|
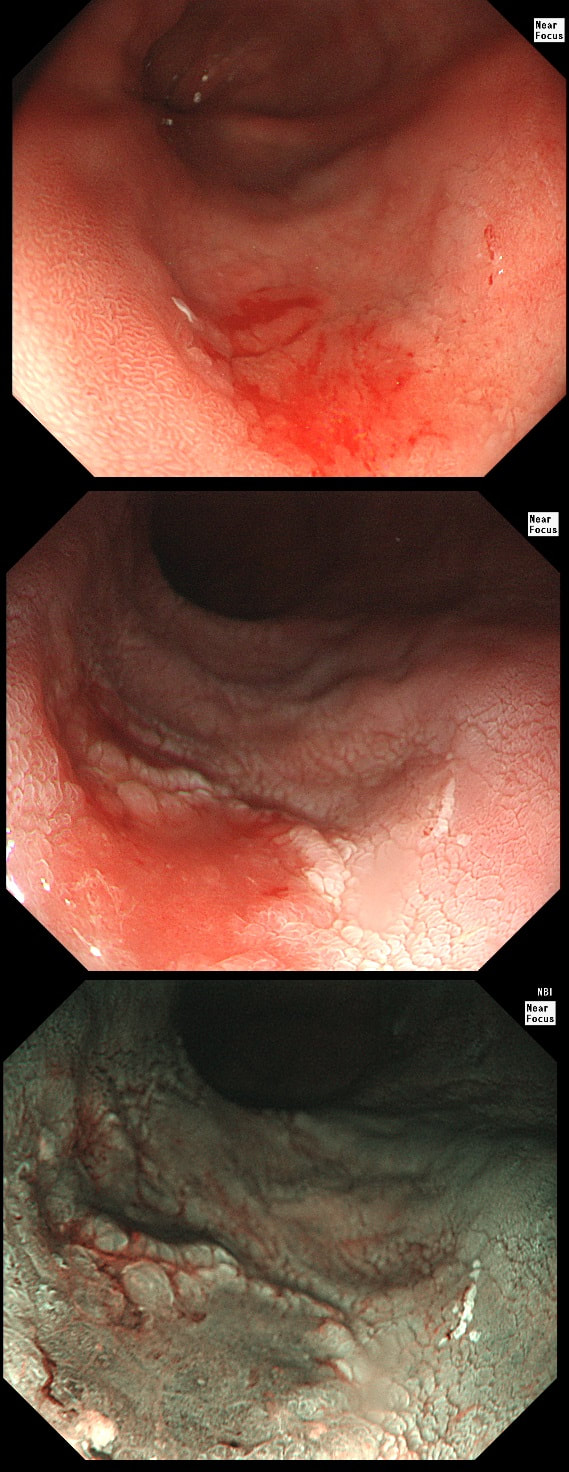
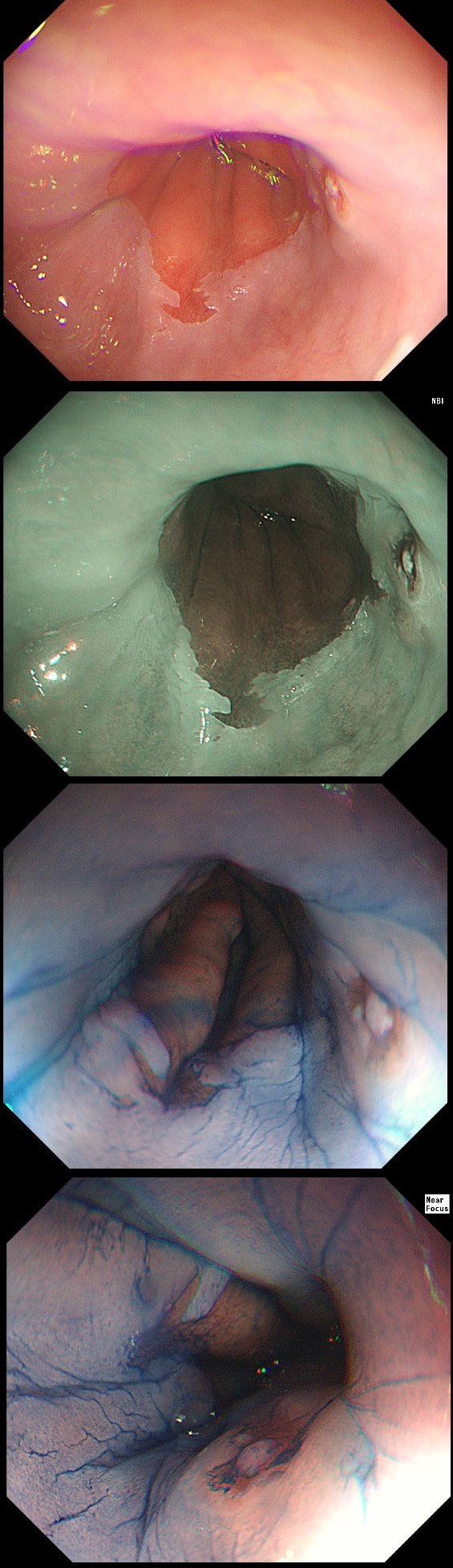
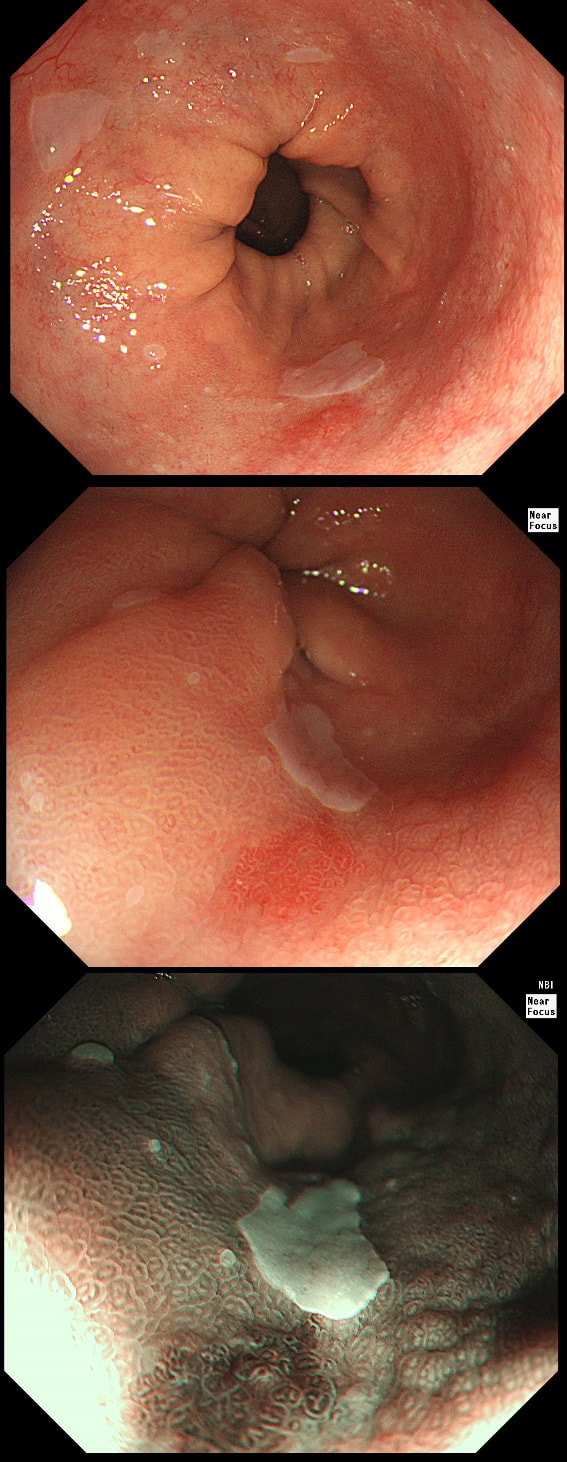
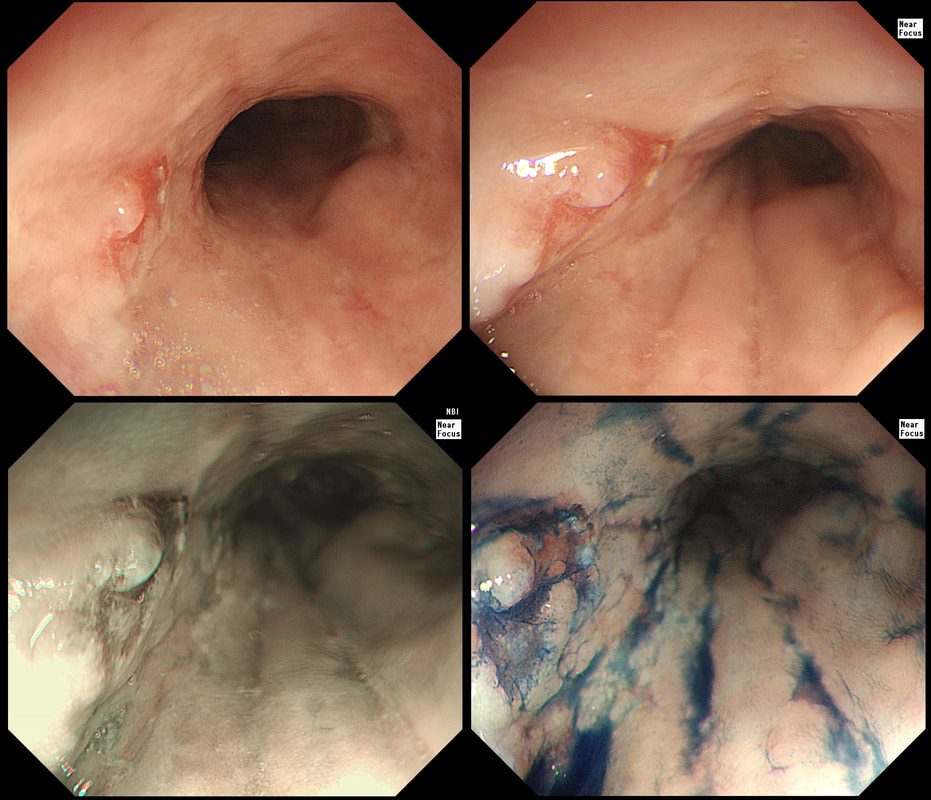
a) Reflux oesophagitis
An irregular squamo-columnar junction does suggest previous reflux oesophagitis but in this case there is more...
