|
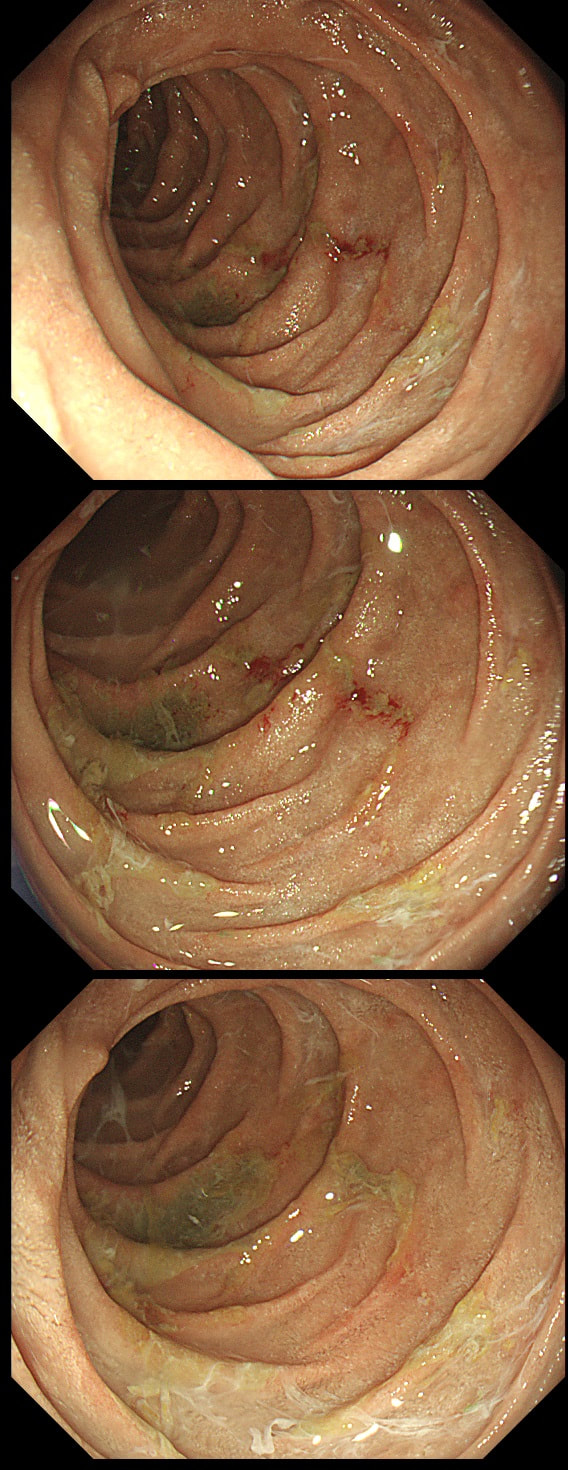
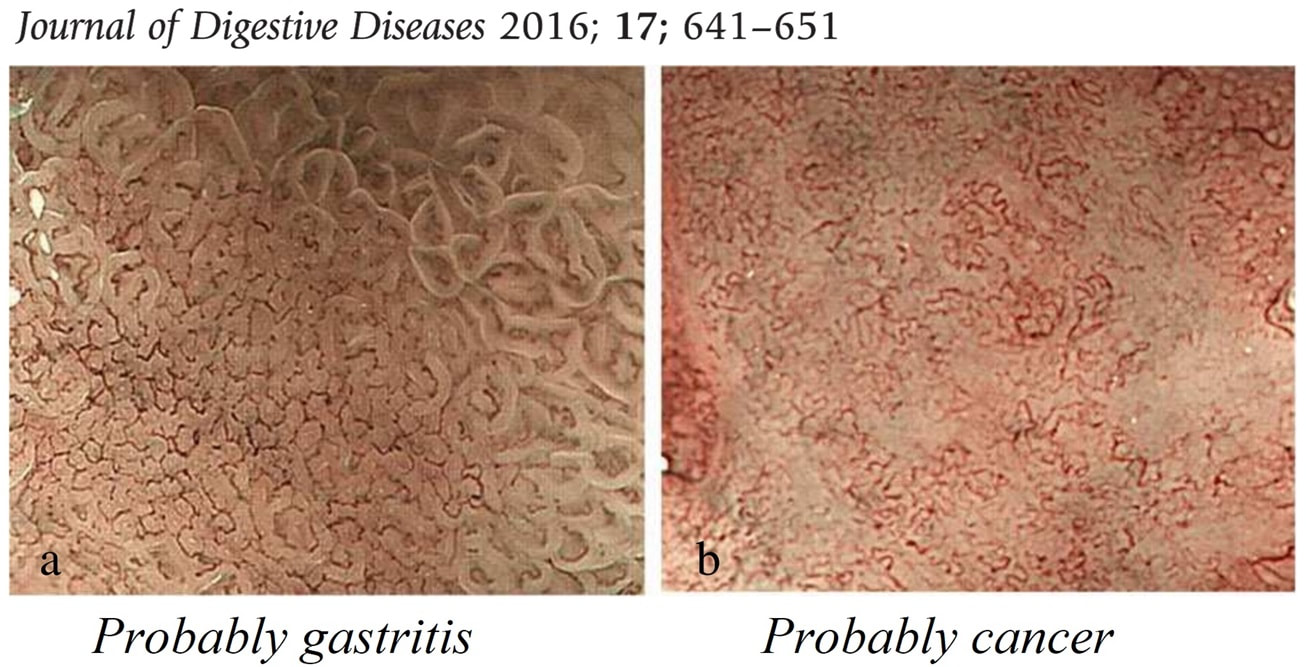
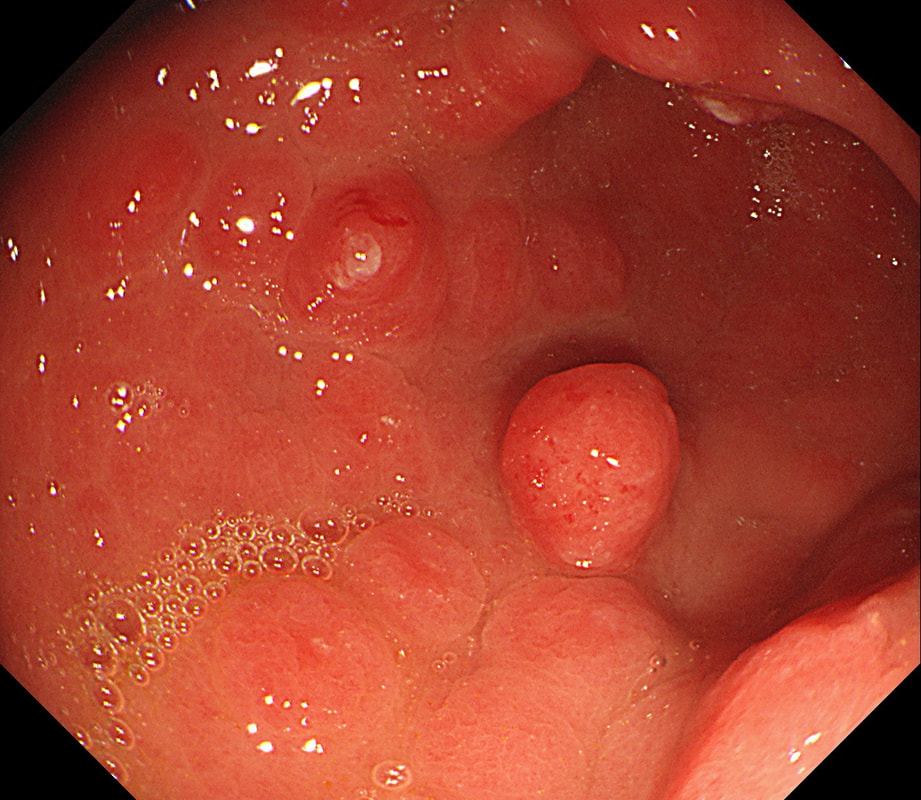
This patient was undergoing a gastroscopy for iron deficiency anaemia. A small ulcer is found in D2.
