|
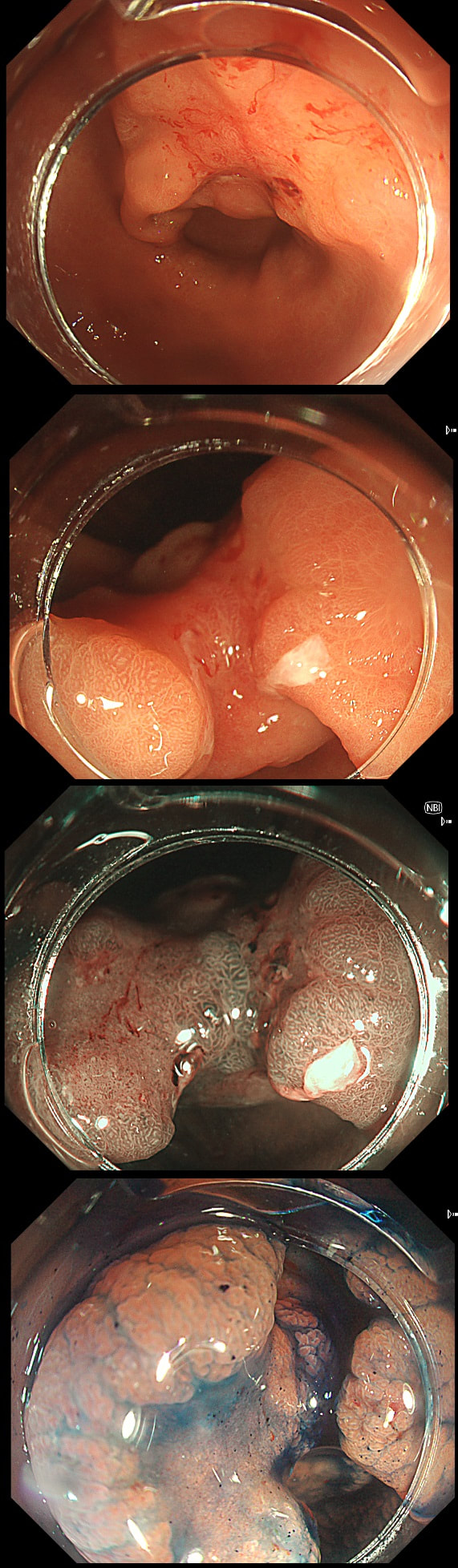
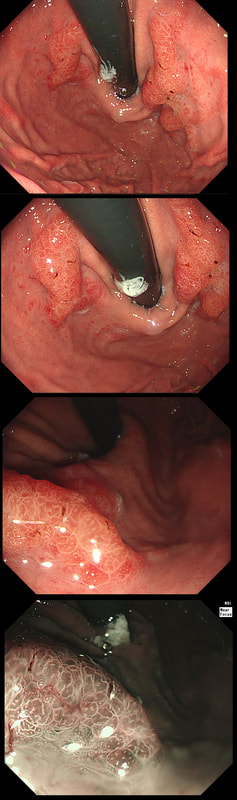
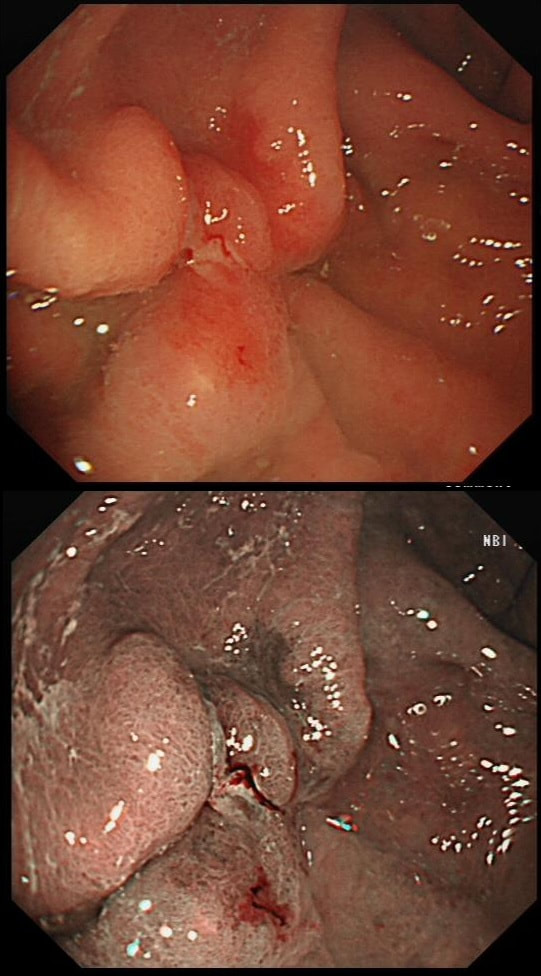
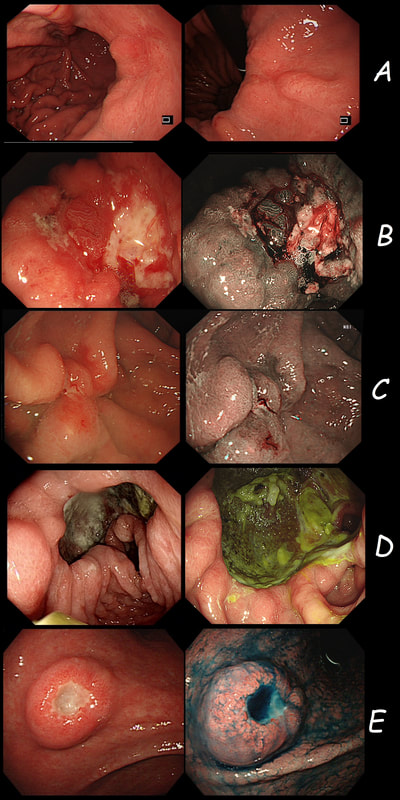
This gastric lesion is found in an 85 yr old man. Samples have confirmed a moderately differentiated gastric cancer but the lesion can't be seen on CT. The patient is frail with multiple comorbidities and declines both an endoscopic resection and surgery. However, he does ask how long it will take before he dies from the cancer?
WHAT WILL YOU ANSWER?
a) 1-2 yrs
Definitely too pessimistic!
b) 2-3 yrs
Hmm, perhaps if the lesion is poorly differentiated but it doesn't look it!
c) 3-4 yrs
This is probably how long it will before that cancer is at an advanced stage but patient will probably survive for another couple of years beyond that 'transition point'
d) 4-5 yrs
Probably too pessimistic still!
e) >5 yrs
With limited information this is the only figure backed up by some data.
Explanation
This question is something of an homage to Sir David Cox who developed the well known 'Cox Regression analysis'. Professor Cox died in 2022. As you know, the 'Cox Regression analysis' basically explores the relationship between the length of survival and more than one variable.
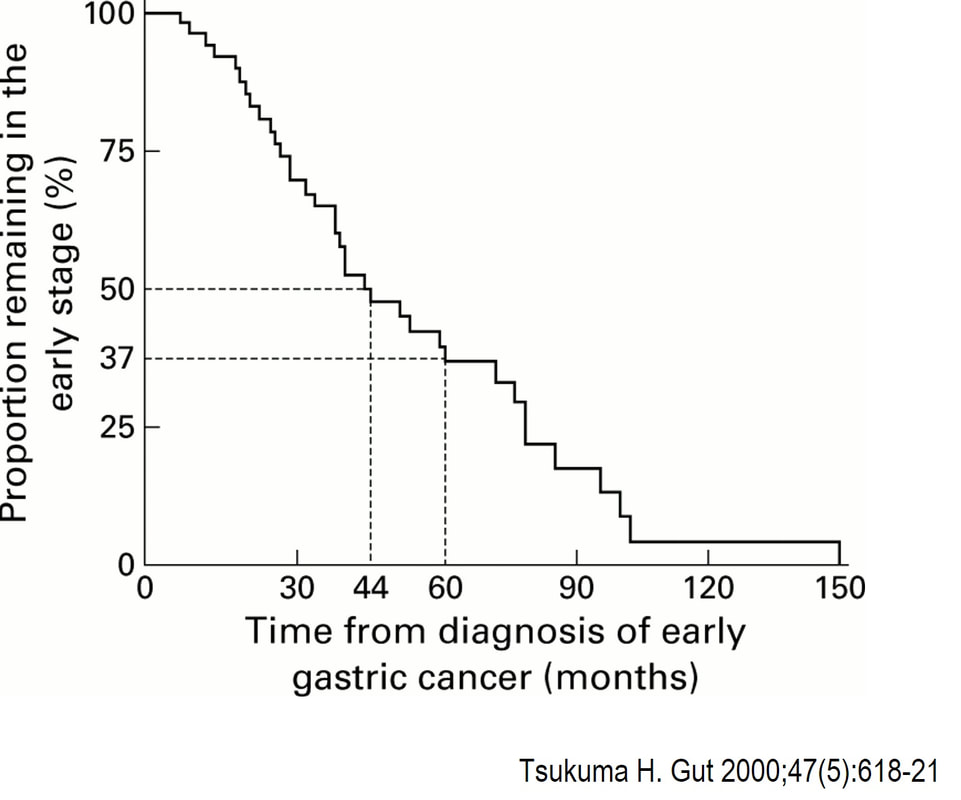
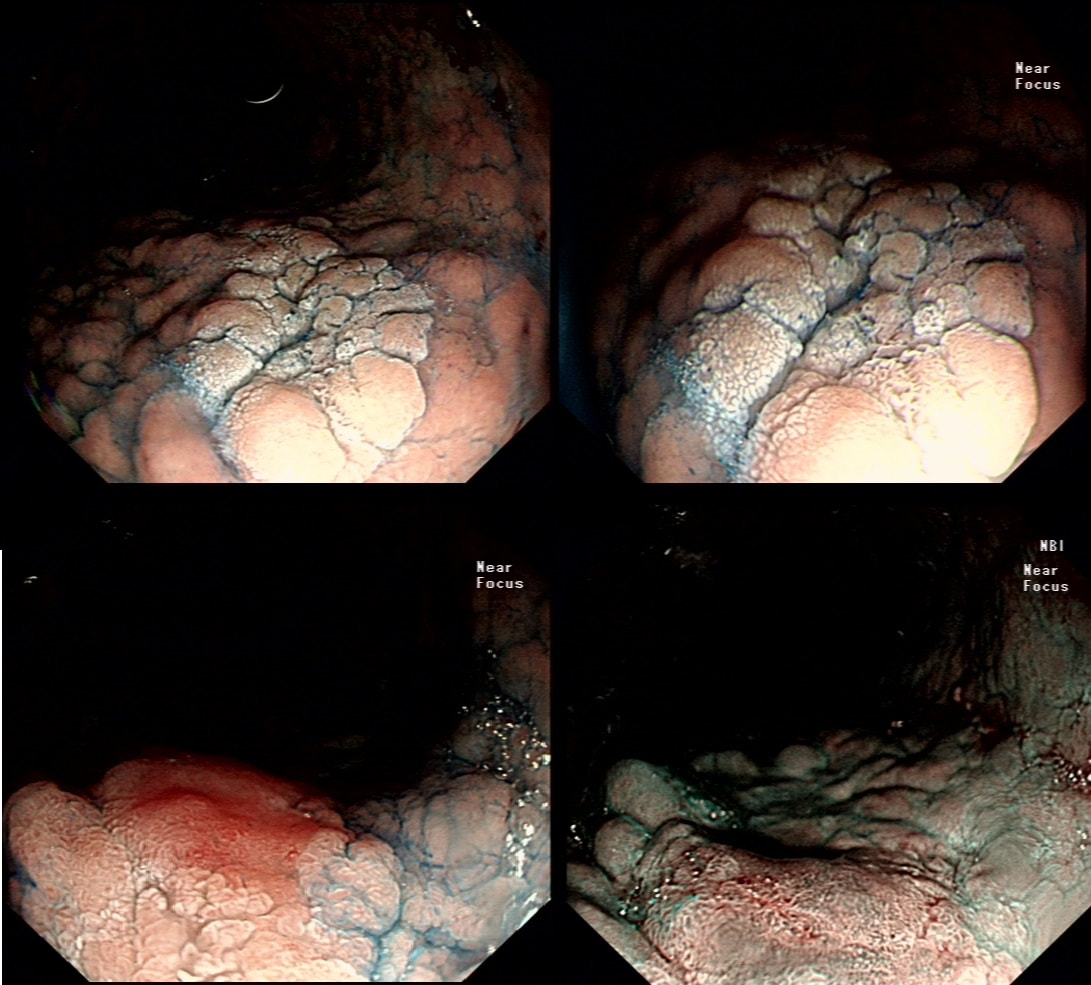
You would expect the following 'variables' to influence cancer-specific survival; histology, size of lesion, stage of lesion and perhaps the macroscopical type of early gastric cancer. Perhaps the location within the stomach also influences survival. There is some data that lesions at the gastric cardia are worse. Of course we don't have data on any of these 'variables'. However, I know of one paper by Tsukuma et al [Gut 2000;47(5):618-21] which did look at the natural history of 56 early gastric cancers in Osaka, Japan. This paper was actually a riposte to an earlier editorial from Leeds by one of my colleagues arguying that EGC is a 'pseudo-disease [Everett S. Lancet 1998;351:1350–2]. As you would expect, 56 EGC's is not enough for any sub-analysis of the above variables. Also they didn't actually look at survival. They looked at the average time it took lesion to progress from what appeared to be 'early' gastric cancer (i.e. confined to the mucosa or submucosa) as diagnosed by endoscopy. And here we reach the first issue! How sure can we be that the lesion in the image is an EGC and hasn't started to invade into the muscle propria (thus being advanced)? The lesion is about 2cm across and there is a an organised crypt pattern in the centre (can't comment on the vessel pattern as there are no magnification images). It does look 'early' and I stand at least an 80% chance of being correct in that diagnosis. Incidentally, 'lifting' is not a reliable sign in the stomach as any ulceration of the mucosa will tether it down, regardless of stage of invasion. The authors were pleased to report that if patients are left for long enough, those EGC's will grow and progress. No surprise there then! But the rate of progression was slow!!! The median time it took for 'early' gastric cancers to progress to advanced gastric cancers (i.e. T2 or worse) was estimated to be 44 months but probably shorter in patients with poorly differentiated cancers. Furthermore, the cumulative 5 year corrected survival in patients who never underwent surgery was about 63%. The answer to the question is therefore 'e'. This patient is likely to live more than 5 years from diagnosis of his early gastric cancer. Better than expected here is the Kaplan-Meyer curve below !
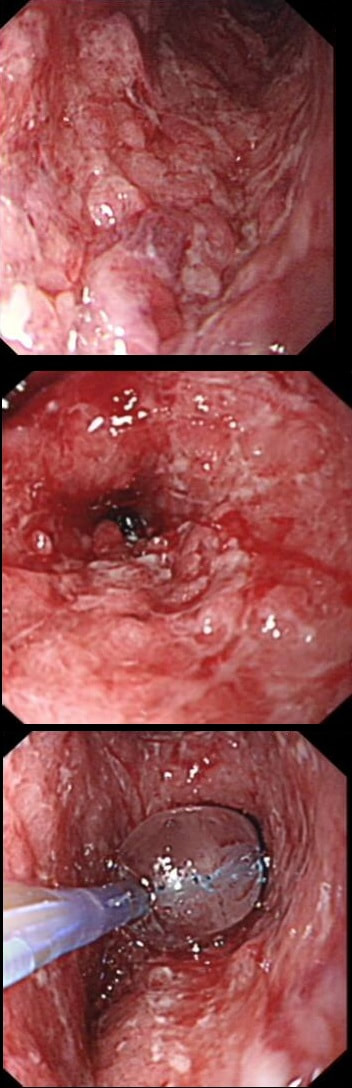
This is the stomach of a middle aged lady undergoing gastroscopy because of anaemia.