b) Non-dysplastic short segment Barrett's
Short-segment Barrett's is defined as Barrett's up to 3cm. In contrast, 'ultra-short Barrett's is less than 1cm. We sized the Barrett's as C0/M2 (according to the Prague classification). However, it doesn't look non-dysplastic!
c) Barrett's with intra-mucosal cancer
This would have been my own guess. There is no 'thickness' to the lesion and there is no ulceration, two features which would suggest that the lesion can't be removed endoscopically
d) Siewert II invasive cancer
Of course it may be invasive but that would be a little surprising. Endoscopically, this is IMca. Similarly HGD is unlikely as there is a lot of mucosal irregularity and you can see something has infiltrated below part of the squamous mucosa in the 2 o'clock position.
explanation
As you know, most of the time Barrett's neoplasia develops on the right-hand side and in the distal (rather than the proximal) Barrett's segment. In this case, in the 2 o'clock position there is a subtle mucosal irregularity. In fact, there appears to be something infiltrating below the squamous mucosa at this location. Biopsies had suggested IMca and a CT had been reassuring. Because the lesion is endoscopically resectable, we didn't bother with an EUS. The lesion was removed and confirmed as a poorly differentiated adenocarcinoma with signet ring morphology. On the other hand, all margins were clear and there was no LVI (lympho-vascular invasion). In the hierarchy of poor prognostic features, LVI and depth of invasion (>500µm) are the most important whilst poor differentiation is the least important. Nevertheless, I was a little worried when the patient declined surgery...
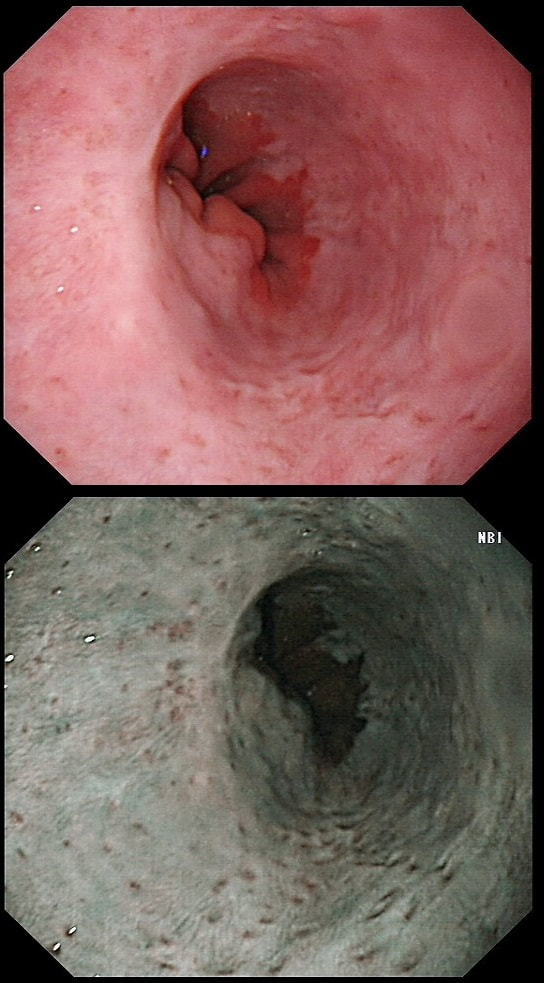
This 'lesion' was barely visible within in a Barrett's segment on white light. However, after acetic acid and with NBI it's more obvious.
WHAT IS THE LIKELY HISTOLOGY?
a) Barrett's LGD
Usually invisible or just a red, flat patch
b) HGD
Subtle change in crypt pattern but with little nodularity?
c) IMca
This was my own guess but it was wrong...
d) Invasive cancer
Yes, superficial invasion (100 microns) and poor differentiation!
explanation
I removed this lesion without worrying too much about the subtle 'depressed' growth pattern and the small, round crypts in the centre of the lesion. However, I was surprised to see the pathology report of a superficially invading adenocarcinoma, with poor differentiation to boot!!! This finding makes the advice on 'further treatment' more complex. As you know, in both the upper and lower GI tract, the finding of 'lymphovascular invasion' (LV) is probably the most 'ominous sign' that a patient needs surgery (or chemo-radiotherapy in case of the oesophagus). Poor differentiation is 'bad', but less bad than LVI.
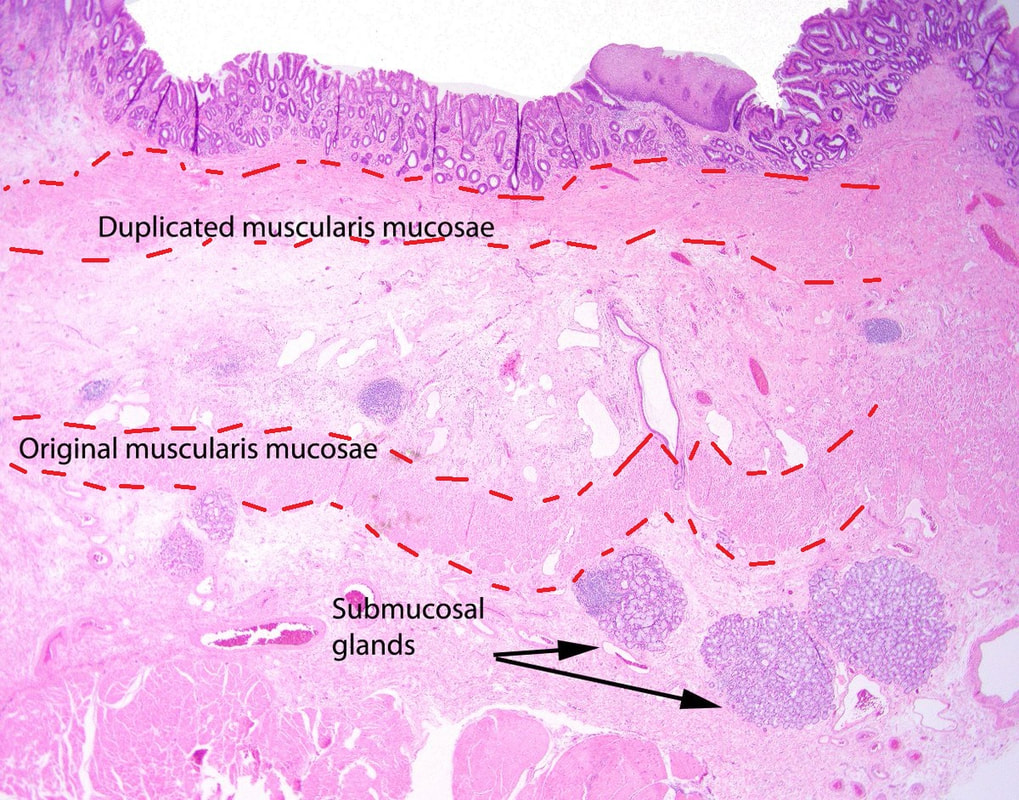
Depth of invasion is also important and in Barrett's you are 'allowed' invasion to about 500 microns below the muscularis mucosa. The corresponding 'safe margin' in SCC's is only 200microns. In this case the depth of invasion was only about 100 microns, leaving 'poor differentiation' as the only 'bad sign'. The patient wasn't a surgical candidate and refused CRT. This was 3 years ago and so far all is well! By the way, the histopathologists do have a more difficult job than you perhaps imagine, measuring the depth of invasion in Barrett's cancer. This is because they often see several bands of muscularis mucosa, so called 'duplication of the muscularis mucosa'. Elsewhere in the GI tract, the muscularis mucosa is a single band of smooth muscle. They measure the depth of invasion from the top of the muscularis mucosa down the the deepest point of invasion. However, if there are several bands of muscularis mucosa, which one do you measure from?!? Below is an example to illustrate the dilemma.