WHAT IS THE LIKELY AETIOLOGY?
■ Peptic ulcer
Statistically probably most likely diagnosis
■ Pill ulcer
Yes, but what pill?
■ Adenoma
Often do appear depressed here (IIc lesions) but not in this case
■ Early cancer
Early duodenal cancer usually look like sessile adenomas or, less commonly like indurated ulcerated lesions
explanation
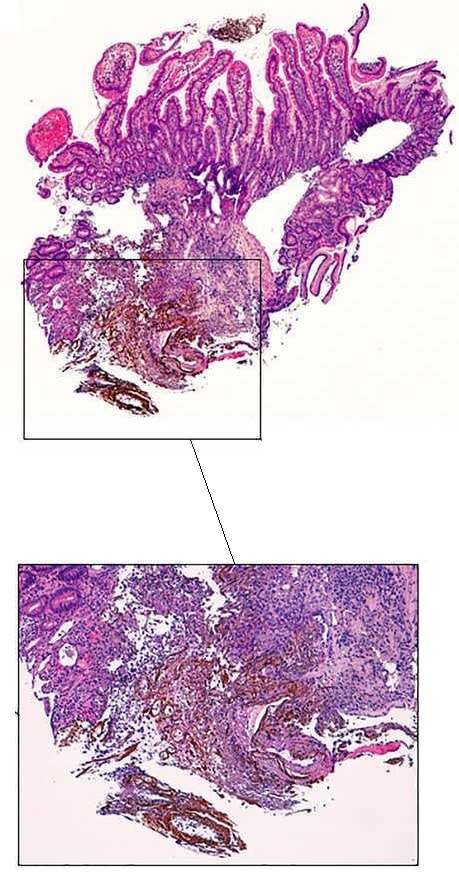
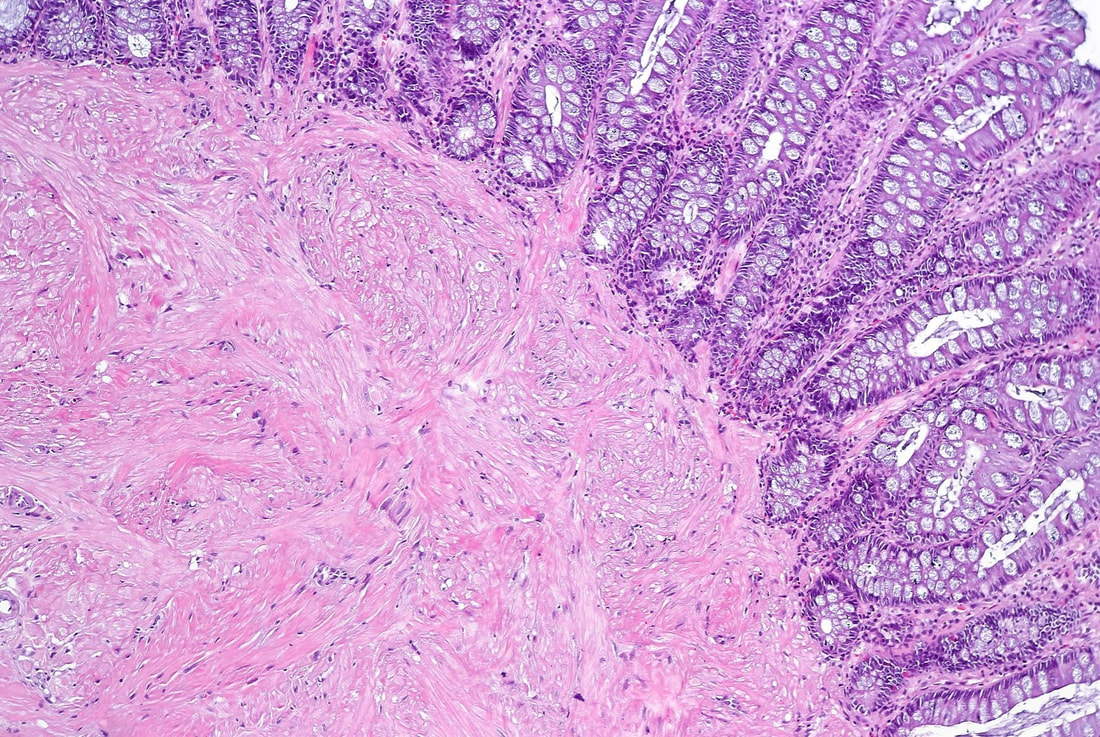
If it wasn't for the somewhat odd, elongated appearance of the ulcer, it wouldn't seem odd. However, the only clue to the aetiology is that yellowish appearance. You may be familiar with 'iron pill gastritis' but can you get iron pill duodenitis? Yes you can! It has even been reported in the hypopharynx and the oesophagus. Those dark spots on the histology slides are crystalline iron deposits are easy to spot for the pathologists.
Of course, ferrous sulphate (Fe2+) tablets is the non-toxic form of iron (naturally). However, the tablets can produce mucosal inflammation if the iron becomes oxidised to Ferric iron (Fe3+). You may remember from medical school that most dietary nonheme iron is in the toxic, oxidized form of ferric iron (Fe3+). To absorb iron it first needs to be reduced, in the presence of acid, to ferrous iron (Fe2+). After this, the iron is easily transported across the enterocytes via a divalent metal-ion transporter 1 (DMT1) protein. Here is a link to an open access article if you want to brush up on iron absorption. Naturally, the treatment is to change to the liquid form of iron supplement which is much less toxic to the mucosa than solid iron tablets.