WHAT IS THE DIAGNOSIS?
a) HP associated gastritis
This is beyond gastritis! The fundal mucosa is pale and atrophic rather than red and inflammed!
b) Atrophic gastritis
The fundal mucosa does appear atrophic apart from a few scattered red patches which is all that remains of the normal gastric lining. But how about the antrum?
c) Autoimmune gastritis
Correct!
D) Coeliac disease
That duodenal mucosa is unremarkable. The 'polyp' in the 3 o'clock position is obviously the duodenal papilla.
Explanation
Those red spots in the gastric fundus is what remains of the more normal gastric mucosa whilst most of the surrounding mucosa is atrophic. In contrast, the antrum is unremarkable, as is the duodenum. This is an example of a 'body predominant' gastritis and your endoscopic diagnosis should be an autoimmune gastritis! The antral G-cells, found deep within the antral pyloric glands are pumping out huge amounts of gastric which is leading to ECL cell hyperplasia and multiple small NET's. In fact, most patients with type I gastric carcinoids have an autoimmune gastritis! The anaemia was due to vitamin B12 deficiency.
WHAT IS THE MOST LIKELY DIAGNOSIS?
a) Portal hypertensive gastropathy
Doesn't really look like 'snake skin' does it?
b) HP associated gastritis
HPAG is a great mimicker but not this time
c) Gastric vascular ectasia
Absolutely, affecting the whole stomach!
d) Lymphocytic gastritis
LyC gastritis is ulcerative!
e) Linitis plastica
Another great mimicker but this is a vascular issue rather than mucosal infiltration!
explanation
Of course, this is a case of Gastric Vascular Ectasia. As the whole of the stomach is affected, I don't think that we can call it 'antral' ectasia. Gastric antral vascular ectasia “GAVE” (watermelon stomach) has been described with; • Cirrhosis • Atrophic gastritis/achlorhydria/hypergastrinaemia • Connective tissue diseases (especially systemic sclerosis and hypothyroidism) • Diabetes • Heart failure • Chronic renal failure It has been proposed that the underlying link between all the different conditions associated with GAVE may have autonomic dysfunction in common leading to prolapse of the antral mucosa through the pylorus. Must admit that I'm sceptical! A study suggested that a “punctuate pattern”, as seen in this clip, is typical of cirrhosis whilst a “striped type” was more common in non-cirrhotic cases. Indeed this patient had NASH cirrhosis. Portal hypertensive gastropathy probably has a different aetiology and is a different condition as gastric antral vascular ectasia can develop with a normal portal pressure. Patients with vascular ectasia of course usually present with iron deficiency anaemia. Patients with bleeding from GAVE were previously treated with antrectomy. Now, the first line treatment is “thermal therapy”. Octeotride and oestrogen-progesterone have also been used in small series but would now only be considered if endoscopic therapy fails. It is far easier to treat these lesions using the argon coagulator than the heater probe. Using a banding device may be quicker in patients who tolerate gastroscopies poorly. RFA is also effective but is VERY expensive. This is a solitary gastric polyp WHAT IS YOUR ENDOSCOPIC DIAGNOSIS?
a) Hyperplastic polyp
Spot on!
b) Hamartomatous polyp
A bold guess but wrong!
c) Cystic fundic polyp
Too red and should be several of them!
d) Adenomatous polyp
But there is no defined crypt pattern?!
e) Malignant polyp
May be a 5% chance that you are right!
explanation
A missing piece of information, which I perhaps should have provided, was the H.pylori status of this patient! This lesion was arising from a H.pylori associated gastritis.