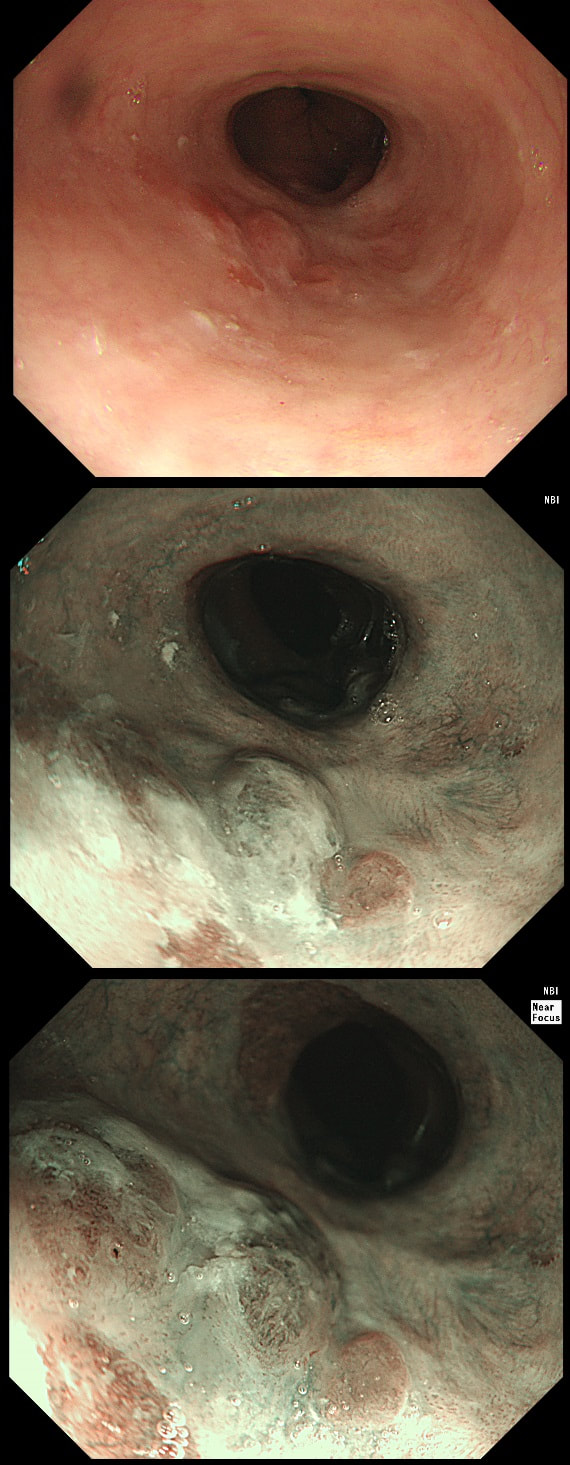
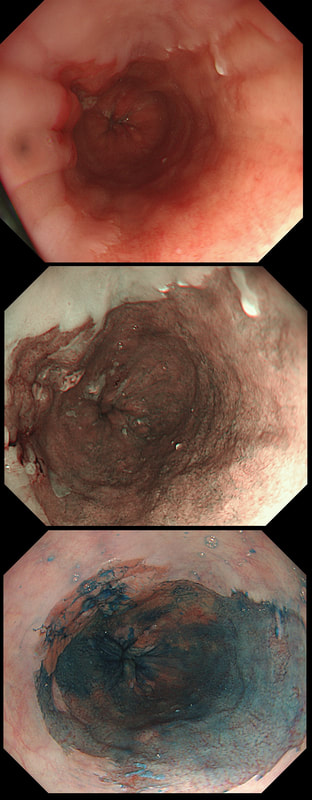
Patient with Barrett's harbouring HGD has been treated with RFA. He has now returned for the second RFA session when this is found.
WHAT IS YOUR ENDOSCOPIC DIAGNOSIS?
a) Benign scarring
Doesn't look benign - Looks evil!
b) Local recurrence of non-dysplastic Barrett's
Barrett's are re-emerging in several places. Doesn't look benign!
c) Barrett's adenocarcinoma
Absolutely! Nodules are never allowed in Barrett's!
explanation
Actually, there is a re-emergence of several brown nodules below the squamous mucosa. Biopsies confirmed this as an invasive adenocarcinoma, re-emerging from below the 'neo-squamous mucosa'. Clearly, to try RFA again would be a mistake! The histology showed a 'poorly differentiated' cancer and we are recommending either surgery or chemo-radiotherapy next (CRT). Even if histology hadn't shown poor differentiation, this 'smells' like bad disease to me which we may well 'undertreat' endoscopically.
This patient attends for Barrett's surveillance WHAT IS YOUR DIAGNOSIS?
a) Barrett's without dysplasia
A nodule within Barrett's is NEVER 'normal'!
b) LGD
LGD is flat, not nodular!
c) HGD
Good guess! Although your guess actually proved to be wrong
d) IMca
Small crypts + nodule = correct!
e) invasive cancer
Nodule should have been larger and without those small crypt openings
explanation
My 'rules of thumb' when assessing Barrett's is as follows: 1) crypts look different in a distinct patch but the area is flat = LGD 2) areas of superficial ulcers within the Barrett's = widespread LGD 3) crypts look different and there is a distinct nodule = HGD 4) crypts are tiny small and there is a nodule = IMca 5) there are no crypts and there is larger nodule = invasive cancer Of course this isn't fool proof ! After all, it's very difficult to distinguish HGD from IMca even on histology! However, it gives you a starting point on how to assess Barrett's and what you should enter on that Histopathology request form. Remember that your pathologists need your help! WHERE IS THE BARRETT'S CANCER?
a) 12 O'clock
Eagle eye!
b) 3 O'clock
But the round crypt openings say otherwise!
c) 6 O'clock
Nope!
d) 9 O'clock
Where there is hardly any Barrett's?!
explanation
Of course the lesion is situated at 12 O'clock. There you can see a subtle mucosal nodularity with an irregular vessel pattern. It was removed by 'suck within the cap' EMR and confirmed as an IMca.
Patient was complaining of indigestion and reflux symptoms.
WHAT IS YOUR DIAGNOSIS?
a) Normal oesophagus
Not quite normal is it?
b) Reflux oesophagitis
This doesn't look like reflux!
c) Adenocarcinoma
Actually the correct answer!
d) SCC
Red nodules, even if surrounding by squamous mucosa are adenocarcinomas (usually)
explanation
You may be surprised to hear that the small nodule at 3 O'clock turned out to be an IMca! It was removed endoscopically. Of course, it's the question then arises; "Should we offer RFA". The BSG recommends this for patients with Barrett's harbouring flat dysplasia. However, in this case there is only a tiny, tongue of Barrett's in the 6 O'clock position! Actually, we just gave this a quick blast of APC (at a fraction of the cost of RFA) and it was gone. However, the patient remains on annual surveillance!