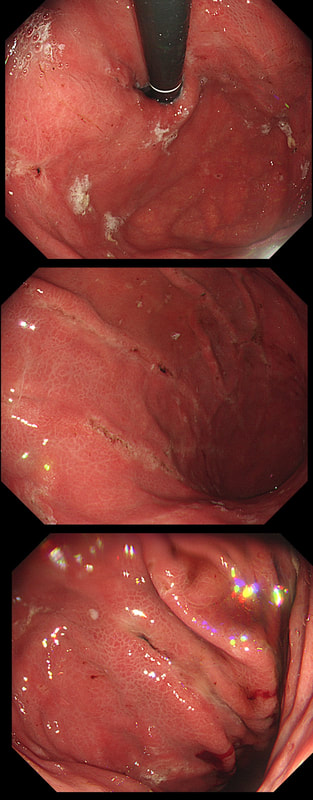
This lesion was found in the rectum of a patient undergoing colonoscopy because of constipation
WHAT IS THE LIKELY DIAGNOSIS?
■ Polyp from mucosal prolapse
Clever!
■ Serrated polyp
Crypts look a little serrated but the rest doesn't
■ Adenomatous polyp
Polyp doesn't look 'neoplastic'
■ Malignant polyp
Now, this doesn't look malignant!
explanation
Well perhaps the crypts look a little like serrated crypt openings but somehow the rest of the polyp doesn't look like a typical serrated lesion. Where is the covering mucus?! Furthermore, the polyp definitely doesn't look adenomatous or malignant!
Actually this has arisen as part of the 'Mucosal prolapse syndrome' which is the umbrella term for entities such as; solitary rectal ulcer syndrome and inflammatory cloacogenic polyps. Patients are often constipated or have difficulty with defaecation with straining on the toilet or undergo the sigmoidoscopy because of tenesmus, altered bowel habits or incontinence. Surprisingly, some patients don't have any straining-related complaints! Most pathologists would recognise the typical mild fibrosis, thickening of the muscularis mucosae and crypt irregularity (dilated, diamond shape crypts). The surface epithelium show regenerative changes.