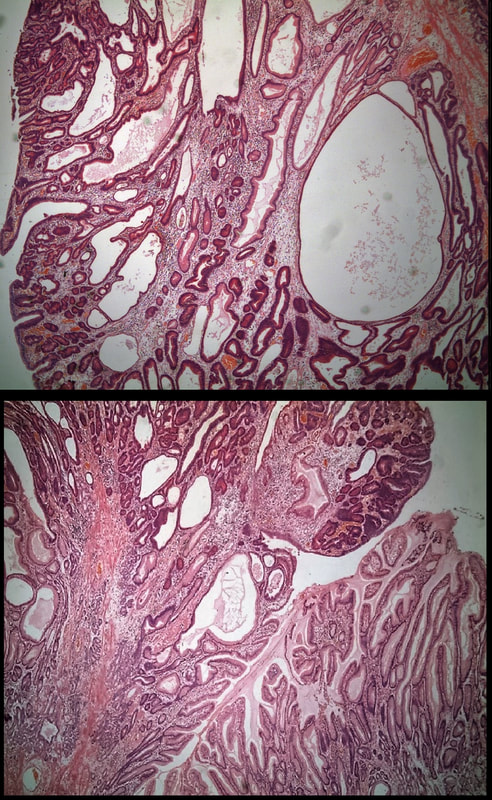
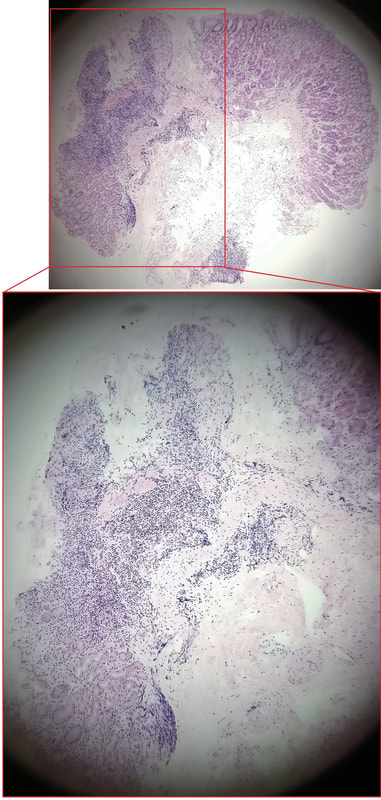
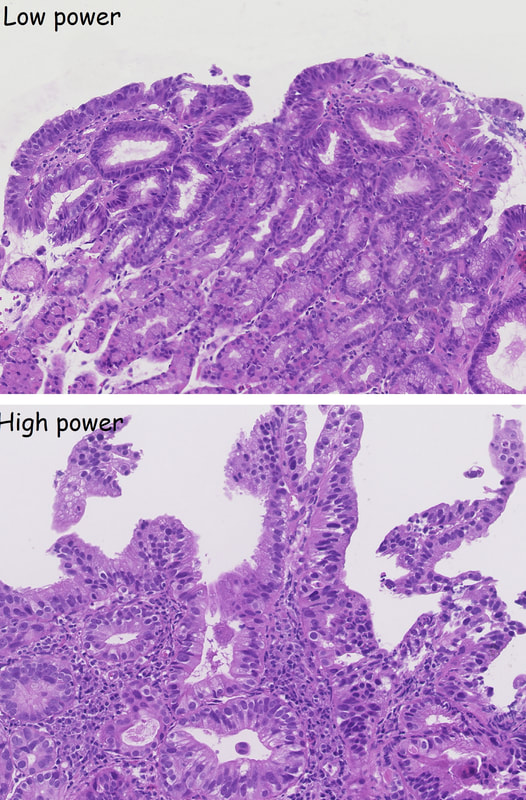
Most polyps found in a stomach with Helicobacter pylori associated gastritis are hyperplastic (inflammatory). They appear angrily red (because they have lots of capillaries) and often with white fibrin caps, making them look a little like mushrooms. A small proportion of hyperplastic looking polyps are actually malignant. In my experience, these 'stealth cancers' are most common close to the gastric cardia and are always solitary. Conversely, multiple inflammatory polyps in the antrum and gastric body are almost certainly benign. Of course, this is a 'solitary' hyperplastic polyp, close to the fundus. Perhaps not unexpectedly it harboured some (low grade) dysplasia (histology below). The dysplasia is in the crypts lined by deep purple/blue cells rather than the normal light pink cells. The contrast is probably best seen in the second histological slide where the dysplastic part is towards the top of the slide and the non-dysplastic is at the bottom half. If the H.pylori test is negative, and particularly if the patient is taking a PPI, the polyp is more likely to be a 'Fundic gland polyp' (cystic fundic polyp) which are full of cystic spaces and therefore look a little translucent like frog-spawn. Rarely polyps are hamartomatous as in Peutz-Jeghers polyps and the polyps arising in patients with 'Cronkhite-Canada syndrome or in Juvenile polyposis. In many cases these lesions have an odd but distinct surface crypt pattern or, alternatively look again look translucent like frogspawn. It's rather difficult to explain the appearance of a hamartomatous polyp and for this reason, I have attached some images below (after the histology slides). Another polyp, most likely to arise in an atrophic gastritis with patches of intestinal metaplasia, is an adenomatous polyp of the 'intestinal type'. To remind you, there are at least 4 types of gastric adenomas which all have an organised and regular surface crypt pattern:
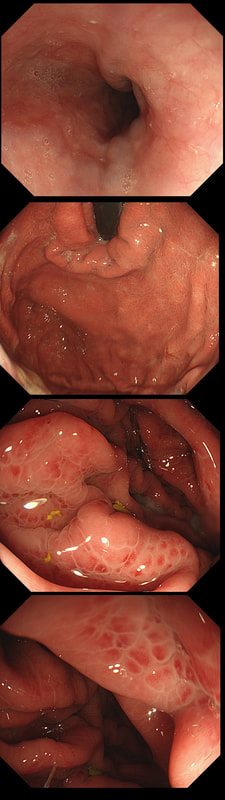
A 21-year-old man presents with a past history of intermittent abdominal pains. For the last year he has suffered with more frequently bouts of abdominal pains and more recently he has started to vomit some 30 minutes after eating. He has been started on lansoprazole for a couple of months with no response. At gastroscopy (photograph) there is a severe gastritis and a tight pylorus requiring dilatation to examine the duodenum (which was unremarkable).
His blood results are as follows: Hb 11.2 g/l MCV 105 fl WCC 11.9 x 109 Plat 395 x 109 Basal gastrin 180 pg/ml (<75) WHAT IS THE MOST LIKELY DIAGNOSIS?
a) Hp associated gastritis
Doesn't explain all the 'issues' !
b) Zollinger-Ellison syndrome
There is a simple explanation to that elevated gastrin level!
c) Crohn's disease
Well done!
d) Gastric lymphoma
The antrum would do but how do you explain the macrocytosis?
e) Diffuse type gastric cancer
Could look like this but he is a bit young isn't he? !
explanation
Although the patient has a raised gastric level, this can be explained by the lanzoprazole. The macrocytosis is more difficult to explain. An autoimmune gastritis would be expected to be worse in the gastric body than in the antrum. In fact, the only other possibility is that the patient has terminal ileal Crohn's disease, causing malabsorption of B12 as well as a Crohn's gastritis. This was indeed the case! Actually, the diagnosis had already been confirmed by antral biopsies before I carried out the dilatation! Wouldn't like to dilate a diffuse type cancer !
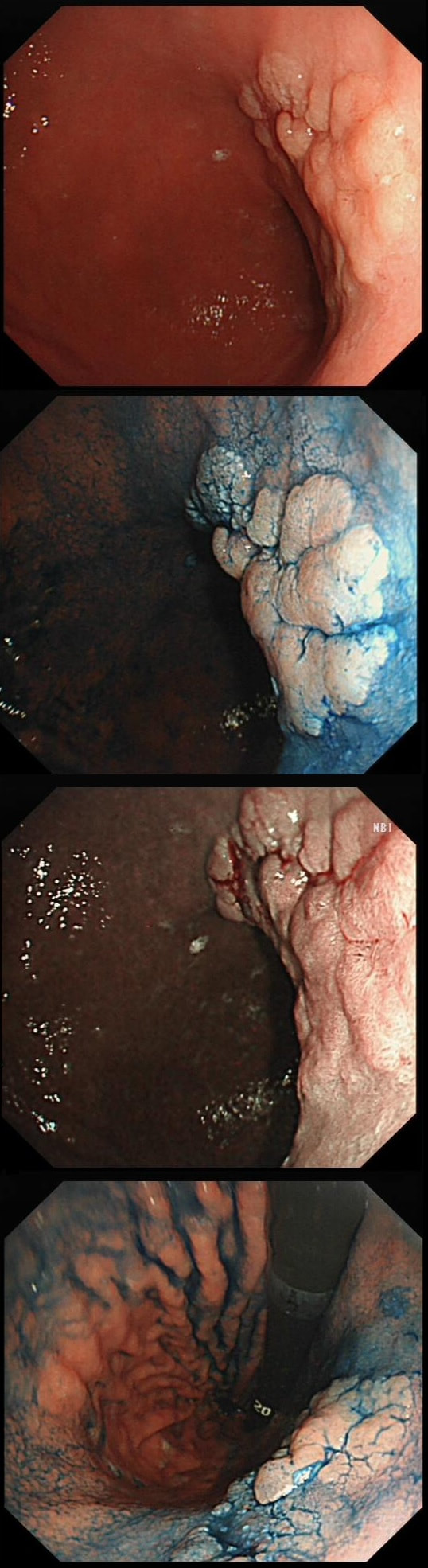
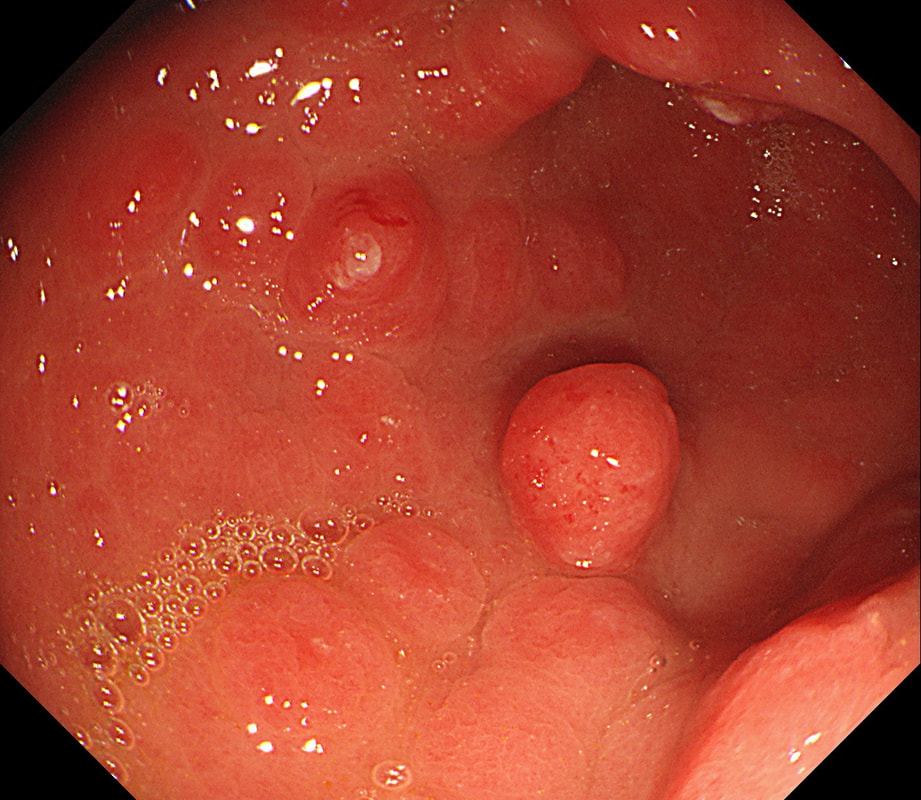
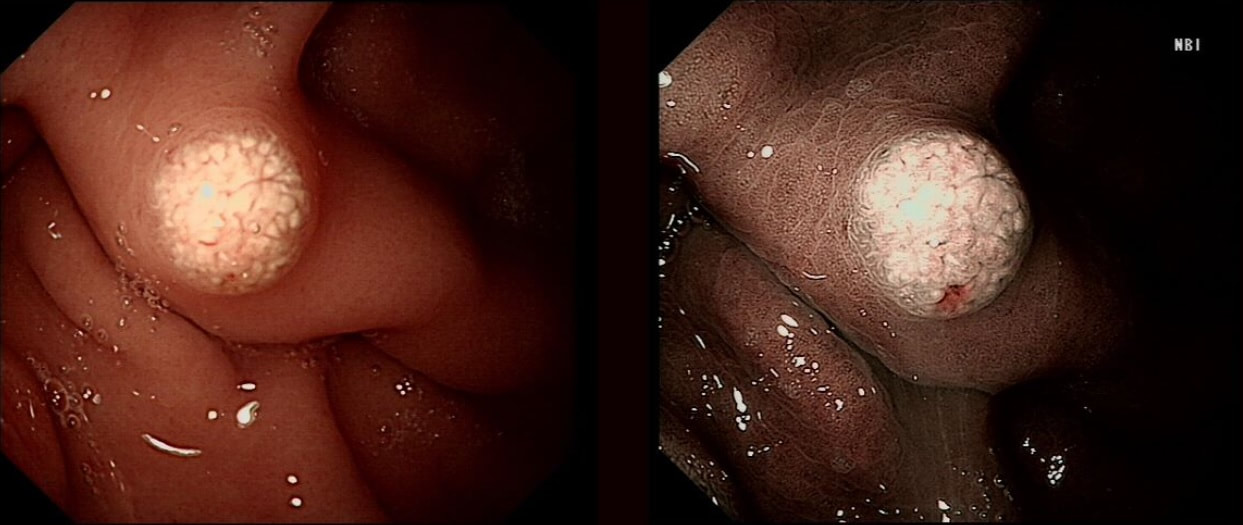
This polyp found on the lesser gastric curve.
