adenomatous!
■ Adenomatous polyp
Slit-like crypts but that thick capillary doesn't fit!
■ Leiomyoma
You're good at histology!
■ Neuroendocrine tumour
That was my guess too but I was wrong!
■ Small CRC
This lesion is submucosal!
explanation
I was quite sure that this was a small NET. It's that vessel crawling up it's side which convinced me. The reason that I enclose the histology is because it's not a neuroendocrine tumour! Histology shows interlacing bundles of spindle-shaped, smooth muscle cells. with bland-looking nuclei probably arising from the muscularis mucosa just below the epithelium.
It's a leiomyoma, which are very uncommon in the colon but of course very common in the oesophagus. Endoscopically, it's usually not difficult to tell the two apart. NET's often have a dip in the centre and/or sizeable vessels crawling up their sides. Colonic leiomyomas are usually covered with entirely normal looking epithelium. With immunohistochemistry it's easy to tell the two apart. A GIST (thought to arise from the 'interstitial cells of Cajar') usually stain with c-kit (CD117) while a leiomyoma is (thought to originate from smooth muscle cells) stain positive for smooth muscle actin or desmin, but not for c-kit. Of course leiomyomas are subepithelial lesions, covered with a normal mucosa. The differential diagnosis of submucosal swellings in the colon include:
A middle-aged woman presented with loose stool and weight loss. Initially, she refused an endoscopy and a diagnosis of coeliac disease was based a high tissue transglutaminase (tTG) antibody titre. She was started on a gluten-free diet (GFD) but her symptoms remained despite adherence to the GFD. After several months she agrees to undergo an endoscopy. The images show the endoscopic view of the duodenal mucosa and the corresponding histology slide stained with haemotoxylin and eosin (H&E).
WHAT WOULD YOU DO NEXT?
■ Refer the patient for a dietary review
You are assuming that the pt has Coeliac disease...
■ Request a clonal analysis of the intraepithelial lymphocytes
But is the diagnosis of Coeliac correct? Look at the histology again!
■ Refer for HLA DQ2/DQ8 testing
But the diagnosis has been made on the biopsy...
■ Refer the patient for capsule endoscopy
Tissue was the issue and you've got that already!
■ Add prednisolone to the dietary restrictions
Congratulations you spotted the collagen band?
explanation
Initially, the patient appeared to have classic coeliac disease, with diarrhoea and weight loss together with a positive serology. The endoscopy was crucial as the duodenal biopsies identified the presence of a thick band-like deposit of collagen just below the duodenal epithelium. On the basis of this finding, collagenous sprue was diagnosed.