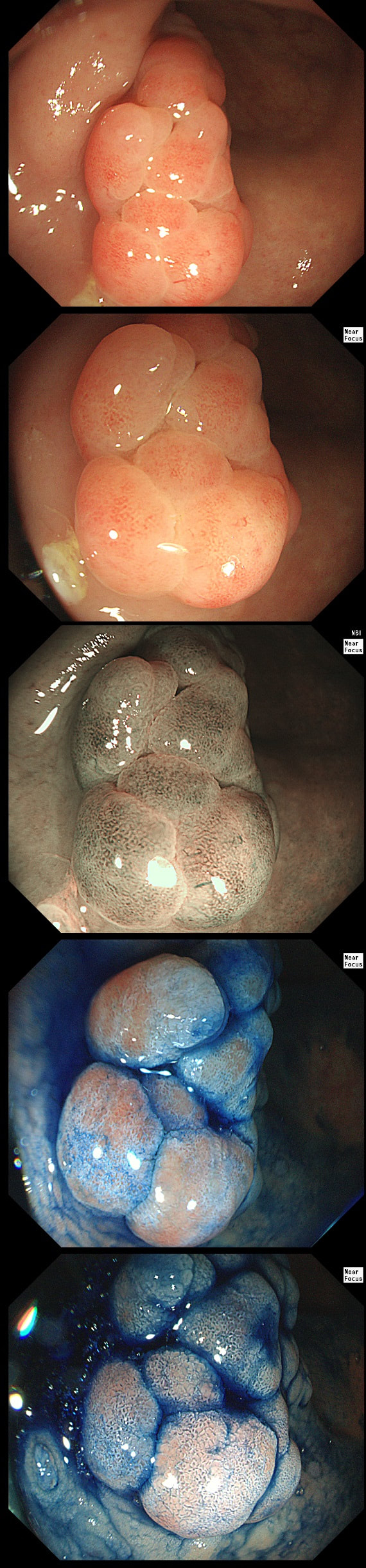
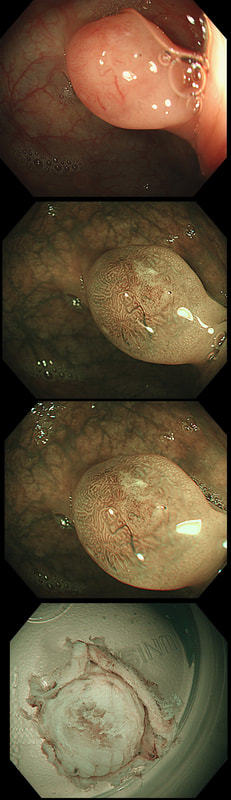
This polyp was found in the sigmoid.
HOW IS IT BEST REMOVED?
■ Cold snare polypectomy
Wouldn't be my first thought!
■ EMR
I agree - single fragment EMR!
■ ESD
Would just add time and expense to procedure (unless you need practise, I guess)
explanation
This 10-12mm polyp is covered with a beautiful gyrate crypt pattern typical of a TVA. The risk of cancer deep within, in spite of the normal surface is far below 1%. But it's not 0% and it would also be a little difficult to cut through this amount of tissue. For these reasons, I think that 'cold snaring' would be wrong.
At the other end of the spectrum, we have an ESD. It would ensure a single fragment resection but would take about 20 minutes or so. The in between the two, we have an EMR. If course, a single injection of 3-4ml would lift this nicely and then a 15mm snare would resect it, single fragment, within a minute. I think that the best method of removal of polyps up to about 2cm, is by EMR as polyps up to this size can usually be removed as a single fragment. This is an elderly patient undergoing an OGD because of IDA (iron deficiency anaemia). A lesion catches my eye on the anterior gastric wall WHAT IS THE LIKELY HISTOLOGY?