WHAT IS THE MOST LIKELY DIAGNOSIS?
a) Hyperplastic polyp
HP polyps are usually subpedunculated and angry red
b) Intestinal metaplasia
IM is also 'pale' but not 'flat elevated'
c) Gastric adenoma
Would be my first guess!
d) Early invasive cancer
This is a plaque without a depression or dominant nodule to suggest invasive cancer
explanation
Of course, this could have been rather easy to miss! An insignificant pale, plaque-like lesion but with distinct borders best seen after indigo carmine dye spray (as usual). Endoscopically this is what a gastric adenoma looks like and it was removed endoscopically. Somewhat surprisingly, histology reported a small spot of IMca and therefore its at actually an early cancer - not invasive though!
Most invasive, gastric EGC's are of course shallow depressions as in the example below. In fact the lesion below was invading into the middle of submucosa and was confirmed as poorly differentiated. Afterwards the patient underwent a distal gastrectomy where no further cancer was found. 'Poor differentiation' is the least reliable and lymphovascular invasion (LVI) is the most reliable predictor of nearby lymphnode micro-metastases.
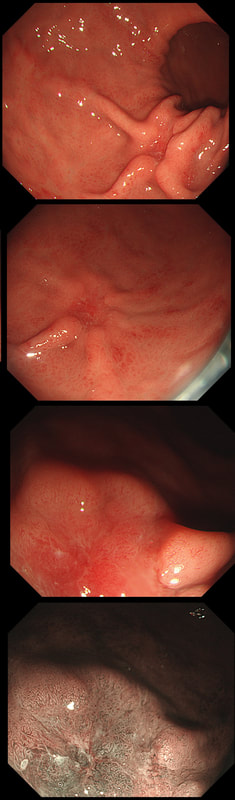
This was found on the anterior gastric wall in an elderly patient with iron deficiency anaemia
WHAT IS THE MOST LIKELY DIAGNOSIS?
■ Gastric ulcer scar
Quite possible but you would take samples surely?
■ Gastric erosion
But there is a nodule in the centre?
■ Benign gastric ulcer
But there is no 'ulcer' in the centre?!
■ Early gastric cancer
Surely, the most likely diagnosis?
explanation
Gastric folds are being pulled into this 'lesion'. Of course most EGC's are depressed or flat elevated with a central depression. This is not easy to classify into the Paris classification system. I guess that you can argue that its' an elevated lesion with a central depression? Anyway, at the centre of the lesion there is a nodule! Not an ulcer! It's most likely an EGC. In fact, the lesion was resected endoscopically and proved to be poorly differentiated invasive cancer with a positive deep margin ! As the patient was 84 yrs old, no surgery was offered. That was 6 years ago and the patient remains well with only an unremarkable scar in the stomach to remind him ! Wonderful !
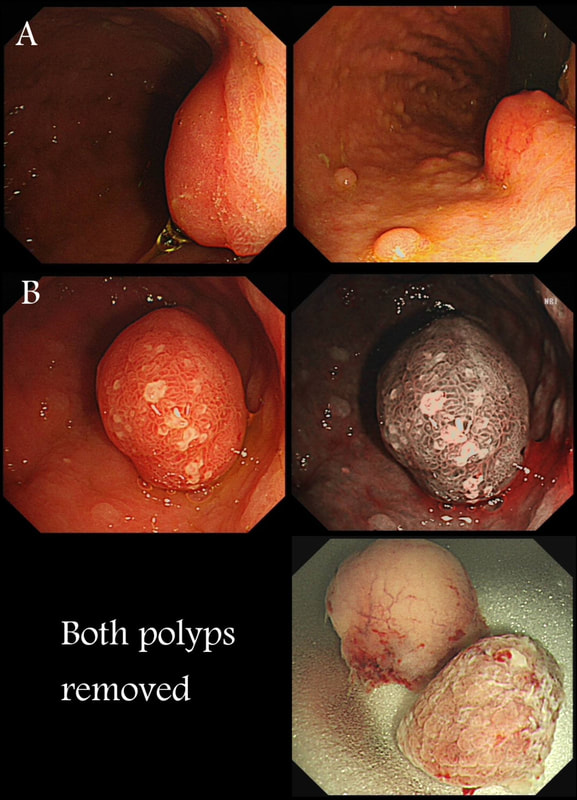
Two gastric polyps (labelled A & B) were both removed from the stomach of a 60 year old woman who complained of indigestion.
WHAT IS THE LIKELY DIAGNOSIS?
■ Both are hyperplastic
But the look different to each other!
■ NET + HP polyp
Well done ! And presumably you know which is which?
■ Both are NET's
But they look different!
■ HP + Adenoma
How about those fine vessels seen in the second image?
■ Both are adenomatous
But they look different!
explanation
You can see the thin vessels crawling up the side of polyp A. This appearance is typical of a gastric NET. Polyp B has a more villous surface with a few white spots - all typical of a hyperplastic gastric polyp. Some of these may contain some malignant cells which somehow generate a hyperplastic/reactive/inflammatory reaction around themselves. However, most hyperplastic gastric polyps are benign and arise secondary to a Helicobacter pylori gastritis. For this reason, you should always do a CLO test in these cases.
Every few weeks, I look up the notes on Prof Pritchard's' Podcast on gastric NET's to remind myself of the workup of these cases. As you remember, you should take samples to confirm an atrophic gastritis whenever you find a gastric NET. In this case, the patient did NOT have an atrophic gastritis. Instead, there was a Hp associated gastritis which is the reason for the second polyp. We realised that what we had was a TYPE II gastric NET! Analysis confirmed that the 17mm was WHO grade II. The finding of anything else than an innocent Type I gastric NET means that further imaging was required. A little late in the day of course but fortunately, the following imaging investigations were unremarkable:
Anyway, below is a reminder of what to do at gastroscopy, when you have a case of gastric NET:
This is an elderly patient undergoing an OGD because of IDA (iron deficiency anaemia). A lesion catches my eye on the anterior gastric wall WHAT IS THE LIKELY HISTOLOGY?
■ Gastric xanthelasma
The colour is suggestive and gastric atrophy would be the link but it's not quite right...
■ Healed GU scar
But how about that funny crypt pattern?
■ Gastric adenoma
Yes, doesn't this look like a tubular adenoma?
■ Early gastric cancer
Wouldn't the crypt pattern then be very disorganised or perhaps very small?
explanation
The pale colour is odd and reminiscent of gastric xanthelasma which as you know is linked with gastric atrophy which is the likely cause of this patients IDA. However, when I zoom in on the area, the crypt pattern is different here. Of course, this does not fit with a xanthelasma or a scar from a healed gastric ulcer (GU) either for that matter. Interestingly, almost everyone thought that this was an EGC. However, THERE IS a distinct crypt pattern in the centre of the lesion. Furthermore, the lesion isn't red. Remember that cancers encourage the growth of small irregular capillaries which gives them a red colouration. Finally, it doesn't have the typical flat-elevated with a central depression (IIa+IIc) growth morphology. Therefore, your first guess should be a gastric adenoma! This is actually a gastric tubular adenoma which we found in an elderly frail patient with atrophic gastritis some 10 years ago. As she had some comorbidities and it was only harbouring LGD, we decided to keep an eye on the lesion on a yearly basis. The risk of progression is supposedly only 5% with tubular adenomas in the stomach. In contrast, villous adenomas are much more likely to progress (40%). The BSG gastric polyp guidelines have the references if you want to look this up. Of course, the issue is not entirely clear-cut as risk of progression also increases with the size of the lesion (and this is probably 2cm in size) and also with age (patient is now 86 yrs). In some ways, making an initial decision to either 'attack' or 'abort' would be easiest. After all, regular surveillance drains valuable resources and leaves you open to the possibility that at some point in the future, the patient is no longer a candidate for anything more invasive than a haircut but now the lesion under surveillance shows evidence of progression. Then your patient could well ask the legitimate question why you didn't go ahead when he was younger and fit enough but instead wasted his time with pointless surveillance?! My own preferred way to navigate this minefield is to openly discuss the three options with the patient;
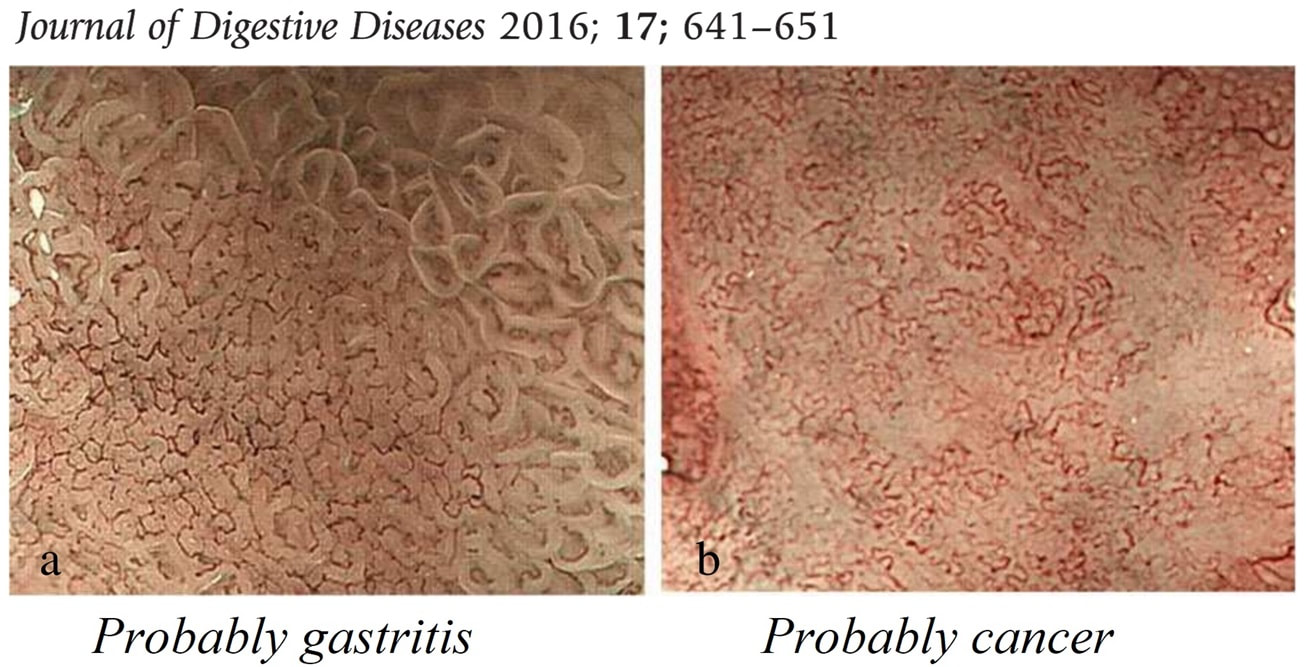
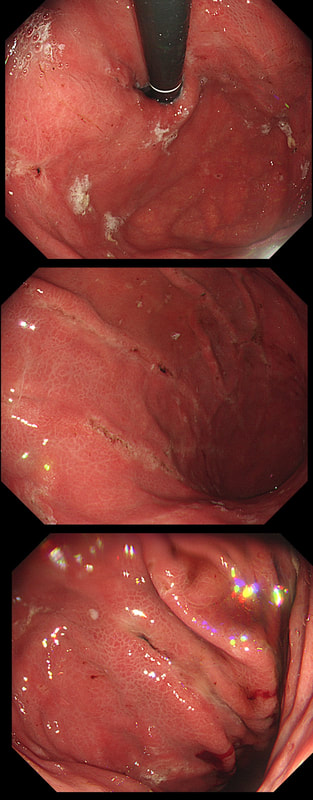
I often wonder if its the personality type which dictates what patients prefer. Perhaps, those who think 'my glass is half empty' usually want to have their lesion resected immediately whilst people who regards their 'glass to be half full', prefer to hope for the best and get on with their lives? This patient is under surveillance after having had two previous EGC's. The stomach has been sprayed with indigo carmine dye when this lesion caught my eyes. WHAT IS THE LIKELY DIAGNOSIS?