Collagenous sprue was first described in 1947,1 but it was not until 1970 that Weinstein et al. introduced it as a diagnostic term to the medical nomenclature.2 Collagenous sprue is more frequent in females and in individuals who have other autoimmune diseases.3 It is now recognised that collagenous sprue shares similar clinical features with coeliac disease, such as chronic diarrhoea, anaemia and weight loss. In addition, the endoscopic and histological features of both diseases are similar, with an atrophic and scalloped duodenal mucosa. However, the histological hallmark of collagenous sprue is the presence of a thick subepithelial collagen band. Such collagen bands may also be found in collagenous gastritis and collagenous colitis.4 It has been proposed that collagenous sprue may be a heterogenous condition of collagenous gastroenteritides, including conditions such as collagenous colitis and coeliac disease.5,6 Unlike coeliac disease, in collagenous sprue, the typical histological changes may also be found in the stomach and colon.7 Furthermore, greater numbers of IgG4 plasma cells have been reported in the duodenal mucosa of patients with collagenous sprue when compared with the numbers in patients with coeliac disease, duodenitis or normal duodenal mucosa.8 As in our case, patients with collagenous colitis usually also have positive coeliac serology. For this reason, some believe that the collagen band is simply a marker of particularly severe coeliac disease, which may also be associated with ulceration, perforation and T-cell or B-cell lymphoma.9 An interesting report of paraneoplastic collagenous colitis was reported by Freeman et.al.10 In this case a patient with collagen deposits in both the small and large intestine was also found to have a coincidental colon cancer. After surgery, both the malabsorption and the histopathological changes completely resolved! For this reason, a search for underlying malignant disease should be considered. The response to a GFD is usually disappointing and for this reason, the outlook used to be grave for patients with collagenous sprue. However, remission of the condition has been reported with courses of corticosteroids11 or immunosuppressive agents such as infliximab.12 References
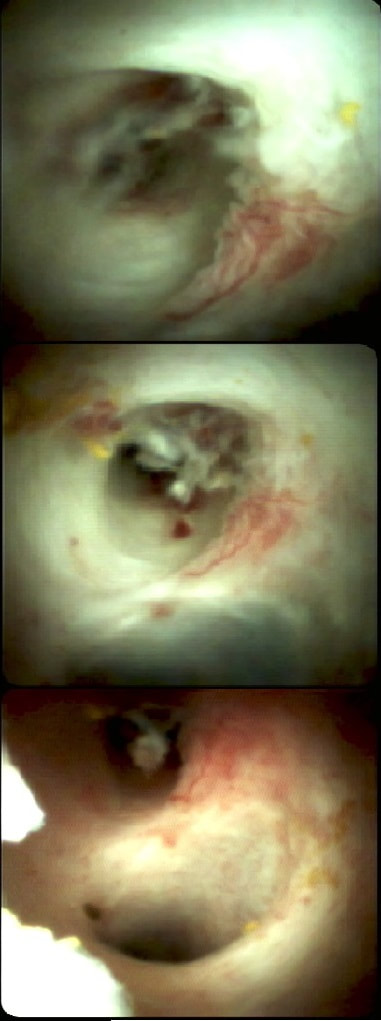
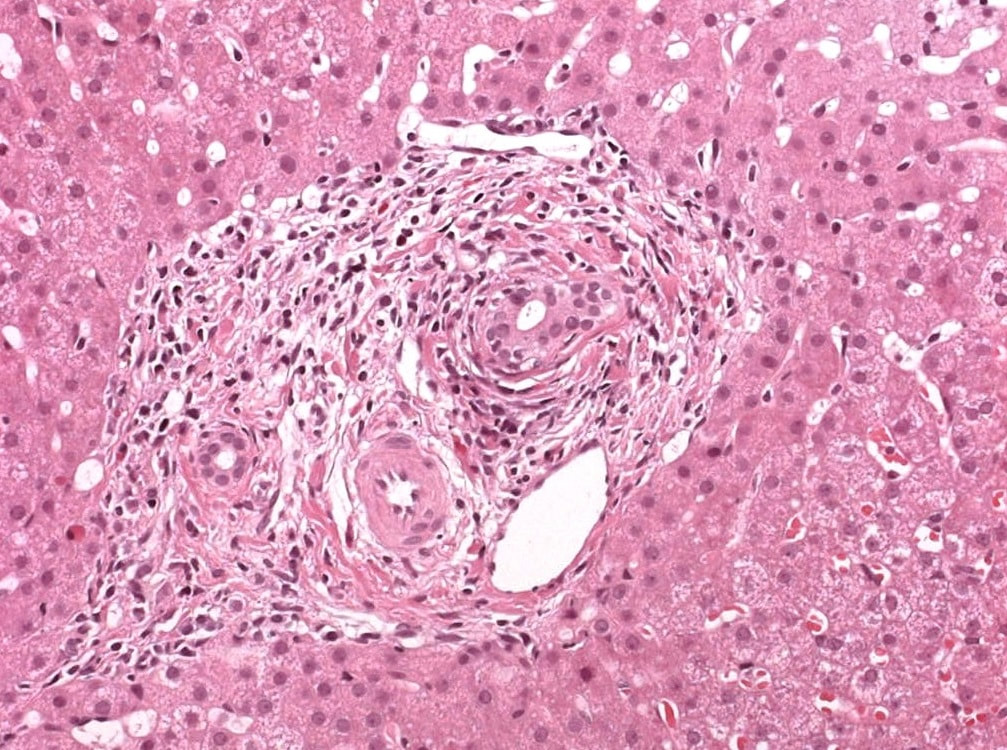
A full house of cholangiogram, cholangioscopy and histology of a 35 year old man with jaundice!