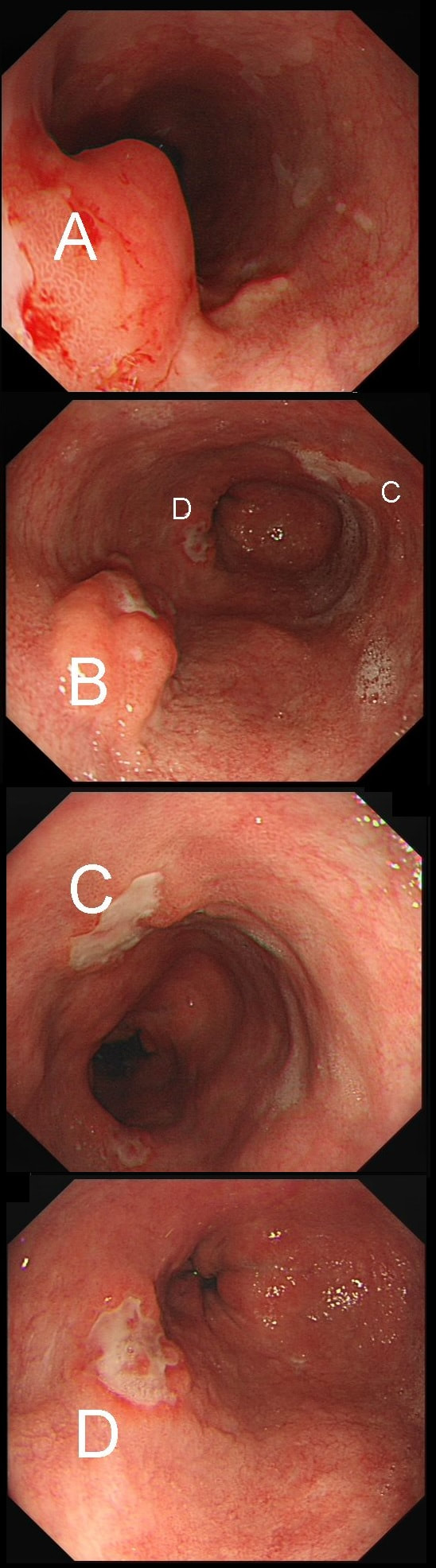
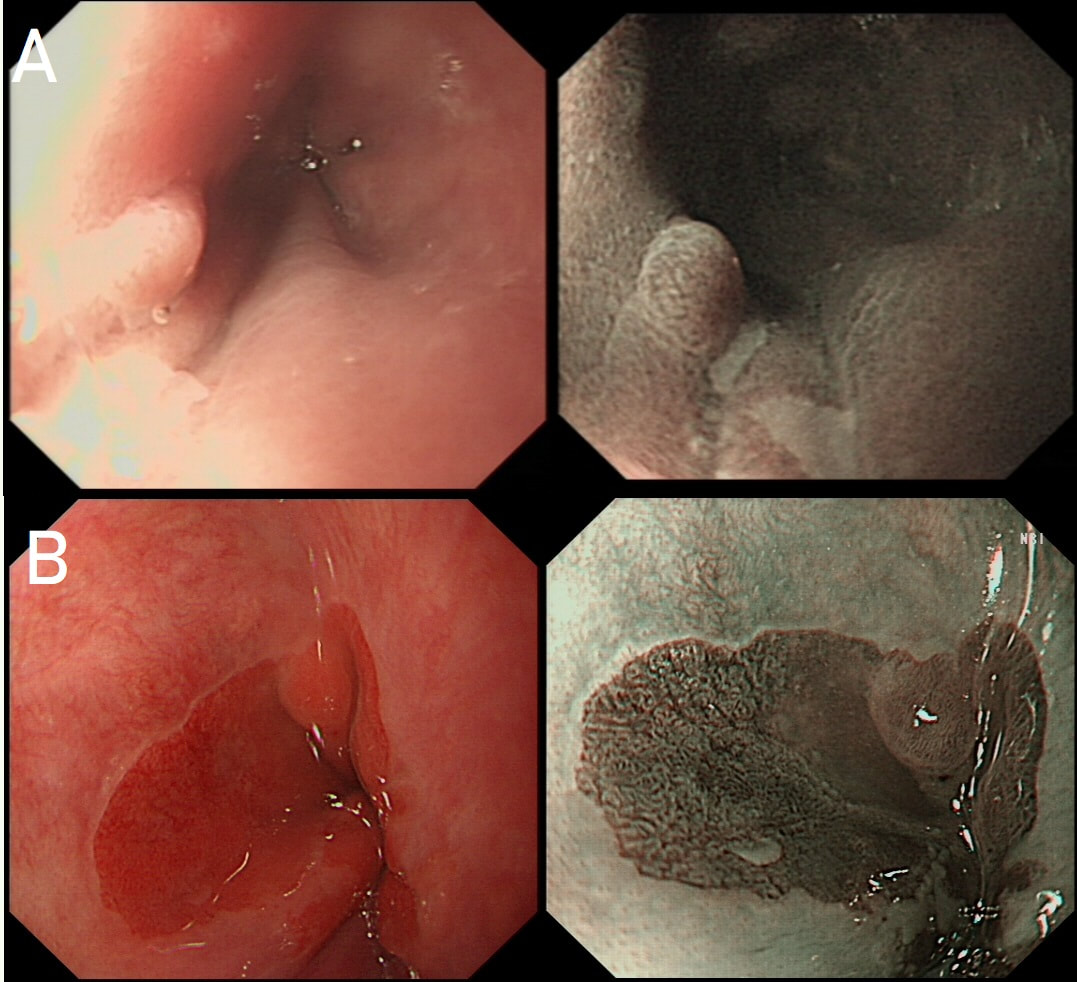
55 yr pt with a Barrett's nodule is referred for an endoscopic resection. I'm surprised to find 4 abnormalities within his 10cm stretch of Barrett's
WHICH OF THESE IS THE MOST LIKELY TO BE ENDOSCOPICALLY RESECTABLE?
a) Lesion A
I agree! Of these 4, A is the only one which isn't ulcerated and therefore is least likely to be invading deeply.
b) Lesion B
Would be my second guess as the ulcerated area seem superficial
c) Lesion C
Don't like the ulceration!
d) Lesion D
Would be my least favourite lesion to attack as the ulceration suggests deeper invasion and poor lift into my cap.
explanation
This may be something of a record, 4 synchronous lesions! Clearly A, B and D are malignant. At first, ulcer C seem more innocent without an elevated edge but on closer assessment, it also has a slightly elevated rim.
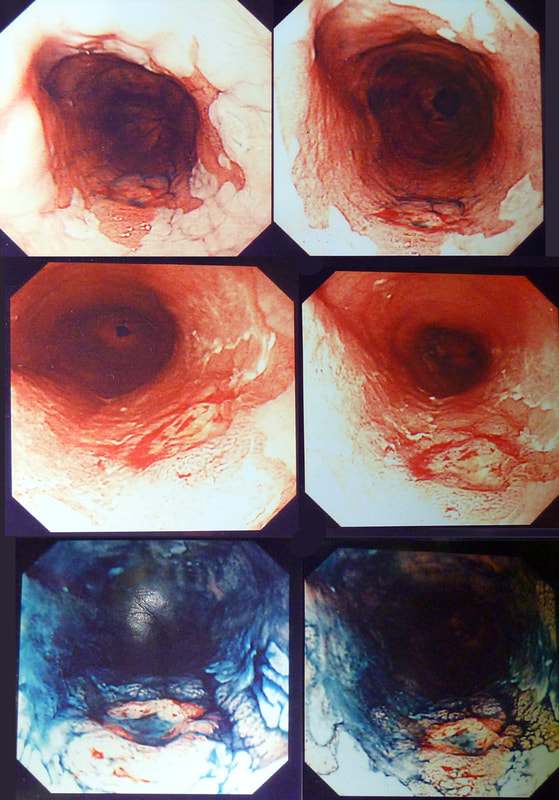
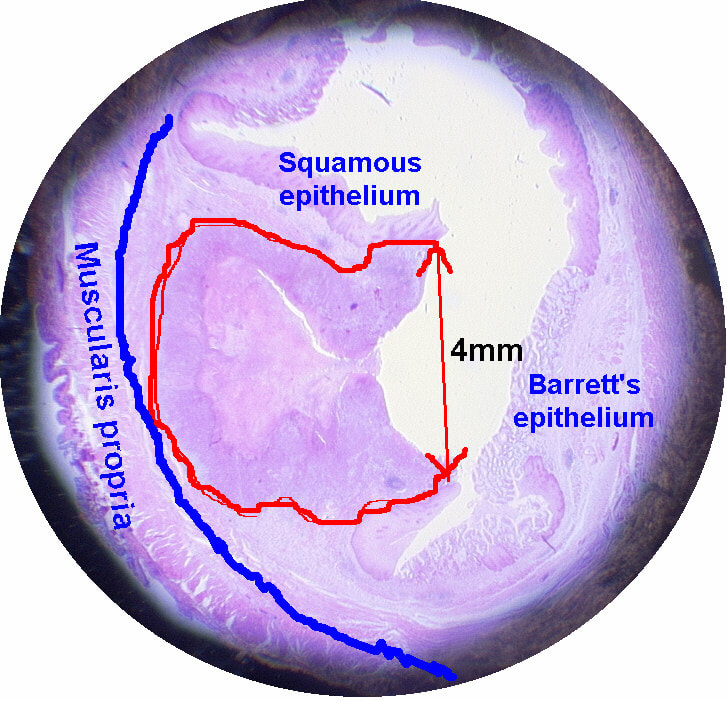
Of these 4, A is the only one which isn't ulcerated and therefore is least likely to be invading deeply. Of course, they are assessed together as there is no point in EMR'ing one only. Either all are curable endoscopic means or none are ! Rather than going ahead with attempting to resect these, I actually bailed out and took samples from each lesion. Biopsies showed invasive, poorly differentiated adenocarcinoma at each location! Clearly, this patient has multifocal 'bad disease' which endoscopy is unlikely to cure in my opinion. I believe that surgery is a far better option and the patient is currently awaiting his oesophagectomy. If you still are not convinced of the pitfalls in trying to deal with ulcerated Barrett's lesions, have a look at the lesion below. Two rounds of sampling had indicated that the lesion harboured HGD. However, I failed to remove the lesion and ultimately the patient underwent an 'Ivor-Lewis'. You can see the histology yourself. The 4mm surface is literally the tip of the iceberg and below you can see the cancer (red line) invading up to the muscle propria. This patient attended for dilatation of his biopsy confirmed peptic oesophageal stricture. Clip has been speeded up somewhat. WHAT WOULD YOU ORGANISE NEXT?