■ Likely to be gastritis only
The non-staining is suspicious and rarely lie...
■ Likely to be a patch of intestinal metaplasia
There appears to be a crypt pattern in the centre but it can still be neoplastic!
■ Likely to be neoplastic
Non-staining, slightly raised and in an 'unstable stomach' - surely!
explanation
Clearly this is an unstable stomach having given rise to two previous EGC's. In patients attending for surveillance, I always spray indigo-carmine dye throughout the stomach using a spray catheter. I then look carefully at any spots where the dye seems to have slid off. This is such a 'lesion' which was confirmed as a subtle EGC. After my samples had confirmed my suspicion it was removed by ESD and was confirmed as HGD/IMca. I consider the both as the same histology as one pathologists HGD is another pathologists IMca. There was no LVI. However, I was surprised to find that one mucosal resection edge was involved by cancer. Looking back at the clip, I think that it's the 4 O'clock border which has an irregular extension which I failed to spot.
Most EGC's look like this, a shallow depression with a subtle elevated margin. I find the crypt pattern more difficult to interpret. However, as you know, the more destroyed it is, the more likely the lesion is to be poorly differentiated.
■ Inflammed granulation tissue
Was my first too! But it doesn't quite look right ...
■ Adenomatous polyp
Adenomatous polyps are usually smooth topped
■ Malignant polyp
An intramucosal cancer arising from the anastomosis
explanation
This patient had in the past undergone a Billroth II operation. You can see that the nodule is arising from the surgical anastomosis. Distal gastrectomy is a well-known risk factor for developing an anastomotic cancer later [Sitarz R. World J Gastro. 2012;18(25):3201–6]. The risk of anastomotic cancer steadily increases after surgery. About 15 years after surgery, the risk exceeds that of the background population (age- and sex-matched). For this reason, surveillance has been suggested to start 15 yrs after surgery. The case for surveillance was strengthened by the fact that dysplasia can often be found in random biopsies from the anastomosis several years before cancer develops. In spite of this surveillance in not recommended. A surveillance study in Amsterdam traced 500 patients who had undergone a distal gastrectomy for benign disease and only detected 10 cancers (6 were in an early stage). Furthermore, there was no survival advantage in the screened group after 10 years follow up [J Clin Pathol. 1984;37:748–54]. Other studies have also put the cancer yield by surveillance at around 2% and concluded that regular surveillance could not be recommended [Am J Surg 1977;134:581-4], [ Lancet 1977;ii467-9] [ Scan J Gastro 1981;suppl 16:169-71]. Nevertheless, my practise is to always take do 'opportunistic screening' by obtaining 6 biopsies or so from the gastric side of the anastomosis when I come across a case. On first glance the nodule appears to be inflamed granulation tissue only. However, there is an odd cleft in the centre and the base from which it arises is also nodular. The polyp was removed and was confirmed as harbouring intramucosal cancer. Of course, after finding dysplasia (which often does not progress) or IMca, these patients should be offered surveillance.
This was found at gastroscopy in a middle aged patient with anaemia
WHICH STATEMENT IS CORRECT?
■ The severity is related to the severity of the underlying disease
This is generally thought to be the case!
■ Its generally more marked in the distal vs the proximal stomach
It's the other way around actually
■ Beta blockers don't help
Beta blockers do reduce gastric perfusion
■ The condition is irreversible
A reduction in portal pressure improves the condition
explanation
Congestion secondary to portal hypertension is thought to be the primary cause of Portal Hypertensive Gastropathy. However, other factors may also play a part such as mucosal protective mechanisms, inflammatory response, local vascular tone, hepatic function, gastric mucosal perfusion, endotoxin, and gastric sucrose permeability, have been suggested to influence the development of Portal Hypertensive Gastropathy. However, because Portal Hypertensive Gastropathy is a dynamic condition which improves after liver transplantation or after TIPS, suggests that it IS portal hypertension which is the primary driver.
There is a correlation between Portal Hypertensive Gastropathy and Child-Pugh stage, HVPG (hepatic vein pressure gradient), MELD score, albumin level, bilirubin, platelet count, INR and even survival [Bang CS. BMC Gastro 2016;16(93] You are right, there is also a small oesophageal varix visible!
This is an odd looking, almost 'nodular' antral gastritis. You may be surprised that this appearance is typical for a certain type of gastritis ...
WHICH TYPE OF GASTRITIS?
■ Lymphocytic gastritis
Mucosa is usually ulcerated in LyC gastritis
■ Eosinophilic gastritis
You are correct!
■ H.pylori associated gastritis
Can look weird but not classically nodular
■ Collagenous gastritis
Is endoscopically invisible!
■ CMV gastritis
Usually ulcerative
explanation
This nodular appearance is typical of an 'eosinophilic gastritis'. In textbooks you also find that lymphocytic gastritis can give rise to this appearance. However, in my experience LyC gastritis is usually intensely red with superficial linear ulceration. See my example below. CMV gastritis look similar but the ulcers are usually round rather than linear.
Anyway, finding more than 30 eosinophils per high power field (HPF) in at least five HPFs has been suggested as diagnostic. There is an argument that pathologists shouldn't make a diagnosis which doesn't change management. Not sure that I agree with this. After all, their job is to describe what they see! Nevertheless, it's probably prudent to ask about food or drug allergies, and consider if patient is atopic. A search for Helicobacters is probably also indicated. I would probably also consider taking further samples from the oesophagus as unrecognised and untreated eosinophilic oesophagitis can ultimately develop into a stricturing disease. Apart from this, it's difficult to know what to make of this diagnosis! Would you do a colonoscopy to chase after the possibility of asymptomatic IBD?
This patient has recently undergone a bone marrow transplant when she develops abdominal pain. PPI therapy has not improved her symptoms and she referred for a Gastroscopy
WHAT IS YOUR ENDOSCOPIC DIAGNOSIS?
■ CMV infection
A good guess although there are no ulcers?!
■ HSV infection
Never seen it! Does it really happen?
■ C.diff infection
Definitely doesn't happen in the stomach!
■ Acute GVHD
Actually, you are not wrong!
■ Checkpoint inhibitor gastritis
Not as crazy as you think!
explanation
Well this is a striking appearance! I'd never seen anything like it and tentatively suggested HSV gastritis on the histology request form. I didn't consider CMV because there was no mucosal ulceration.
Histology confirmed finding lots of oedematous CMV infected cells with granulation tissue and there was no need for immunostaining for cytomegalovirus proteins. This is a case of CMV gastritis! Actually, there were also some features suggestive of acute GVHD and I can therefore not be entirely sure that these 'plaque-like appearance on the mucosa isn't due to the combined effect of CMV+GVHD+PPI! It's not uncommon, one study, reported that approximately one-quarter of patients who were unresponsive to first-line acute GVHD treatment, actually had coinfection with CMV [Bone Marrow Transplant. 47, 694–699]. Of course, immunosuppressants is the trigger for CMV reactivation but CMV gastritis can occur in apparently immunocompetent patients, although a link with occult cancer has been suggested [Scand J Gastroenterol 2011;46:1228–35]. My guess of HSV was of course unlikely as HSV infects squamous mucosa or the colon (surprisingly), not the stomach. In the oesophagus you may see vesicles and/or small ulcers (left when the vesicles rupture). If I had seen this appearance in the colon I would have called this 'pseudomembranous colitis but of course C.diff doesn't affect the stomach. Can you get gastritis with checkpoint inhibitors? Yes, you can! It can cause a severe, haemorrhagic gastritis as outlined in this article [Case Rep Oncol 2018;11(2):549–556]. There is more information on CMV gastritis at this site. These polyps were found in the stomach of a 40 year old man on a Barrett's surveillance programme WHAT WOULD YOU ADVICE?
■ We should eradicate your Hp
This patient is VERY unlikely to have H.pylori
■ We should remove these endoscopically
An unnecessary job unless one look wonky, like perhaps that single red one!
■ We should stop you PPI
But the patient has Barrett's ?!