WHAT IS THE DIAGNOSIS?
■ Autoimmune hepatitis
But this should be diagnosed by blood tests?!
■ Alcoholic hepatitis
The last thing a patient would need is an ERCP
■ Primary Billiary Cirrhosis
But it's a man!
■ Primary Sclerosing cholangitis
Got it!
explanation
This is of course something of a curveball. Why would a patient with any of these causes of jaundice undergo an ERCP? Surely a clinical assessment, blood tests and an MRI would be the investigation of choice? Without any risk of giving the patient cholangitis!? Actually, the three images (cholangiogram, cholangioscopy and histology) are from three historical cases ☺!
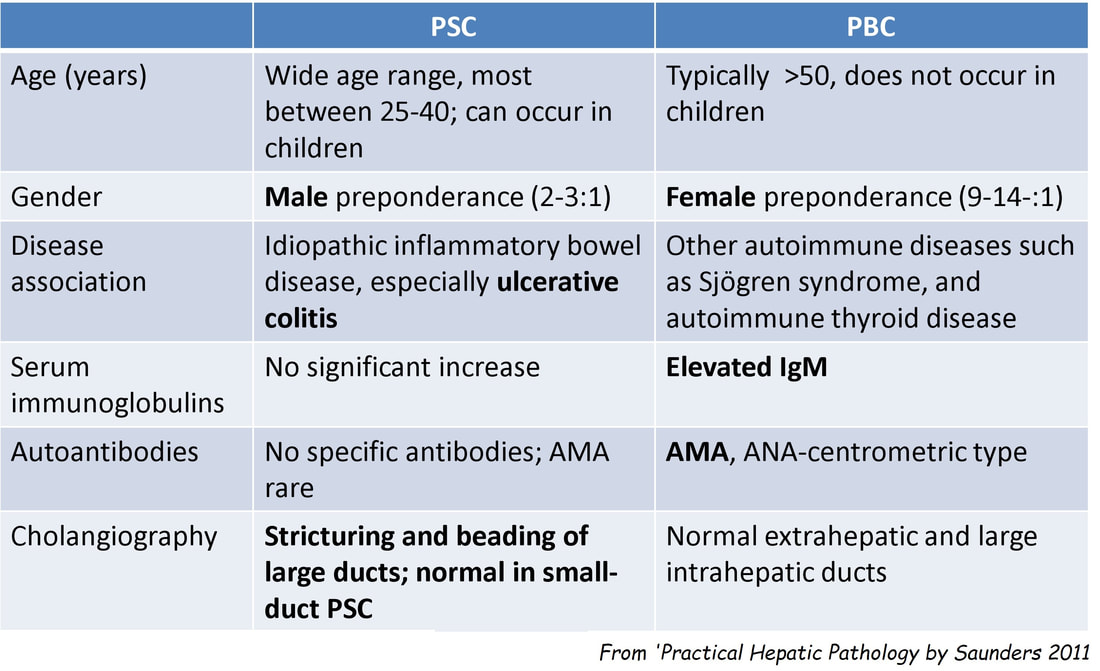
MRI has a sensitivity of about 85% and specificity of around 95% for PSC. In most cases of PSC (87%), both intra and extra-hepatic bile ducts are involved , intrahepatic ducts alone in 10-25% and very rarely only the extrahepatic ducts (2%). Cholangioscopy shows patches of inflammation on the wall of the CBD and the cholangiogram did suggest some 'pruning' of the intra-hepatic biliary tree. A subsequent liver biopsy (we really did go all out), shows the typical peri-portal 'onion skin' inflammatory changes in keeping with PSC (although histology can't usually completely rule out other causes of biliary obstruction).