■ Gastric xanthelasma
The colour is suggestive and gastric atrophy would be the link but it's not quite right...
■ Healed GU scar
But how about that funny crypt pattern?
■ Gastric adenoma
Yes, doesn't this look like a tubular adenoma?
■ Early gastric cancer
Wouldn't the crypt pattern then be very disorganised or perhaps very small?
explanation
The pale colour is odd and reminiscent of gastric xanthelasma which as you know is linked with gastric atrophy which is the likely cause of this patients IDA. However, when I zoom in on the area, the crypt pattern is different here. Of course, this does not fit with a xanthelasma or a scar from a healed gastric ulcer (GU) either for that matter. Interestingly, almost everyone thought that this was an EGC. However, THERE IS a distinct crypt pattern in the centre of the lesion. Furthermore, the lesion isn't red. Remember that cancers encourage the growth of small irregular capillaries which gives them a red colouration. Finally, it doesn't have the typical flat-elevated with a central depression (IIa+IIc) growth morphology. Therefore, your first guess should be a gastric adenoma! This is actually a gastric tubular adenoma which we found in an elderly frail patient with atrophic gastritis some 10 years ago. As she had some comorbidities and it was only harbouring LGD, we decided to keep an eye on the lesion on a yearly basis. The risk of progression is supposedly only 5% with tubular adenomas in the stomach. In contrast, villous adenomas are much more likely to progress (40%). The BSG gastric polyp guidelines have the references if you want to look this up. Of course, the issue is not entirely clear-cut as risk of progression also increases with the size of the lesion (and this is probably 2cm in size) and also with age (patient is now 86 yrs). In some ways, making an initial decision to either 'attack' or 'abort' would be easiest. After all, regular surveillance drains valuable resources and leaves you open to the possibility that at some point in the future, the patient is no longer a candidate for anything more invasive than a haircut but now the lesion under surveillance shows evidence of progression. Then your patient could well ask the legitimate question why you didn't go ahead when he was younger and fit enough but instead wasted his time with pointless surveillance?! My own preferred way to navigate this minefield is to openly discuss the three options with the patient;
I often wonder if its the personality type which dictates what patients prefer. Perhaps, those who think 'my glass is half empty' usually want to have their lesion resected immediately whilst people who regards their 'glass to be half full', prefer to hope for the best and get on with their lives? This patient is on a surveillance programme due to alcoholic liver disease but has never had a bleed. He is maintained on a non-cardioselective β blocker. WHEN WOULD YOU RECOMMEND THE NEXT SURVEILLANCE EXAMINATION?
■ None, this patient should be offered banding
Correct, if you decide that there are 'red signs' or the varices are grade II or III
■ In 12 months time
Correct, if you decide that there are no 'red signs' and that the varices are grade I
■ In 2-3 years time
Would only be correct if there are no varices at all!
explanation
I must admit that I don't like the current surveillance guidelines for patients with portal hypertension. This is a good example why! Although the LFT's had remained stable and the patient had remained abstinent, you could argue that there are 'red signs'. Of course, the presence of 'red signs' predicts progression of varices [J Hepatol 2003;38:266–72] and because the patient is already on a β-blocker, band ligation should now be started. The truth is that 'red signs' are common and have a poor agreement value between endoscopists.