■ A clinic appointment
To ask pt about the swallowing? Hmm, somethings missing!
■ Another dilatation in 2 weeks time
Stricture not very tight and the dilatation should last longer
■ an early follow up OGD for biopsies
The mid-oesophageal stricture is peptic but what of the mucosa below?
explanation
The background to this case is a recent audit which we did in Leeds on missed upper GI neoplasia. We have had several instances of oesophageal cancers being missed when the endoscopist focused too much on the 'task in hand'. For example, we have had several SCC's missed when the endoscopist was carrying out a Barrett's surveillance endoscopy. This is another example of a missed (intramucosal) adenocarcinoma, glimpsed about 10 seconds into the clip in the 3 O'clock position. With mid-oesophageal peptic strictures there is often a stretch of Barrett's below which of course must be assessed and sampled at the earliest convenience. Of course, one can argue that when the job is to do something therapeutic, such as placing a PEG or removing a large polyp, it is 'permissible' to miss a cancer elsewhere. After all, the objective is not to undertake a careful diagnostic examination but to 'do a job'! I'm a strong believer that 'diagnostic' examinations and 'therapeutic' examinations must be clearly separated when you are looking at 'missed lesions. There is a distinct 'therapeutic window' during all endoscopic procedures done without a general anaesthetic. In the upper GI tract, it's up to 20-30 minutes and in the colon I think that it's up to 45-60 minutes. Your patient will not thank you for wasting the valuable minutes of your therapeutic window on carrying out a full diagnostic examination. Of course, after your therapeutic procedure you should consider if an early diagnostic gastroscopy or colonoscopy is needed. But carrying out a full assessment of the squamous portion of the oesophagus at the time of a Barrett's surveillance examination doesn't add much to the procedure. Similarly, when a mid-oesophageal peptic stricture is found, one should realise that the reason that the peptic stricture is in the middle of the oesophagus and not the gastro-oesophageal junction, is probably that there is a a Barrett's segment below the stricture. It doesn't add much time to the procedure and that IMca could have been spotted earlier!
During a Barrett's surveillance OGD, I notice this small red spot at 6 O'clock.
WHAT IS THE LIKELY HISTOLOGY?
■ Reflux ulcer
I don't think that you get this within Barrett's without dysplasia!
■ LGD
Clear crypt pattern, not that different from surrounding mucosa? Good guess!
■ HGD
Quite possible and would have been my guess IF there was some nodularity associated
■ Early cancer
Impossible as lesion has an organised crypt pattern!
explanation
Previous surveillance samples, 3 years earlier had not revealed any dysplasia. However, that red spot shouldn't be there! A reflux ulcer is unlikely as the Barrett's is there because it protects the food pipe from acid. Of course it does this by developing crypts with Goblet cells which secretes protective mucus (of course the pathologists call this process 'metaplasia'). Early texts usually stated that ulceration is common within Barrett's. In my experience, ulceration within Barrett's only happens when dysplasia develops. With the arrival of dysplasia, you begin to see a disappearance of the Goblet cells and of course when the Goblet cells start to dwindle, so does the protective mucus.
In the bottom zoom image you can see that there is a distinct surface crypt pattern at the site of the red spot. Therefore the cells can't be too disorganised, making cancer unlikely. Actually this turned out to be a spot of LGD. I would have guessed HGD/IMca (pathologists struggle to tell the difference) if there was a visible nodule at the site of the red spot. Isn't it interesting that even at the very earliest stage of dysplasia, the unstable cells signal the need for more oxygen to nearby vessels? This is the GOJ of a 60 yr old woman with dyspepsia. WHAT IS YOUR DIAGNOSIS?
■ Reflux oesophagitis
No this is a nodule not an ulcer!
■ Hyperplastic/reactive nodule
Hyperplastic nodules have villous surface!
■ Unremarkable Barrett's
A nodule within Barrett's is always suspicious!
■ Adenocarcinoma
Absolutely!
■ Squamous cell carcinoma
No abnormal vessel pattern within nearby squamous mucosa!
Explanation
Actually, this patient was on a Barrett's surveillance programme. Five years earlier a small IMca had been removed following which annual surveillance had been reassuring. However, then this lesion was found and referred for removal.
Histological assessment of the resection fragment reported early invasion into the submucosa (which is 'allowed' provided that the depth of invasion is less than 500 microns (0.5mm). There was no LVI but unfortunately, there was poor differentiation. Of course, the 'worst' feature to find is probably LVI. Poor differentiation is worrying but less worrying than LVI. The reason for this is that 'poor differentiation' is a rather poorly defined entity. For example, how many crypts should be involved to call something 'poor differentiation' rather than 'focally poor differentiation'? Anyway, the patient was young and ultimately underwent an Ivor-Lewis which confirmed that the EMR had been curative with only some HGD remaining within the residual short stretch of Barrett's. We urgently need a prospective study following this type of Barrett's patient closely over time with regular EUS and CT to see if we can detect the small number of patients who subsequently turn out to have lymphnode involvement This patient has been referred for an oesophageal dilatation after biopsies have been reassuring WHAT WOULD YOU DO?
■ Abort and take more samples
You are a wise as a fox!
■ Dilate to 12mm
Hmm, cautious but not cautious enought !
■ Dilate to 15mm
Seems reasonable but you've missed something!
■ Dilate to 18mm
You are clearly brave but look again!
■ Dilate to 20mm
If this stricture was benign, I would also aim for 20mm but it isn't!
explanation
Of course this stricture doesn't look right! There is a peculiar plaque-like area in the 10 O'clock position. The dilatation was cancelled and another set of samples were requested together with a CT. Samples indicated that this was a case of poorly differentiated intramucosal cancer and an EMR was organised. Have a look at the clip below. Clearly this lesion is firmly tethered to underlying structures and is beyond endoscopic cure. Some options may pop into your head such as injecting some fluid below the lesion or using the 'pull-within-the-snare' EMR technique. The problem is that these will not change the basic fact that endoscopically the lesion is beyond endoscopic cure! You run the risk of 'muddying the waters' with scrappy histology reporting 'intramucosal cancer at least' and with uncertain margins. Far better is to recognised the endoscopic irresectability of the lesion which provides a clear steer towards the 'next treatment level'. In this case the patient wasn't a surgical candidate and was offered chemo-radiotherapy (CRT).
The squamous portion of this oesophagus looks very unusual !
WHAT IS THE LIKELY REASON FOR THIS APPEARANCE?
■ Previous reflux disease
Nope, that's not it!
■ Previous EMR
Gives a pale scar, usually covered with squamous mucosa
■ Previous RFA
Absolutely. You've also seen it?
■ Previous radiotherapy
Gives telangioectatic vessels!
explanation
This patient had undergone RFA, leaving the squamous mucosa looking a little odd. It's actually the openings of the 'oesophageal glands' which you can see. In my experience, there is no point in taking any samples as histology will be reported as normal. In most cases, Barrett's returns at the gastro-oesophageal junction. The return is slow and subtle and for this reason you should take samples from what appears to be the 'columnar side' of the squamo-columnar junction. As intestinal metaplasia (Goblet cells to you and me) are common at the cardia, the pathologists will not be able to tell you if the Barrett's is returning. However, they CAN tell you if the epithelium look dysplastic again!