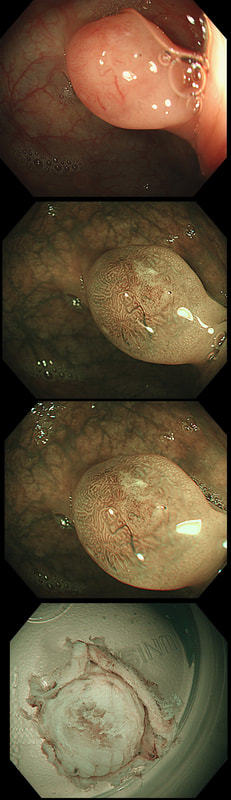
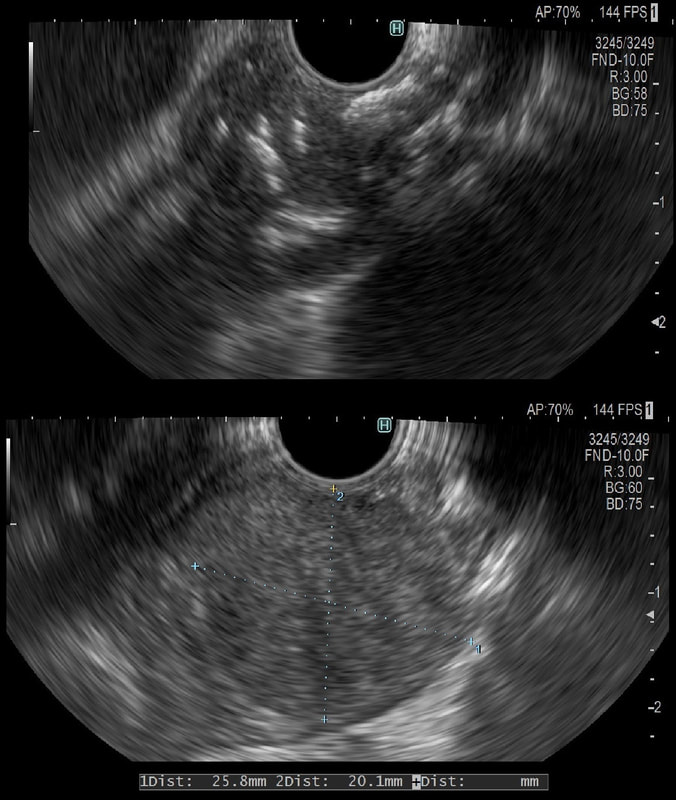
This was an incidental finding in the mid-oesophagus. Subsequently an EUS is organised and the lesion is ultimately removed (histology in the last image)
WHAT IS THE DIAGNOSIS?
■ Neuroendocrine tumour
I've never actually seen an oesophageal NET
■ Granular cell tumour
But it isn't a small white submucosal disc?!
■ Leiomyoma
A shrewd guess!
■ GIST
These are VERY rare in the oesophagus (but common in stomach)
■ Melanoma
Usually look brownish endoscopically!
explanation
Leiomyomas are the most common submucosal lesion found in the oesophagus. No idea why they are so rare in the stomach, where GIST's are the most common submucosal lesion. Correspondingly GIST's are very rare in the oesophagus.
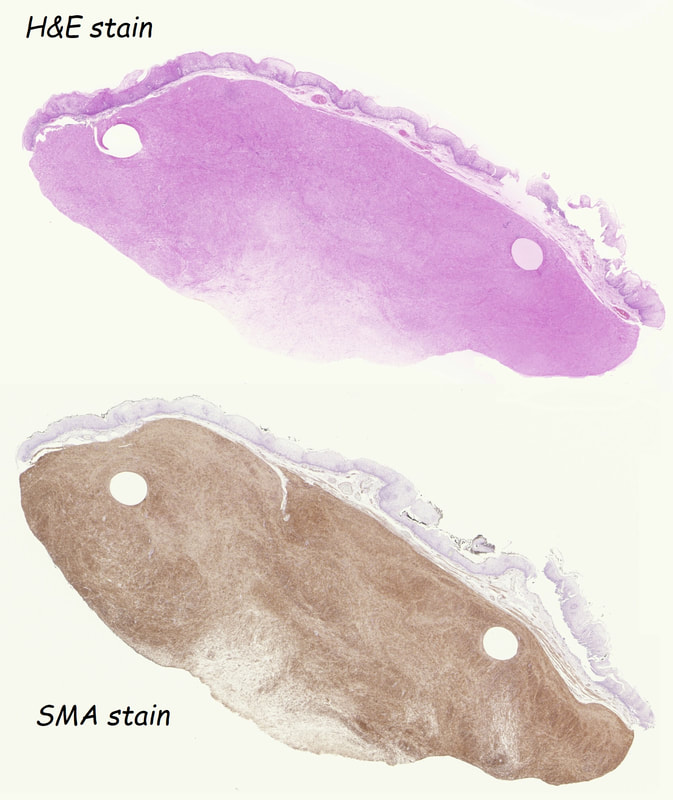
These usually arise from the circular muscle propria layer. On CT they are often calcified and there are of course no nearby nodes to suggest malignant disease. Indeed, malignant transformation is extremely rare and for this reason many centres would ignore a small submucosal lesion such as this, when found in an elderly person. Larger (around 5cm usually) and symptomatic lesions (which are more likely to be leiomyosarcomas) are enucleated surgically. "- They