■ We should stop you PPI
That's what I would do !
explanation
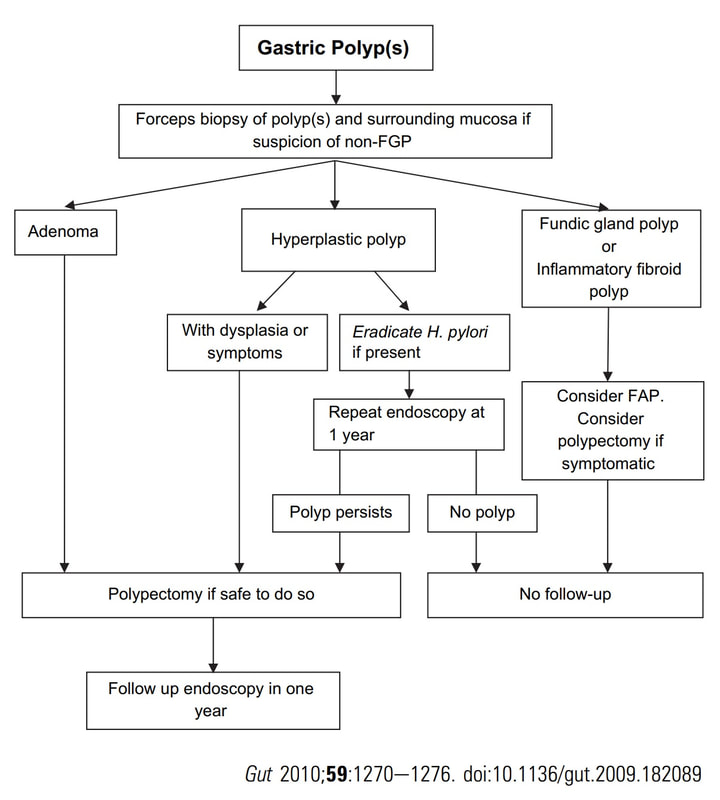
Of course, this is a Fundic Gland Polyps/Cystic Gland Polyps . They are usually multiple, somewhat transparent sessile polyps, usually in the 1-5mm in diameter and located in the body and fundus.
Histologically there are cystically dilated glands lined by gastric body type mucosa. Of course, there is no need to sample these. A study patients with FGPs reported that they can be diagnosed with a high degree of accuracy based on endoscopic appearance alone (J Clin Gastroenterol 2003;36:399-402). The sporadic polyps are caused by activating mutations of the beta-catenin gene. Not sure how an APC gene mutation (you may remember are involved in FAP), could give rise to FGP’s though! Furthermore, I don't know how the use of PPI's can be linked to the development of FGP's. Presumably its something to do with the prolonged hypergastrinaemia and enterochromaffin cell-like (ECL) hyperplasia? I have seen a couple of cases of cancer developing within a FGP. In both cases the patient had FAP and the polyps were markedly larger than all the surrounding cystic fundic polyps. There have been case reports of dysplasia arising within sporadic fundic gland polyps. A few series have reported a <1% risk of sporadic FGPs harbouring dysplasia (Am J Surg Pathol 1998;22:293-298, Eur J Gastroenterol Hepatol 2003;15:1153-6, Endoscopy 1995;27;32-37). I must admit that I don't believe the figure of <1% risk of dysplasia in a sporadic FGP. I suspect that these series consist of rather selected cases. Of course by only selecting polyps 10mm or larger, you would be filtering away 99% of the FGP's, leaving only the 'far above average risk lesions' behind. Nevertheless, the BSG recommend sampling some of them to confirm the diagnosis but not to set out on any attempt to remove them. Of course, this advice is different to the advice for Hyperplastic polyps which ARE usually linked with H.pylori and also a greater risk of developing cancer. The BSG advice is summarised in the algorithm below. Sporadic FGPs are not associated with either H.pylori gastritis or atrophic gastritis. In fact, it has been proposed that H. pylori infection may have an inhibitory effect on the development of FGPs. Of course, you wouldn't stop the PPI in a patient with Barrett's. Actually there is no evidence that patients with unstable Barrett's are any more or less likely to progress to cancer if they stop their PPI. However, there must be a point (I presume) when the use of PPI is beneficial in the 'evolution' of Barrett's. Are patients on a PPI less likely to develop Barrett's or to develop damage the stem cell DNA? Not sure! The BSG do recommend a colonoscopy in patients below the age of 40 yrs. to rule out the possiblity of FAP. However, as the average age of colorectal cancer in patients with 'attenuated FAP' is 55 yrs, I would personally recommend a colonoscopy in patients above the age of 40 yrs. This patient presented with an emergency GI bleed. WHAT IS THE DIAGNOSIS?
■ Benign gastric ulcer
Hmm, multiple GU's ?!
■ Ulcerated GIST
Multiple ulcerated GIST's. Really?
■ Adenocarcinoma
Multifocal adenocarcinoma ?!
■ Lymphoma
Too crater-like !!!
■ Melanoma
Got it!
explanation
There are actually several odd lesions in the stomach. Apart from the 'multi-focality', the lesions look a little like craters on the moon. Of course, this is what metastatic disease looks like. The dark colour clinches the diagnosis of melanomatous deposits. Here is a link to Sanjeev's and Ashok's case published in Digestive and Liver Disease [2020;52(12):1512] which is an even better demonstration of that dark pigmentation. Malignant melanoma has a peculiar predilection for the gastrointestinal tract. Melanoma can spread anywhere and is the most common metastatic tumor of the GI tract. More often the haematogenous spread is to the small bowel or colorectum. Gastric metastases are uncommon. It’s one of the more common findings in the small bowel of younger patients with anaemia. In an old post mortem series, the prevalence of GI metastasis in patients with melanoma was only 0.9% in 1000 melanoma patients (Arch Surg 1964:88:969-973). However, when you look at cases with advanced disease, up to 50% will have GI metastases (Cancer 1964;17:1323-39). As one may expect, the most common presentation is with anaemia or obstructive symptoms. Interestingly, surgical resection has been linked with a longer survival! Of course, its important to distinguish between a primary GI mucosal melanoma and metastatic melanoma. The criteria for a diagnosis of primary intestinal melanoma include 1) no evidence of concurrent melanoma or atypical melanocytic lesion of the skin, 2) absence of extra-intestinal metastatic spread of melanoma, and 3) presence of intramucosal lesions in the overlying or adjacent intestinal epithelium In a small series, the 5 year survival was 38% (Arch Surg 1996;131 :975-980). Without surgical intervention, the prognosis used to be grim and Amer et al. (Gynecol Obstet 1979;149:687-92) reported a mean survival time of 9.7 months, whilst others have reported an even shorter median survival of 4.7 months. Nevertheless, it is perhaps likely that case selection is the reason for the apparent survival benefit of surgery. The new checkpoint inhibitors and other immunotherapies are revolutionising the treatment of advanced stage disease. Just a bit of fun! Above are four types of benign polyps which can be found in the stomach, duodenum and colon respectively. I've used a 'plugin' which will hopefully make it easier to see the polyps on a small screen. BUT WHAT ARE THESE FOUR POLYPS ? A = B = C = D = You'll have to make up your mind and then click 'Explanation' below to reveal the correct answers ! EXPLANATION
The 3 polyps in column A are all adenomatous polyps. Flat and plaque-like in the stomach, pale in the duodenum and a sessile TVA in the colon. Column B shows NET's with the prominent vessels along it's side in the stomach and colon whilst the duodenal NET has the typical central depression. Column C shows 'hamartomatous' polyps seen in Peutz-Jeghers syndrome. There is hardly ever any dysplasia within these polyp and it's uncertain what the 'precursor lesion' is for these patients GI cancers Column D show inflammatory polyps which are typically angry red and sometimes with pretty white spots !
Just a bit of fun! Five 'gastric ulcers', three of which are malignant.
WHICH 3 ULCERS ARE CANCEROUS?
■ ABC
Bloody hell ! You are good!!!
■ BCD
Good guess but one is of these is benign
■ CDE
Good guess but two of these are benign
■ ACE
Good guess but one is of these is benign
explanation
Image A is a diffuse type gastric adenocarcinoma, which is strictly speaking not an ulcer (they rarely ulcerate of course). These are notoriously difficult to pick up and only show up as either a tiny pale submucosal spot, a red patch as in this case or as a more extensive patch of inflamed gastric mucosa.
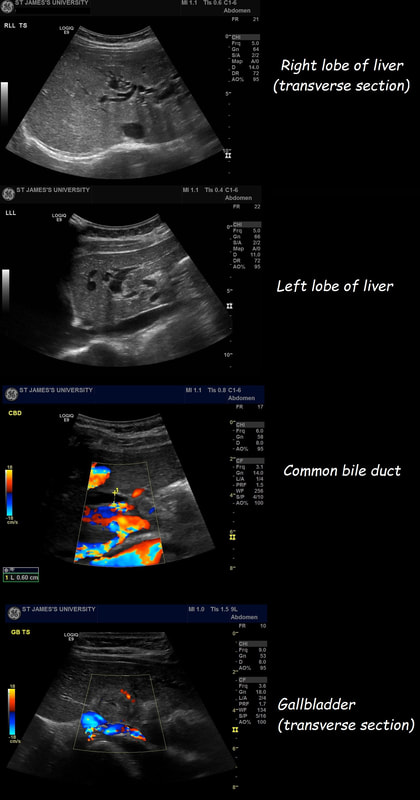
Image B was a T2 gastric cancer in which initial biopsies had been entirely benign, only reporting 'reactive atypia'. However, endoscopically there is a rim of thickened, red indurated mucosa surrounding the ulcer. Image C is an early gastric cancer pulling on the nearby gastric folds. It was removed endoscopically and turned out to be intramucosal only. Image D is of an entirely benign 'giant' gastric ulcer. ‘Giant gastric ulcers’ are (arbitrarily) what we call ulcers which are ≥3cm in size. In our Leeds series of 111 ulcers, 42 were malignant (34 adenocarcinomas and 5 lymphomas as well 3 rare tumours). In our series, logistic regression revealed that the (predictable) predictors for malignancy were; 1) increasing patient age, 2) larger size of giant the gastric ulcer and 3) the endoscopist making a diagnosis of a likely malignant ulcer. Finally E is an ulcerated fibroid polyp and not an ulcerated GIST! Of course you can't tell the two apart on endoscopic grounds! A middle aged patient presents with obstructive jaundice. There is a history of upper abdominal pain for 2 months and jaundice for about 4 weeks. She has also lost some weight loss and had night sweats. On examination she is apyrexial, obviously jaundiced and slightly tender in the epigastrium Hb 98 MCV 71 WCC 4.17 Plat 338 CRP <5 (<10) Amylase 102 (<110) Bili 75 µmol/L ALT 910 iu/L ALP 302 iu/L Albumin 37 g/L INR 1.0 An abdominal ultrasound is carried out (images below) If you work outside of the UK, and actually do abdominal ultrasound examinations you will be able to see that the gallbladder is extremely thick walled and hyperaemic. There is also intrahepatic biliary duct dilatation but the CBD is of normal calibre (6mm) and no stones can be seen. I haven't included the images showing that the pancreas, pancreatic duct, liver, abdominal aorta, spleen and kidneys were all normal. WHAT IS YOUR CLINICAL DIAGNOSIS?
■ Biliary colic
There is no pain!
■ Acute cholecystitis
How then do you explain the jaundice?