In a minority of cases, perhaps 1:4 the recurrence happens below the squamous mucosa which can be very hard to see. Of course, the only way to deal with 'buried Barrett's', is to remove the patch by EMR! This 65 yr old patient attends following EMR of an IMca for RFA WHAT WOULD YOU DO NOW?
■ Go ahead and RFA
No that nodule is a show-stopper!
■ First take another set of biopsies
Or go ahead and EMR!
explanation
The BSG guideline as of 2013 (and updated on the topic of RFA for LGD in 2015), is very simple; RFA may be offered for flat dysplasia. The reality is often a little less clear-cut. For example, dysplastic Barrett's often appear slightly lumpy or with areas of superficial ulceration. When does a 'bumpy Barrett's' cross over into 'non-flat dysplasia'? I think that this is an example of 'not flat enough for RFA'. In fact, analysis of the original EMR specimen had reported a positive lateral margin! Expecting a local recurrence I actually went straight ahead and EMR'd the nodule which was confirmed as IMca. You will get the odd patient with Barrett's who fails to get his RFA because each time there is another little nodule to remove. Of course there are no hard and fast rules but these patients probably have 'bad disease' and are better served by an oesophagectomy. Of course, many of these are too old for surgery in which case you have no option but to keep nibbling away at their Barrett's mucosa until some adverse histological feature sends them along for chemoradiotherapy (CRT). In my experience these patients often don't do well. When chromosomal copy number analysis becomes a mainstay tool, I think that we'll see why ...
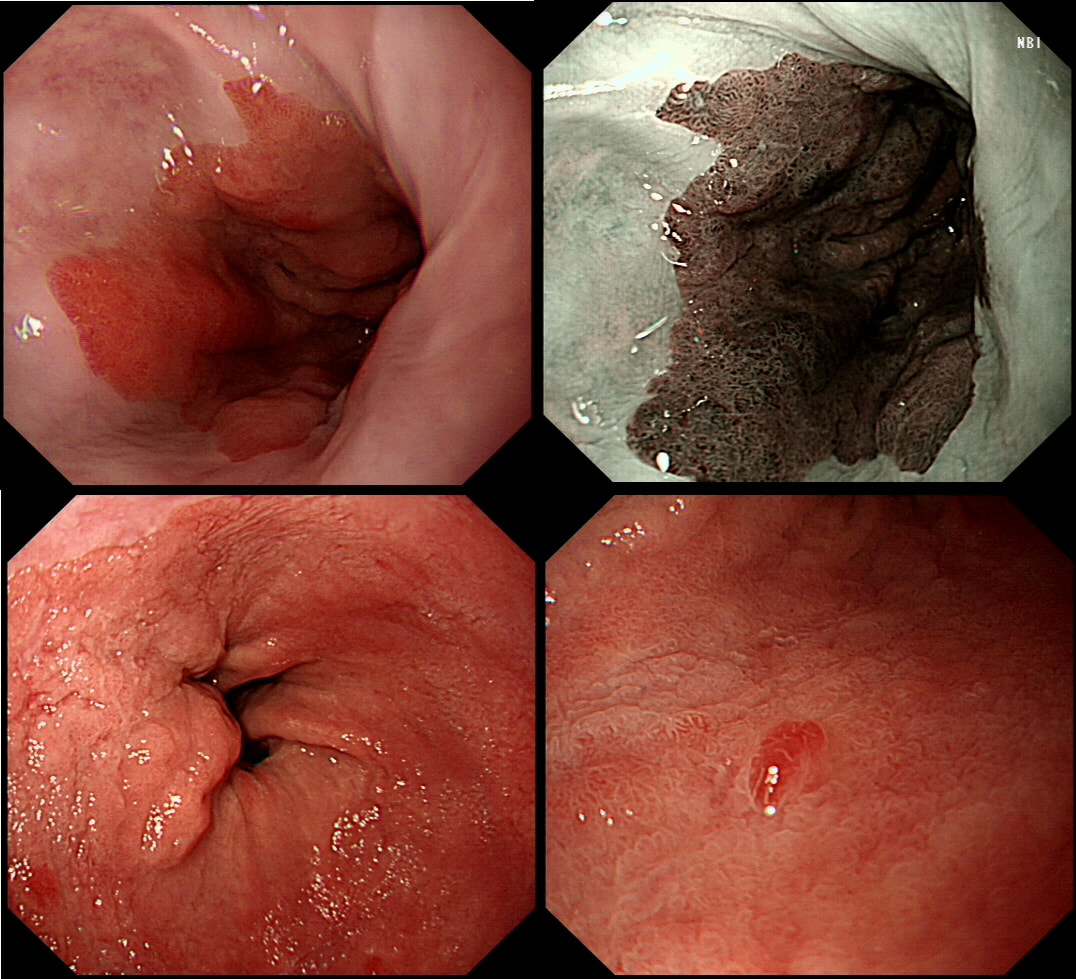
This is a C2M4 Barrett's under surveillance
WHAT IS THE LIKELY HISTOLOGY?
■ Non-neoplastic Barrett's
Nodularity within Barrett's is always bad!
■ LGD
Visible nodularity is never truly just LGD
■ HGD/IMca
A good guess but you are missing something!
■ Invasive cancer
Yes! That small ulcer is the spot of invasive cancer!
explanation
Of course you should initially scrutinise the right-hand part of the Barrett's between 12 and 5 O'clock or so. Furthermore, dysplasia is most common in the distal rather than proximal Barrett's. However, in this case the subtle nodularity is situated in the 11 O'clock position. That is why I call this an 'unusual case' of Barrett's
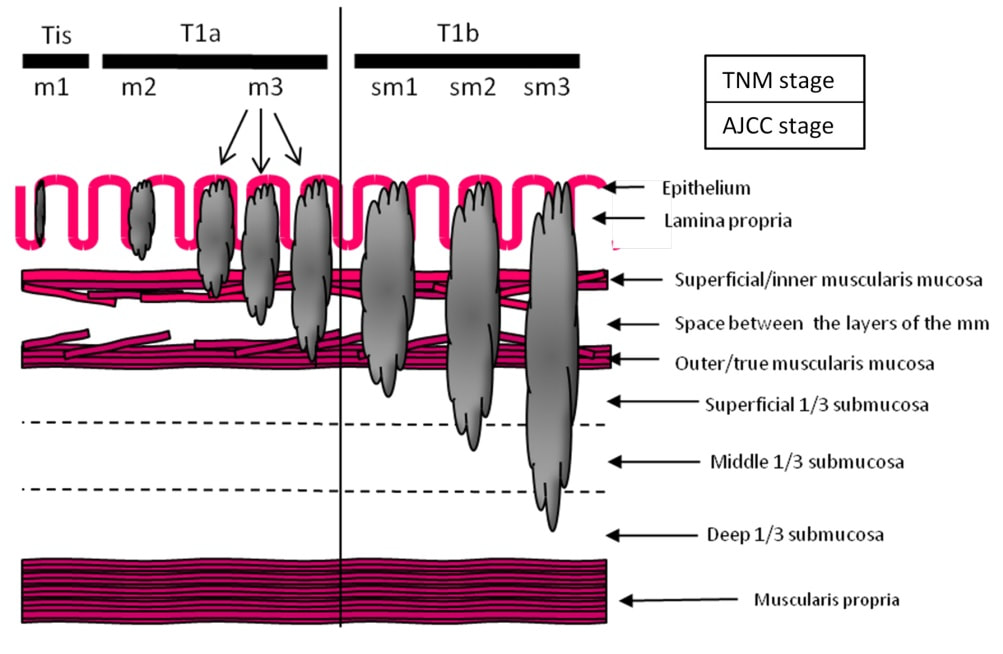
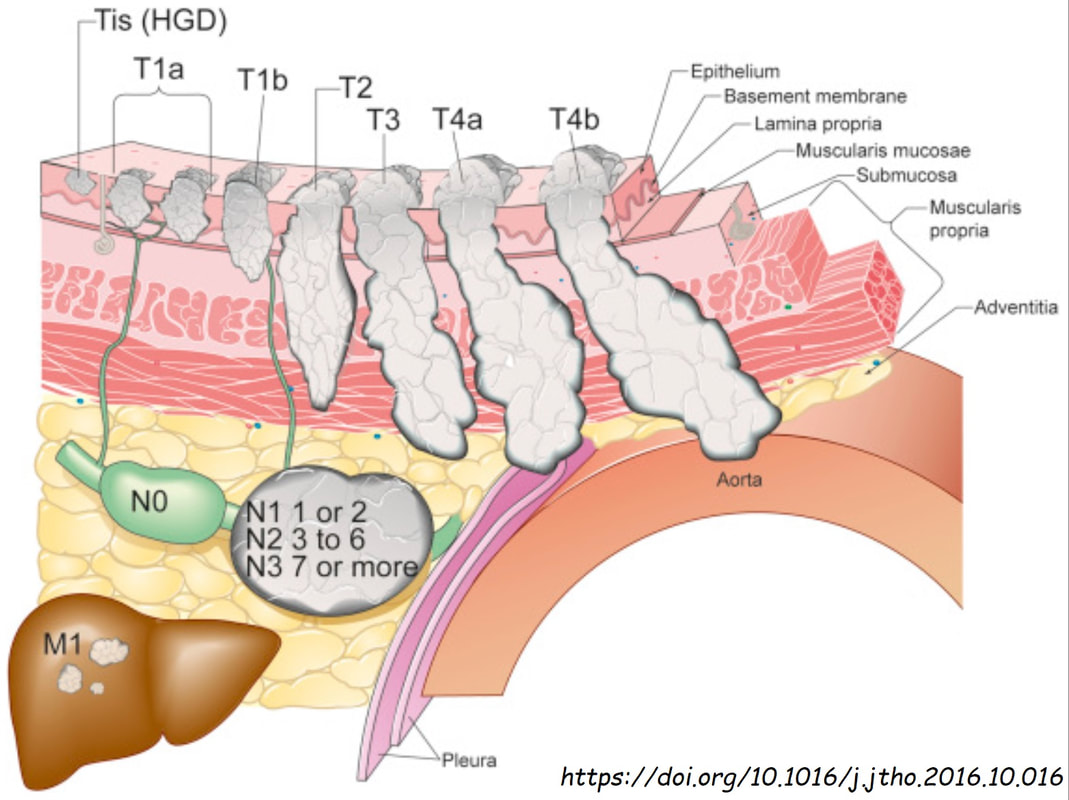
I don't think that LGD is visible endoscopically or possible to recognise even by AI systems. However, in this case there is a definite nodularity and HGD/IMca seems the most likely diagnosis. However, worryingly there is a small depression in the centre of the lesion (seen best on NBI) without any crypt pattern at all. That is the location of the poor differentiation and invasion below the muscular mucosa. Furthermore, biopsies revealed LVI and the patient subsequently had chemoradiotherapy (CRT). CRT is a great option in elderly patients but I do worry when young patients opt for CRT. This is because they are at high risk of developing further lesions and they have decades of life expectancy ahead of them for this to happen. The ultimate staging was T1b, N0 disease. I've attached a reminder about TNM staging because it's easy to forget it unless you see it every day!