shell out like peas" according to one of our UGI surgeons. However, our local surgeons are also careful to point out that resection usually result in some damage to the muscle propria layer, which require repair as otherwise a diverticulum may develop. With endoscopists spending more time in the 'third oesophageal space' (i.e. the submucosal space), doing ESD's and POEM, the suggestion that we should also remove leiomyomas endoscopically has arisen. May seem like a good idea but I would advocate caution for the simple reason that endoscopically, we are unable to repair the defect we cause in the muscle propria layer ... Oh, how about that 'SMA stain' used in the histology. This is 'Smooth Muscle Actin' stain which does exactly what it says on the tin ...
This ulcer was found on the lesser curve of a middle aged man with dyspepsia. Biopsies are taken and stained with H&E (below)
WHAT IS THE AETIOLOGY OF THAT GU?
■ Benign GU
Don't you think that the mucosa look odd near to the ulcer?
■ Lymphoma
Yes! That odd atrophic mucosa next to the ulcer look weird but not "carcinoma weird" !
■ Adenocarcinoma - intestinal type
With a 'standard cancer', the surrounding mucosa should be thickened and red not atrophic looking
■ Adenocarcinoma - intestinal type
This was actually my own diagnosis but histology shows lots of small round lymphocytes not ballooned up signet ring cells.
EXPLANATION
There are only two possibilities here. Of course the ulcer itself doesn't tell you much. The mucosa surrounding the ulcer tells you more! With a benign ulcer, you would expect a thin rim of 'reactive' mucosa (looks red and a little villous). In the case of an intestinal type of adenocarcinoma a broad band of surrounding mucosa is red, indurated and thickened. In gastric lymphomas, the surface area of abnormal mucosa is usually far greater than the surface area of the ulceration. There are usually areas of atrophic and inflamed mucosa. Mucosa infiltrated by a diffuse type adenocarcinoma could look like this BUT it's unusual for it to ulcerate!
This patient has a MALT lymphoma ! Of course, I took samples for Helicobacters. Of course not every MALT lymphoma is linked with HP infection (about 2/3 cases are though). In this case our haematologists were optimistic of a good response to Hp eradication because all the 3 good prognostic criteria were met:
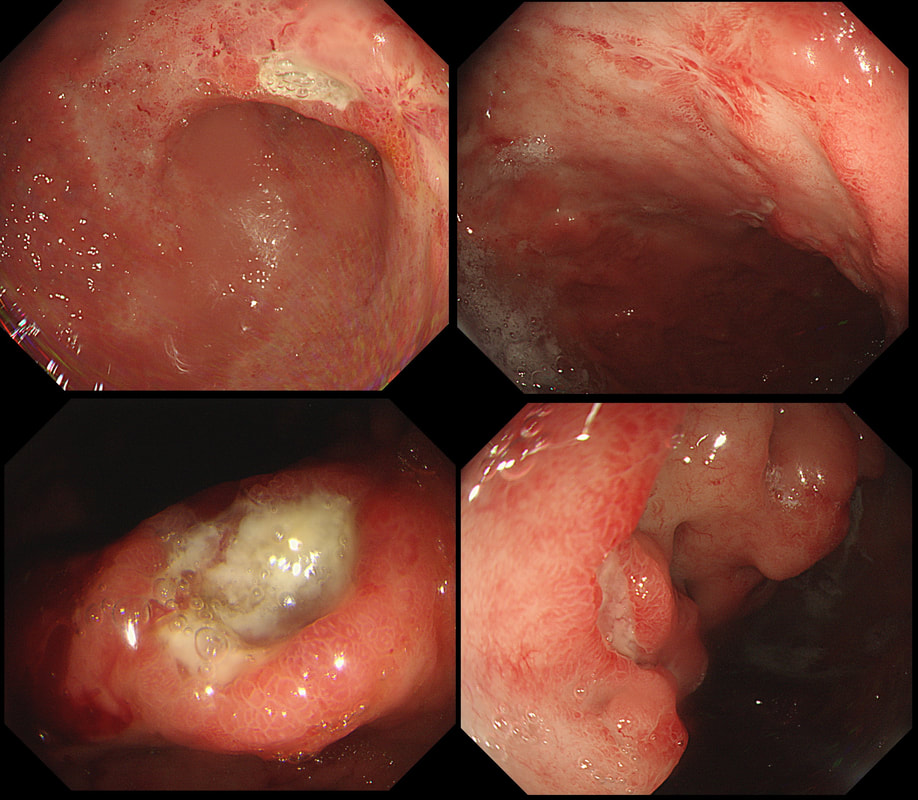
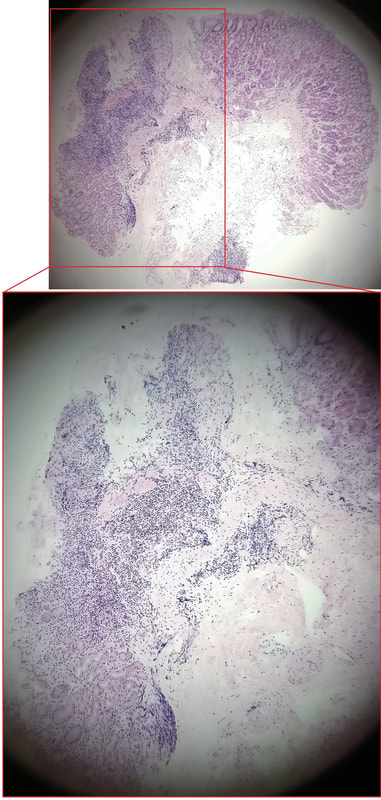
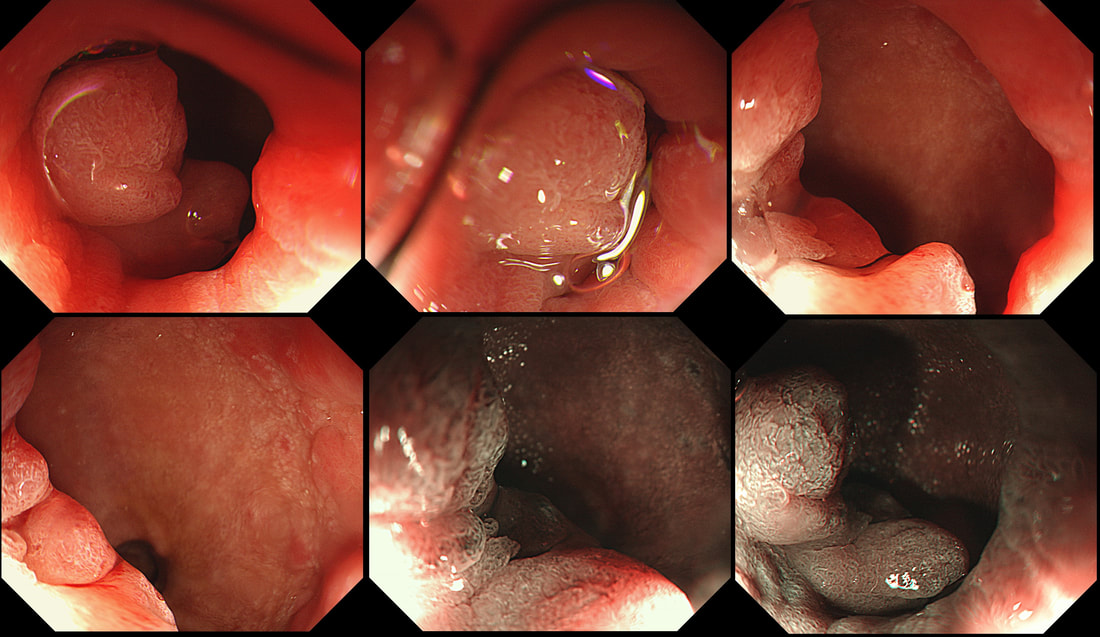
A beautiful polyp removed from the ascending colon. WHAT IS THE LIKELY DIAGNOSIS?