If you decide that there are no 'red signs', and that the varices are grade I only (which depends on the degree of inflation of the oesophagus), the recommendation is to offer surveillance in 1 year. Finally, if you decide that the varices are now grade II (or III), variceal band ligation would be the logical next step. Thus, you can make an subjective argument for any of the above three treatment options. Of course, what you decide will be judged in hindsight. If you decide that these are not red signs and the patient re-presents with a bleed in 6 months time, you could be open to criticism for missing signs of progressive liver disease. Surely, in the modern era of FibroScans, it's possible to predict progression of portal hypertension non-invasively!!! Five years ago, the BAVENO VI workshop only mentioned in passing, that surveillance endoscopies may be avoided in patients with elastography values <20 kPa and a platelet counts >150,000 as these patients are at low risk of progression. Similarly increasing size of the spleen is another warning sign and could be looked for when these patients attend for screening for HCC's. The American guidelines suggest that there is no need to offer patients with untreated viral cirrhosis a screening endoscopy to search for varices IF elastography is <20 kPa and the platelets are >150. They concede that annual elastography and platelet counts may be less predictive in other causes of cirrhosis. However, the American guidelines advice continued surveillance if varices have been found in the past, particularly if liver injury is ongoing. The next Baveno conference in October 2021 will hopefully recommend non-invasive monitoring rather than endoscopy. It would be cheaper, less arduous for patients and offer less subjective findings! The now rather dated BSG guidelines are summarised in the graph below. These three polyps were removed en-bloc from the proximal colon. In an earlier examination 2 SSL's had been removed. A subsequent colonoscopy finds no further polyps. WHAT WOULD YOU ADVISE AS REGARDS SURVEILLANCE?
■ There is no need for surveillance
You may be right if the patient is old and with multiple comorbidities!
■ Surveillance every 2 years
Yes, this is what guidelines usually recommend for 'SPS'
■ Surveillance every 5 years
Too long if pt fulfills WHO criteria for SPS
explanation
If course, this patient has 'Serrated Polyposis Syndrome'. It's important to recognise that the serrated polyp count is cumulative over multiple colonoscopies. The recently updated 2019 WHO criteria for Serrated Polyposis Syndrome recognize two types of the syndrome: a 'proximal phenotype' with serrated polyps proximal to the rectum, all being ≥5 mm in size, with at least two being ≥10 mm in size (criterion I 2019), and a more 'distal phenotype' with more than 20 serrated polyps of any size throughout the large bowel (criterion II 2019) [Gastroenterology 2020;158:1520–23]. Personally, I believe that there are more than two subtypes of the Serrated Polyposis Syndrome. There is accumulating evidence that the syndrome includes multiple conditions with variable phenotypes and with different risks of progression to CRC [Gut 2010;59:1094–1100]. This would explain the huge range of cancer risk (25%-70%) in published studies [GIE 2016;83:563–65]. Of course, the likely mixture of several 'syndromes', makes writing guidelines difficult. A recent consensus update by the US Multi-Society Task Force recommend offering a follow-up colonoscopy to average risk patients based on number and size of the SSL's found [GIE 2020;91:463–85]. Interestingly, the US guidelines make a distinction between 'hyperplastic polyps' and 'sessile serrated polyps' although pathologists can't reliably make that distinction. Furthermore, the guidelines excludes patients with an increased life-time risk of cancer which of course excludes patients with Serrated Polyposis Syndrome. I find it all somewhat confusing! James East's BSG guideline 2017 [ Gut 2017;0:1–16 ] recommend surveillance every other year whilst the more recent BSG/ACPGBI guideline of 2020 [ Gut 2020;69:201– 23 ] would seem to suggest 3 yr for all 'high risk cases. But these guidelines expressively don't cover pts with hereditary cancers. Hereditary cancers are instead covered by the BSG/ACPGBI 2019 guideline [ Gut 2019;0:1–34 ] which recommends annual surveillance until all polyps are cleared and then every 2 years. Finally, I admit that I also take the age of the patient into consideration as well as the presence of both serrated and adenomatous polyps. A 40 year old person is surely more likely to benefit from surveillance than a 75 year old person with multiple comorbidities? In particular, I would worry about a young patient, perhaps 35 year old, with 1-2 large serrated polyps and perhaps only a single adenoma. Current guidelines don't flag these individuals up but personally, I would organise another surveillance colonoscopy in a few years time. Clearly, more research is needed to unpick the different serrated sub-pathways ! This lesion was found in the sigmoid. You have magnification, NBI, dye spray and lift to help you decide. WHAT IS THE STAGE OF THIS CANCER?