■ Choledocholithiasis
Would explain the jaundice and a CBD stone can be missed on US
■ Ascending Cholangitis
But there is no fever, raised WCC or CRP !!!
■ Acalculous cholecystitis
Sure no stones seen in the gallbladder but why the jaundice?
explanation of the case (so far - there is more to follow!!! )
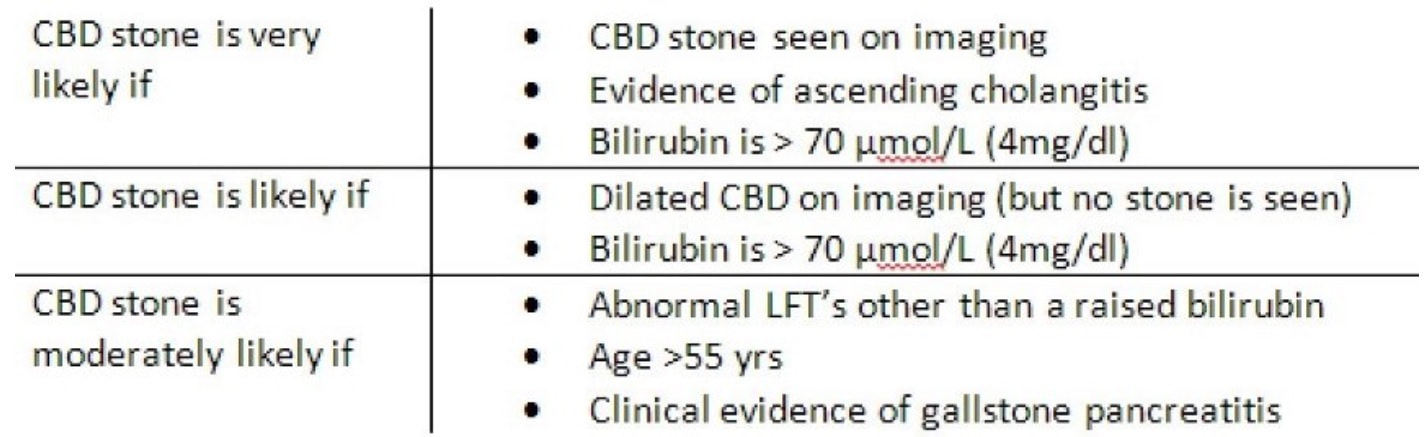
Choledocholithiasis should be your clinical diagnosis at this point. This is because the ‘strong likelihood criteria’ are fulfilled as follows;
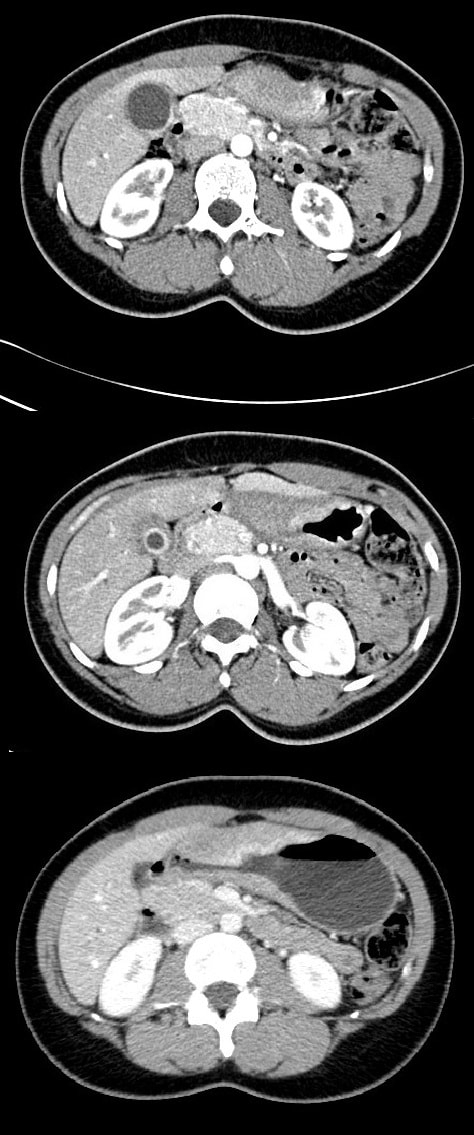
To search further for gallstones, a CT scan was then requested (below). You can probably tell from the CT that The gallbladder is confirmed as thickened with some extrinsic compression of the bile duct at the porta hepatis to explain the intrahepatic ductal dilatation already seen on ultrasound. The distal bile duct is confirmed as collapsed with normal appearance of the pancreas, kidneys, adrenal glands and spleen. WHAT IS NOW THE DIAGNOSIS?
■ Mirizzi’s syndrome
Absolutely
■ Choledocholithiasis
Nope, no stones seen within the CBD
second explanatation (and yes there are further developments around the corner)
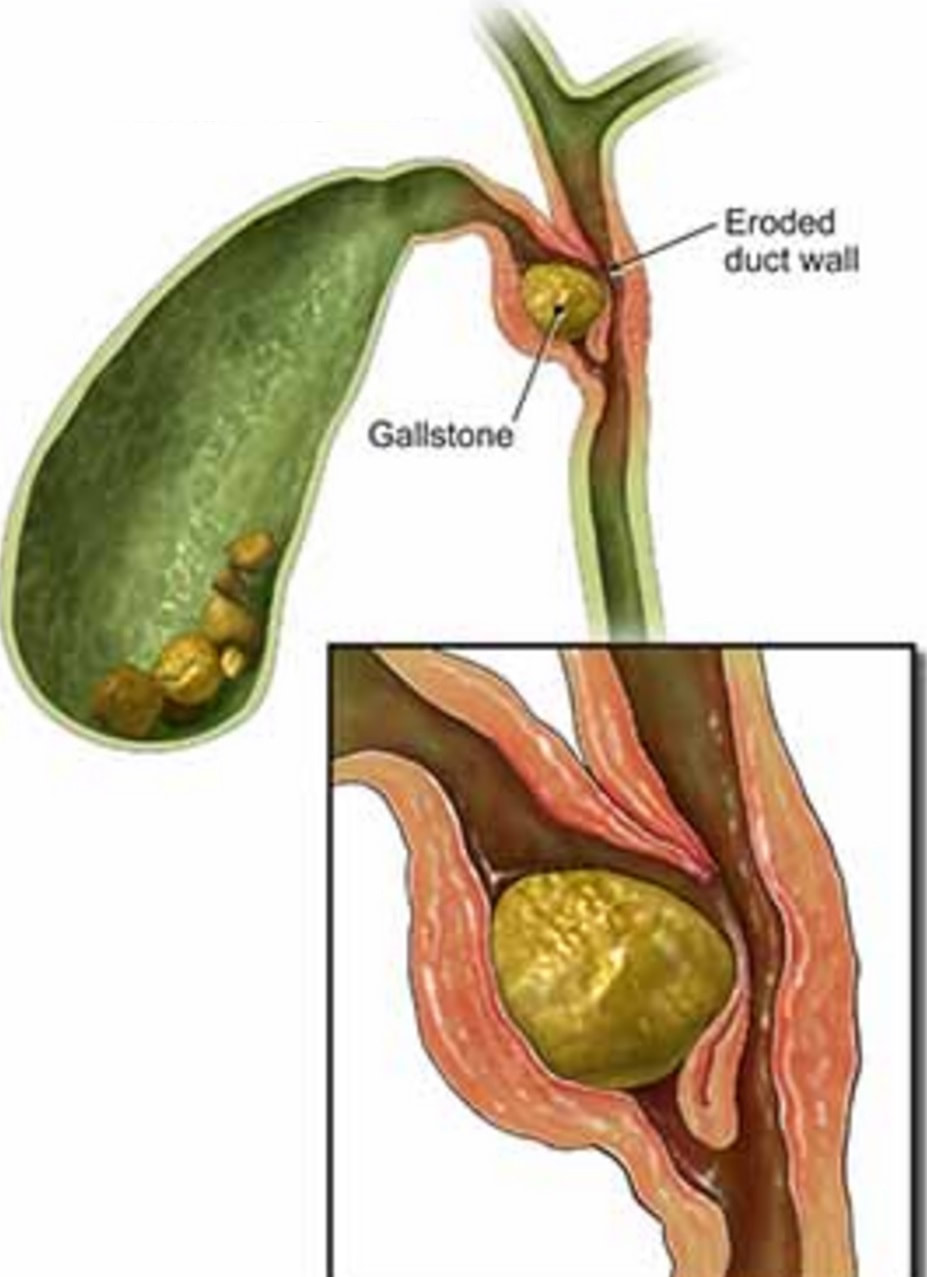
The CT report essentially describes a Mirizzi syndrome type 1. Pablo Mirizzi was an Argentinian Surgeon who first described the obstruction of the common hepatic duct (CHD) by an impacted stone in the cystic duct or Hartmann's pouch of the gallbladder. In Mirizzi syndrome type I there is no fistula between the gallbladder and CHD whilst type II-IV have a fistulous communication. Of course its difficult to tell on imaging if there is a fistula. For this reason the subtype of the Mirizzi syndrome is usually something discovered at surgery.
The patient does undergo a laparoscopic cholecystectomy but to the surgeons surprise there are no stones found within the thickened gallbladder or within the bile ducts. The gallbladder is analysed and the pathologists report that the: “Gallbladder measures 52 x 30 x 20 mm. The serosa is congested and wall thickness is 3 mm. No stone and no focal lesions. There is subacute cholecystitis with myofibroblastic proliferation of the wall and mild acute on chronic inflammation” Strangely enough, the jaundice does not resolve after surgery and for this reason an MRI scan is carried out which confirms a stricture at the common hepatic duct with mild intrahepatic ductal dilatation. At this point we decide to carry out an ERCP to sample the stricture and place a stent. Now you have all the pieces in this jigsaw and should know what's going on! WHAT DID WE MISS?
■ Primary cancer of the gallbladder
No cancer found in resection specimen!
■ Cholangiocarcinoma
You missed something on the video!
■ Gastric cancer
Well done, you spotted this on the video?
■ Duodenal cancer
But the duodenum look fine on video?!
FINAL EXPLANATION
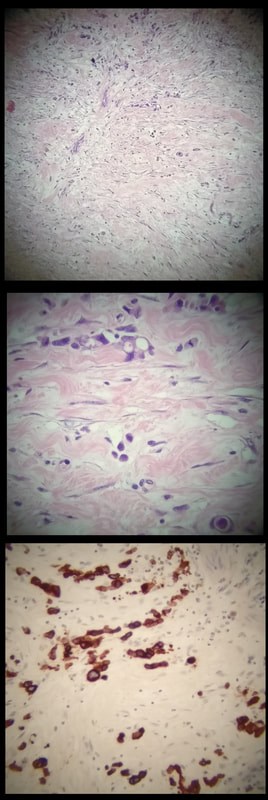
Thanks for sticking with this case until the end! Actually, you did have an opportunity of getting the diagnosis when you heard that the gallbladder histology showed; "markedly thickened wall but with only mild inflammation of the gallbladder mucosa" ...