■ Hyperplastic polyp
CORRECT, or rather more precisely an 'inflammatory polyp'
■ Adenomatous polyp
INCORRECT
■ Malignant polyp
INCORRECT!
explanation
This is a typical inflammatory polyp! The pathologists call these inflammatory/hyperplastic polyps. However, for clarity I wish that they would simply call them 'inflammatory polyps'. After all, pure 'hyperplastic polyps' are actually a different entity in the colon consisting of mutated epithelium. In contrast, 'inflammatory polyps' like this one consists of granulation tissue! As you probably know, the white spots are superficial caps of fibrinopurulent ulcer slough overlying small spots of ulceration. It remains a mystery why such a spectacular lump of granulation tissue developed in this particular patient. There was no history of NSAID use but the patient did have 'neurofibromatosis' and the lesion was found at a surveillance examination.
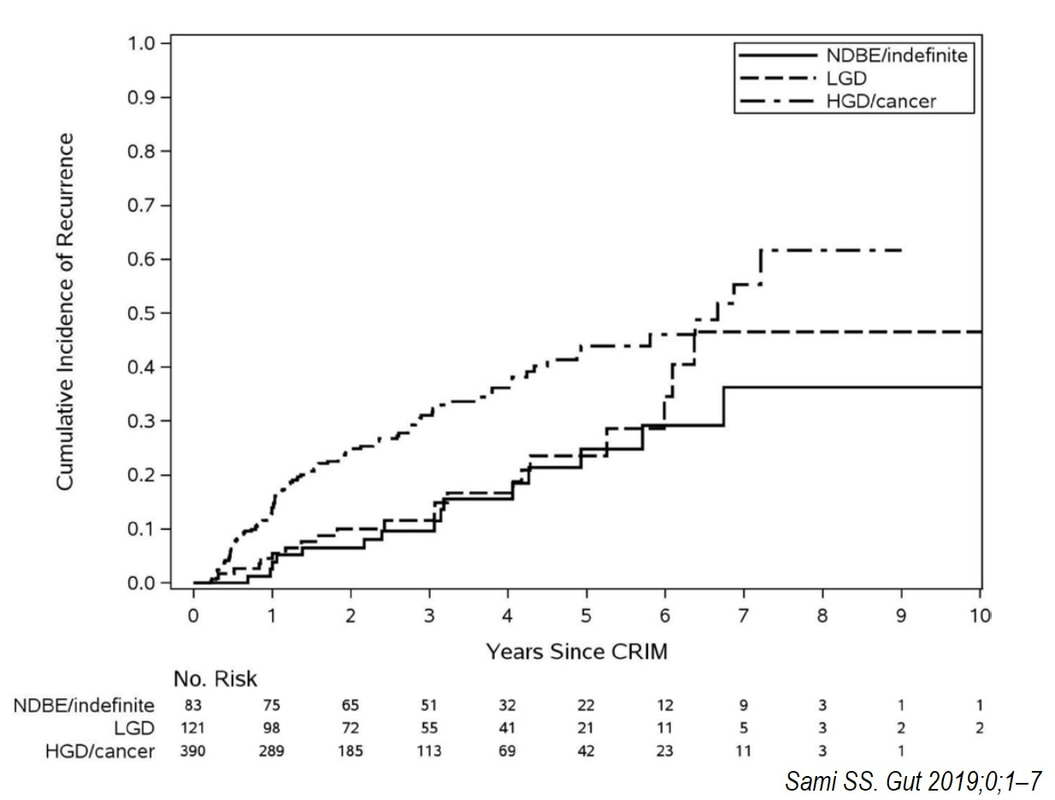
A year ago, this patient underwent RFA for a C5/M6 Barrett's harbouring HGD.