A year ago, this patient underwent RFA for a C5/M6 Barrett's harbouring HGD.
WHAT IS THE MOST APPROPRIATE MANAGEMENT?
■ APC ablation
INCORRECT, the lesion is NOT FLAT!
■ RFA
INCORRECT, because the lesion is too worryingly nodular
■ EMR
CORRECT, the only way to make sure!!!
explanation
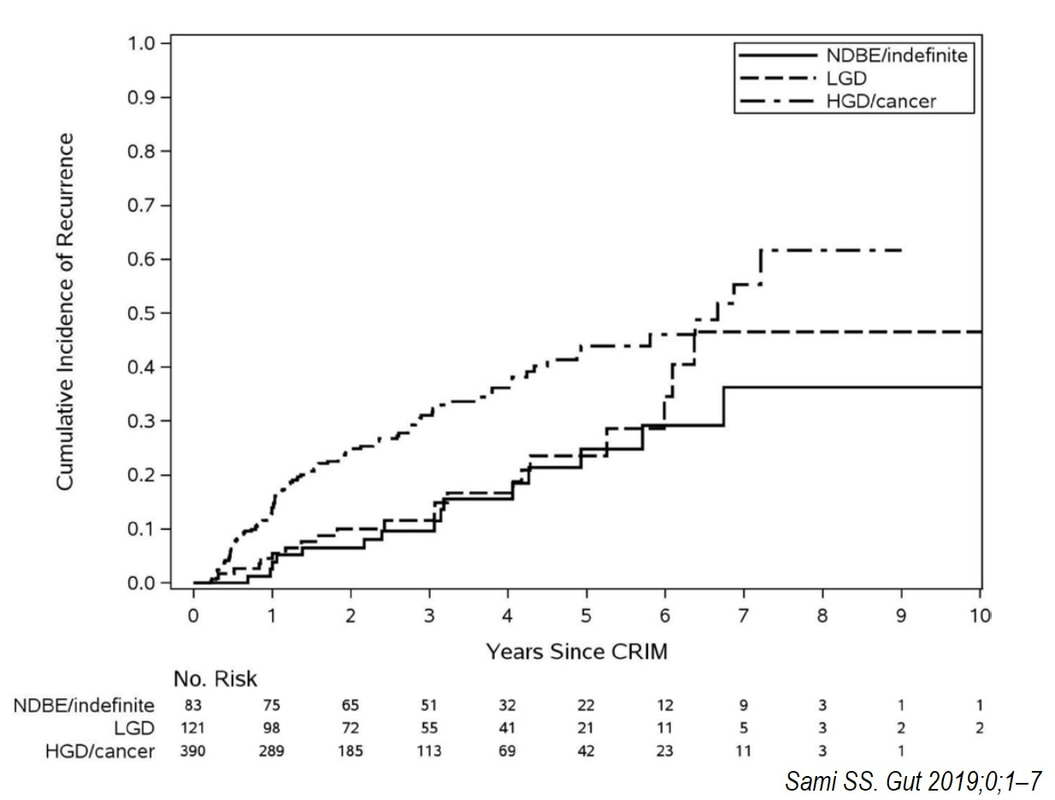
This is the problem with RFA, it doesn't last forever! After 5-6 years, about half your patients will have relapsed. Predictably, it's the patients with the most unstable changes who are the most likely to relapse (see graph below from Dr Sami's landmark paper). This is the reason why surveillance at 12 month and then 3 yearly is recommended by the BSG when the 'index histology' was LGD. However, after HGD or IMca, surveillance after 3 months, 6 months and then annually is recommended. Click here to read those recommendations in full.
Endoscopically, it can be difficult to spot a small rim of Barrett's returning, which over time slowly pushes the squamo-columnar junction proximally. In most cases, this is how Barrett's return over time. It's for this reason that we advice taking 4 biopsies from the columnar side of the SCJ'n. If your pathologists report dysplasia in any of these samples, a "top-up" RFA may well be indicated. Less commonly patients develop a nodule emerging from the 'neo-squamous mucosa' as in this case. Clearly you need to resect this for a full histological analysis. Ablation would be like sweeping it under the carpet! The nodule was removed by EMR and histology revealed an intramucosal cancer! Rather worrying that this patient's disease has in some sense, 'progressed' after RFA. Prof Rebecca Fitzgerald's group at Cambridge has shown us that the DNA is pretty thoroughly mashed up by the time the mucosa looks dysplastic down a microscope. Is it really possible for RFA to 'clean up' and remove all those chromosomal translocations ?
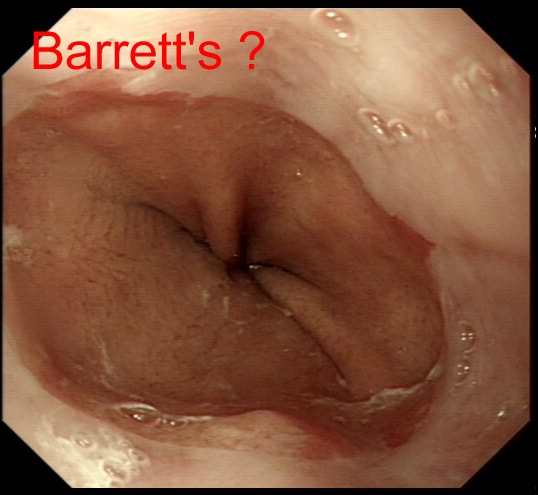
An endoscopic diagnosis of Barrett's has been made and this patient returns for further surveillance biopsies. Three years ago, no specialised columnar metaplasia was found.
DOES THIS PATIENT HAVE BARRETT'S?
■ No, I don't think so
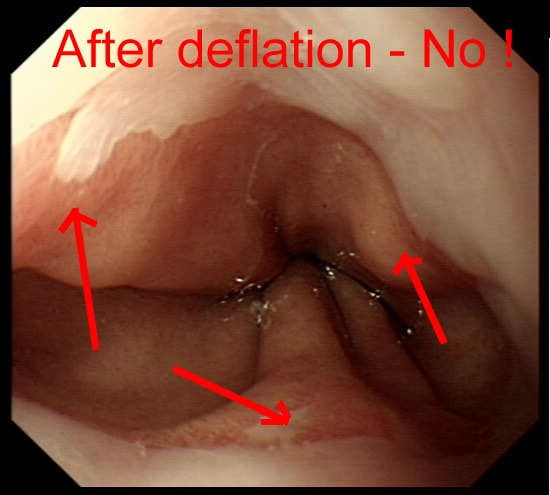
CORRECT, although it's easier to see when deflating that hiatus hernia!