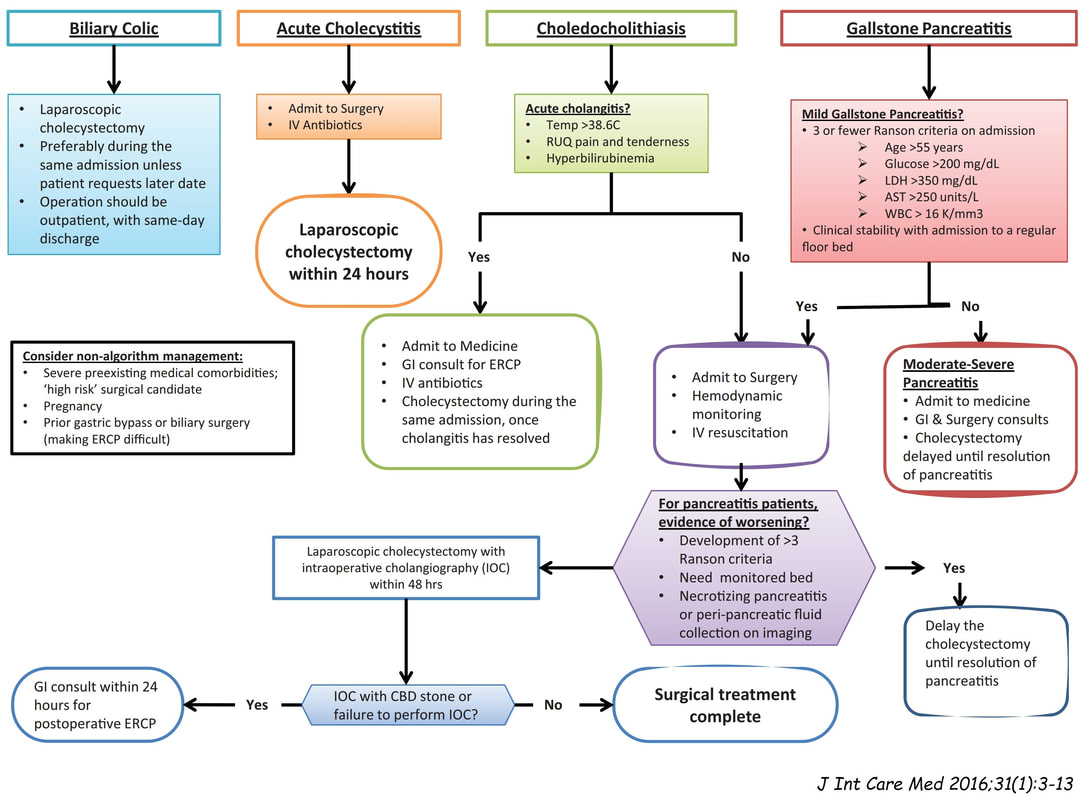
Of course, this doesn't make any sense! In acute cholecystitis there should be an INTENSE inflammation of the mucosa. So why is the gallbladder wall grossly thickened? A second look into the deeper aspects of that thickened gallbladder wall led to a revelation ! The 'second look histology' reported a 'poorly differentiated diffuse type adenocarcinoma deep within the gallbladder wall with single and files of small neoplastic epithelial cells (histology slides below). The pathologists reported that the tumour did appear to be coming from outside the gallbladder. Of course in the video you should have noticed that the gastric antrum was abnormal, a little indurated and thickened. The samples taken from the gastric antrum confirmed the same diffusely infiltrating adenocarcinoma. Those with VERY sharp eyes, would have seen the antral thickening on the initial CT which wasn't commented upon by the radiologist! So what was the final diagnosis? A diffuse type gastric cancer invading into the gallbladder, cystic duct and hepatic duct ! This may a good time to remind you of the management of gallstone related disease. The 'infographic' below from J Int Care Med 2016;31(1):3-13 summarises everything !
I'm carrying out a dilatation (20mm) of a distal gastric stricture. WHAT IS THE LIKELY AETIOLOGY OF THE STRICTURE?
■ Benign pyloric stricture
But the stricture is pre-pyloric!
■ Anastomotic stricture
Doesn't look like an anastomosis!
■ Radiotherapy stricture
But why giving radiotherapy to the stomach?
explanation
The mucosa in the 4 o'clock position looks pale and atrophic. This is a 'scar' left after treatment of a gastric lymphoma. Of course, the scar would look the same following Hp eradication of a MALT lymphoma. However, this was after following chemoradiotherapy (CRT) for a 'Diffuse large B-cell lymphoma. Which is a far more nasty lymphoma and patients present with more advanced stage at diagnosis, and have a worse prognosis (about 75% of patients are alive at 5 yrs) than with a MALT lymphoma. This patient was treated with 6 cycles of R-CHOP chemotherapy and radiotherapy 30 Gy in 15 fractions. The risk of stricturing after gastric CRT is around 5%. My dilatations to 20mm didn't do much and the patient had to come back for a dilatation to 25mm before symptoms improved.
This ulcer was found on the lesser curve of a middle aged man with dyspepsia. Biopsies are taken and stained with H&E (below)
WHAT IS THE AETIOLOGY OF THAT GU?
■ Benign GU
Don't you think that the mucosa look odd near to the ulcer?
■ Lymphoma
Yes! That odd atrophic mucosa next to the ulcer look weird but not "carcinoma weird" !
■ Adenocarcinoma - intestinal type
With a 'standard cancer', the surrounding mucosa should be thickened and red not atrophic looking
■ Adenocarcinoma - intestinal type
This was actually my own diagnosis but histology shows lots of small round lymphocytes not ballooned up signet ring cells.
EXPLANATION
There are only two possibilities here. Of course the ulcer itself doesn't tell you much. The mucosa surrounding the ulcer tells you more! With a benign ulcer, you would expect a thin rim of 'reactive' mucosa (looks red and a little villous). In the case of an intestinal type of adenocarcinoma a broad band of surrounding mucosa is red, indurated and thickened. In gastric lymphomas, the surface area of abnormal mucosa is usually far greater than the surface area of the ulceration. There are usually areas of atrophic and inflamed mucosa. Mucosa infiltrated by a diffuse type adenocarcinoma could look like this BUT it's unusual for it to ulcerate!
This patient has a MALT lymphoma ! Of course, I took samples for Helicobacters. Of course not every MALT lymphoma is linked with HP infection (about 2/3 cases are though). In this case our haematologists were optimistic of a good response to Hp eradication because all the 3 good prognostic criteria were met:
This was polyp found on the greater gastric curve.
WHAT IS THE LIKELY DIAGNOSIS?
■ Hyperplastic polyp
No! Those look like mushrooms usually
■ Fundic gland polyp
No, those look like frogspawn when you look closely
■ Adenomatous polyp
Absolutely, to be specific an 'intestinal type adenoma'!
■ Early gastric cancer
It still has a crypt pattern!
■ Invasive gastric cancer
Impossible as it still has a crypt pattern!
explanation
Clearly there is a discrete nodule containing some small round crypt openings. If a dysplastic (mutated and genetically unstable) lesion produces a visible lesion, the pathologist calls it an 'adenoma'. Dysplasia in a flat mucosa is just called 'gastric dysplasia'. Of course in the colon there is a long established 'adenoma → carcinoma' sequence first proposed by Basil Morson a famous British pathologist. He estimated that about 2/3 colorectal cancer must be arising from an adenomatous precursor. What gives rise to the rest remained a disputed mystery until Jeremy Jass, another famous pathologists (and I really nice guy as I found, meeting him at a conference) proposed that serrated polyps may also give rise to cancer [Histopathology 2006;49:121-31]. His suggestion and data to support it, didn't really go anywhere for some 10 years but after he died in 2008 of glioma, the 'serrated pathway' to bowel cancer has become accepted.
Anyway, in the stomach things are even more complicated! There are at least 4 types of adenomas:
Of course, not every gastric polyp is adenomatous. Most are hyperplastic and look angrily red (because they have lots of capillaries) and often with white fibrin caps, making them look a little like mushrooms and may have a villiform surface reminisient of TVA's in the colon. Another common type are the 'Fundic gland polyps' which are full of cystic spaces and therefore look a little like frog-spawn. These usually develop in patients on PPI's but also in FAP. Polyps may also be hamartomatous as in 'Peutz-Jeghers syndrome', 'Cronkhite-Canada syndrome' and 'Juvenile polyposis'. By the way, the word 'Hamartomatous' implies that the polyp has arisen from overgrowth of multiple aberrant cell lineages rather than from a single, mutated precursor cell as in an adenoma. And then of course there are neuroendocrine tumours (NET's). A whole different thing which has a complete podcast dedicated to it. You can tell that a gastric polyp is a likely NET as they usually have little vessels crawling up it's sides. Finally, the lesion may be a gastric metastasis most commonly from breast, kidney, melanoma or HCC. By the way, there is a pitfall here as 'lobular breast cancer' mimics diffuse type gastric cancer. For this reason you should inform the pathologist if the patient has a history of breast cancer and you suspect a metastasis! This lesion was found in the antrum. The histology is also attached. WHAT IS THE LIKELY DIAGNOSIS?
■ Hyperplastic polyp
Would be red with white caps!
■ Adenomatous polyp
Yes and histology confirms HGD only
■ Superficial gastric cancer
Could be but histology is benign...
explanation
Most EGC's look a little like basal cell carcinomas with an elevated rim surrounding a central depression. This is a good example of what an EGC could look like. As it happens though, it only harbours HGD. Because you can't really tell endoscopically (the pathologists struggled as well to make a distinction sometimes) There are more than one type of 'gastric adenoma':
This was reported as an intestinal type adenoma harbouring HGD !
This was noted on the greater gastric curve
WHAT DOES IT MEAN FOR YOUR MANAGMENT?
■ I'll take a biopsy
No harm in this!
■ I'll check the patients cholesterol levels
No more likely to be abnormal than avg!
■ I'll check the patients urate levels
Don't think that you can get tophi in the gut!
■ I'll look carefully for EGC's
Brilliant!
■ I'll remove it by EMR
You are wasting your time!
explanation
Of course this is a 'Gastric xanthelasma' which histologically consists of macrophages stuffed full of lipid. There isn't a strong link with abnormal serum lipids but there is a strong link with gastric atrophy and gastric intestinal metaplasia ...
You will be familiar with 'Correa's hypothesis' and will know that widespread gastric atrophy and intestinal metaplasia is what's left when Helicobacters has run ravage for decades, leaving the stomach atrophied, achlorhydric and at risk of developing early gastric cancer! I used to regard gastric xanthelasma as a 'curiosity' only but actually, they are signposts that you should slow down and look VERY carefully for EGC's !!! |
Categories
All
|