WHAT IS THE MOST APPROPRIATE MANAGEMENT?
■ APC ablation
INCORRECT, the lesion is NOT FLAT!
■ RFA
INCORRECT, because the lesion is too worryingly nodular
■ EMR
CORRECT, the only way to make sure!!!
explanation
This is the problem with RFA, it doesn't last forever! After 5-6 years, about half your patients will have relapsed. Predictably, it's the patients with the most unstable changes who are the most likely to relapse (see graph below from Dr Sami's landmark paper). This is the reason why surveillance at 12 month and then 3 yearly is recommended by the BSG when the 'index histology' was LGD. However, after HGD or IMca, surveillance after 3 months, 6 months and then annually is recommended. Click here to read those recommendations in full.
Endoscopically, it can be difficult to spot a small rim of Barrett's returning, which over time slowly pushes the squamo-columnar junction proximally. In most cases, this is how Barrett's return over time. It's for this reason that we advice taking 4 biopsies from the columnar side of the SCJ'n. If your pathologists report dysplasia in any of these samples, a "top-up" RFA may well be indicated. Less commonly patients develop a nodule emerging from the 'neo-squamous mucosa' as in this case. Clearly you need to resect this for a full histological analysis. Ablation would be like sweeping it under the carpet! The nodule was removed by EMR and histology revealed an intramucosal cancer! Rather worrying that this patient's disease has in some sense, 'progressed' after RFA. Prof Rebecca Fitzgerald's group at Cambridge has shown us that the DNA is pretty thoroughly mashed up by the time the mucosa looks dysplastic down a microscope. Is it really possible for RFA to 'clean up' and remove all those chromosomal translocations ? This odd little polyp glimpsed just beyond the pyloric ring. It was a bugger to remove as it proved impossible to retrovert in the duodenal cap. In the end it was possible by pulling it into my snare, using a double channelled gastroscope. The histology is attached. WHAT IS THE DIAGNOSIS ? a) gastric heterotopia b) pyloric gland adenoma c) duodenal adenoma d) Brunner's gland hyperplasia e) Neuroendocrine tumour Explanation
OK, there is no way to beat around the bush. This turned out to be a 'pyloric gland adenoma'. I don't think that there is any way that I could have distinguished this from a 'normal' duodenal adenoma endoscopically (you also have the histology to help you though). A Brunner's gland polyp is unlikely as the lesion isn't a beautiful, symmetrical submucosal ball, covered with normal duodenal mucosa. Gastric heterotopia is never this 'polypoid' or 'flamboyant'. Finally, a NET is unlikely as these should also be 'balls', classically with a central depression in the duodenum. The first time I came across a 'Pyloric gland adenoma', I objected to the histology because the polyp was situated in the stomach. Our lovely pathologist patiently educated me and explained that in spite of the name, they don't necessarily arise from the pylorus. Instead, they got their name from the fact that they arise from 'pyloric glands' which are found in the oesophagus, stomach, pylorus and the duodenal cap. Furthermore, I didn't know that gastric adenomas are broadly classified into two types: 1) Gastric type and 2) the Intestinal type of gastric adenoma. Gastric-type adenomas are further subclassified into 'pyloric gland adenomas' and 'foveolar-type adenoma'. However, it's the intestinal-type adenomas which we endoscopists usually refer to as 'adenomas' in the upper GI tract. Pyloric gland adenomas are found in patients with Helicobacter pylori associated gastritis and 'chemical gastritis'. However, they are indeed 'neoplastic' (the cells are unstable) and may harbour low-grade dysplasia, high-grade dysplasia or even cancer. If you would like to learn more about 'Pyloric gland adenomas', this is a good link: http://www.archivesofpathology.org/doi/pdf/10.5858/arpa.2013-0613-RS |
Categories
All
|