■ Yes, probably!
INCORRECT, but deflate to make sure!
explanation
It can be difficult to tell if patients have Barrett's and I rather suspect that Barrett's is overdiagnosed at endoscopy. This is because you need to deflate the stomach to see the top end of those gastric folds. Below is the appearances after deflation of the stomach with the gastric folds reaching all the way up to the squamo-columnar junction. Remember, by definition, it's the top end of the gastric folds which marks the junction between the stomach and the oesophagus.
In most patients this should be where the squamo-columnar junction sits but of course, this moves proximally in Barrett's. How about taking biopsies to leave it to the pathologists to make the diagnosis? Few things upsets pathologists more than receiving some samples with the question: "- is this Barrett's?". The reason is that Barrett's is an ENDOSCOPIC diagnosis. Histologically, the normal cardia mucosa looks IDENTICAL to Barrett's and the pathologists can't tell the difference. OK! It's up to you to make that diagnosis and I've uploaded a second image below to remind you of the landmarks you should be aware of!
Lesion found at the gastro-oesophageal junction
SHOULD I ATTACK OR ABORT?
■ ATTACK!
INCORRECT. SOMETHING HORRIBLE IS LURKING BELOW!
■ ABORT
CORRECT, THERE IS A HORRIBLE LOOKING ULCERATED CANCER SEEN AT CARDIA
explanation
From above this looks like a reactive nodule, perhaps secondary to reflux. Naturally, one couldn't rely on a visual inspection and an EMR seemed indicated. However, on retrovertion (bottom right image), the true nature of the lesion is revealed. There is a depressed, indurated area just at the GOJ (the close-up image at the bottom on the left show some irregular, malignant looking vessels). I diagnosed a type III early gastric cancer (Sievert's type III guess) probably involving the muscle propria (T2 disease) and organised an EUS. Surprisingly, the subsequent EUS upstaged the lesion to T3,N0,M0). I find it tricky to "stage" lesions at the cardia. Even small lesions can be far more advanced that expected.
This pt with short stretch of Barrett's have previously had dysplasia detected within his Barrett's. He is now attending a follow up gastroscopy for consideration of RFA.
WHAT WOULD YOU DO NEXT?
■ TAKE ANOTHER SET OF SAMPLES
NOT INCORRECT☺!
■ GO AHEAD AND RFA
INCORRECT!
■ DO AN EMR FIRST
CORRECT!
EXPLANATION
On Facebook, most (69%) suggested that the nodule needed to be removed first. However, first taking another set of samples, isn't 'wrong'. Provided of course that you also sample that suspicious nodule at 6 O'clock (bottom right photograph)!
I actually removed that nodule at the 'index examination'. As expected, it was confirmed as harbouring IMca. Not a surprise as 'Nodules are NEVER allowed within Barrett's. At the same time, I also obtained a set of samples from the more unremarkable surrounding Barrett's. This revealed a mixture of high and low grade dysplasia (i.e. HGD and LGD). A few months later the patient returned for his RFA. By the way, unstable Barrett's often has a subtly irregular surface, slightly more red than usual and often with patches of superficial ulceration. In particular, superficial ulceration almost certainly means that the Barrett's is dysplastic. Presumably this is because the 'Goblet cells' are the first to disappear when the Barrett's become unstable. Of course, there is no hurry to offer patients RFA. I do take my time, first having a chat with the patient about the 'issues' of; pain afterwards (a prescription of 'oxetecaine & antacid' helps), the need for continued surveillance ('because the cure will not last') and reassure that continued 6-monthly surveillance is no less effective than RFA in reducing their likelihood of dying from Barrett's cancer. What I don't mention is that most patients on Barrett's surveillance die from cardiovascular disease. Of course their risk of dying from oesophageal cancer may well be below the average population risk. Hmm, isn't it odd that we never see this benchmark figure of 'average population risk' in publications? Arguably, it's the ONLY benchmark that matters !!!
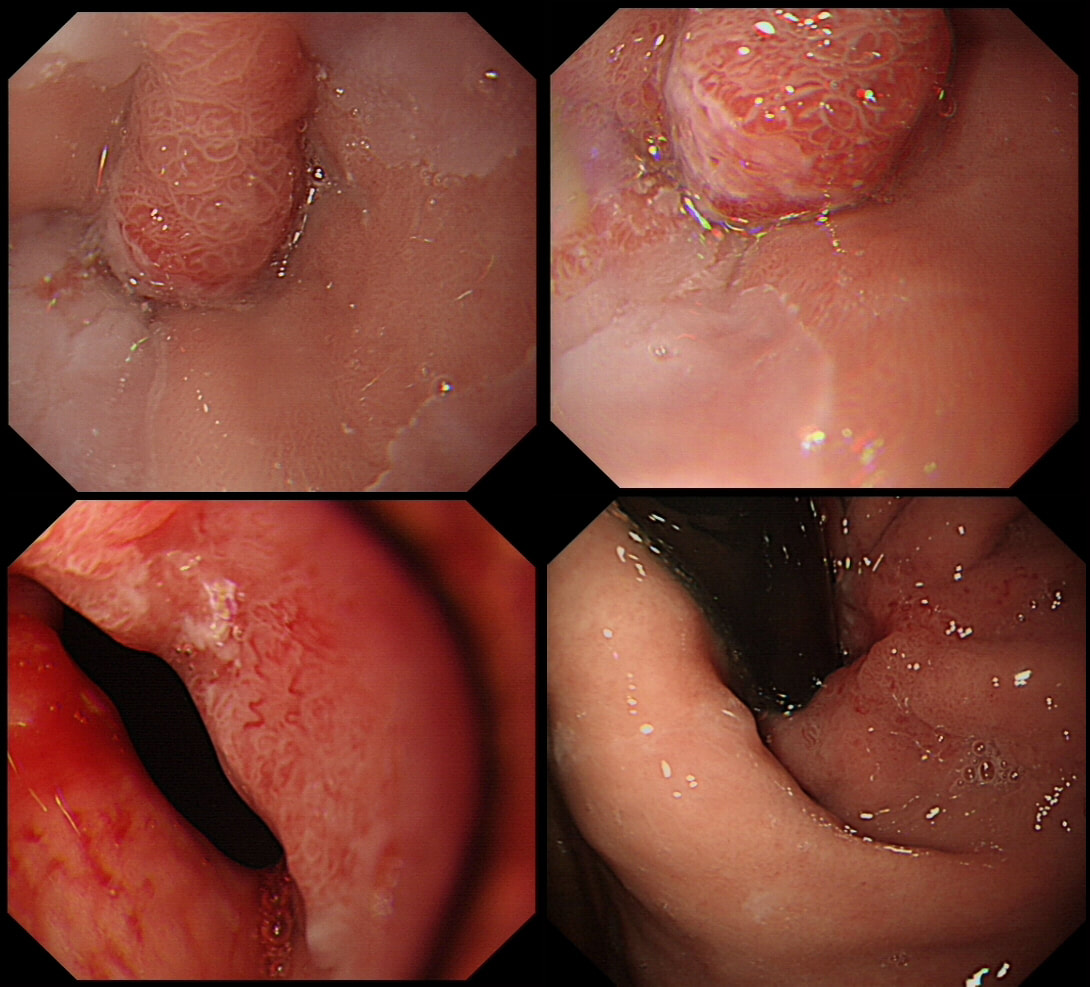
Here are two cases of a small nodule within Barrett's (top and bottom row). Of course you know that a nodule within Barrett's should ALWAYS be viewed with suspicioun. Naturally, both nodules were sampled as well as the surrounding unremarkable looking Barrett's. One case turned out to be non-dysplastic but the other were reported to harbour IMca.
WHICH NODULE IS DYSPLASTIC?
Case A (top row)
INCORRECT!
Case B (bottom row)
CORRECT!
explanation
On facebook, this proved a very difficult question and only half correctly identified Case B (bottom row) as being dysplastic.
Must admit that I felt uncomfortable about both nodules, as in my book, a nodule within Barrett's is never "right". In the end, I actually removed both. Just to be on the safe side. Incidentally, my own simple 'rule of thumb' when assessing Barrett's is as follows:
|
Categories
All
|