■ Intramucosal
There is no such thing!
■ sm1 invasion
You can see a disorganised crypt pattern?
■ sm2 invasion
That was my guess!
■ sm3 or 'massive' invasion
I think that the lift is better than that!
explanation
I think that it's difficult to tell the difference between no crypt pattern at all and a severely disrupted crypt pattern. Of course, when the crypt pattern is 'severely disorganised' but some of it is still visible, the lesion will be sm1 or sm2. In contrast, if there is no crypt pattern at all, the lesion is sm3 or beyond (the Japanese call this 'massive invasion'). Instead, I rather rely on the degree of lifting. In this case, the endoscopist decided that the lifting was insufficient for a resection and backed off, referring the patient to our MDT. Biopsies confirmed that the lesion was likely to be malignant and the patient ended up with sigmoid resection. I think that there is some sort of crypt pattern in the centre of the lesion. Furthermore, looking at the slight degree of lifting, my guess would be that the lesion is sm1 or sm2 and therefore potentially endoscopically resectable. Actually the cancer turned out to be sm2 (T1,N0). These polyps are found in a rectal pouch WHAT IS THE UNDERLYING DIAGNOSIS?
■ Serrated polyposis syndrome
Polyps is SPS are flat (usually)
■ FAP
Looks like a classic example of rectal surveillance in FAP but polyps do look odd don't they?
■ Juvenile polyposis
Yes! Pt had already developed a CRC and after a subtotal colectomy offered rectal surveillance
explanation
These polyps have a funny crypt pattern, but THERE IS A PATTERN! Accordingly, they are likely to be 'hamartomatous' (a non-sensical histological term which basically means overgrowth of normal tissue). Of course, this patient has Juvenile Polyposis syndrome. Although the polyps are 'hamartomatous', as in Peutz-Jeghers syndrome, there is a stark difference. The polyps in JP often turn dysplastic and are presumably the origin of this patients adenocarcinomas. In contrast, the polyps in PJS are extremely rarely reported to harvest dysplasia (I've never seen a dysplastic polyp in PJS). By the way, the WHO criteria for diagnosis of juvenile polyposis syndrome are one of either:
This patient is under surveillance after having had two previous EGC's. The stomach has been sprayed with indigo carmine dye when this lesion caught my eyes. WHAT IS THE LIKELY DIAGNOSIS?
■ Likely to be gastritis only
The non-staining is suspicious and rarely lie...
■ Likely to be a patch of intestinal metaplasia
There appears to be a crypt pattern in the centre but it can still be neoplastic!
■ Likely to be neoplastic
Non-staining, slightly raised and in an 'unstable stomach' - surely!
explanation
Clearly this is an unstable stomach having given rise to two previous EGC's. In patients attending for surveillance, I always spray indigo-carmine dye throughout the stomach using a spray catheter. I then look carefully at any spots where the dye seems to have slid off. This is such a 'lesion' which was confirmed as a subtle EGC. After my samples had confirmed my suspicion it was removed by ESD and was confirmed as HGD/IMca. I consider the both as the same histology as one pathologists HGD is another pathologists IMca. There was no LVI. However, I was surprised to find that one mucosal resection edge was involved by cancer. Looking back at the clip, I think that it's the 4 O'clock border which has an irregular extension which I failed to spot.
Most EGC's look like this, a shallow depression with a subtle elevated margin. I find the crypt pattern more difficult to interpret. However, as you know, the more destroyed it is, the more likely the lesion is to be poorly differentiated. This polyp was found in the transverse colon of a 60 year old man. WHAT IS THE LIKELY HISTOLOGY OF THE LESION?
■ Serrated polyp
Crypt openings look a little serrated but nothing else does!
■ Adenomatous polyp
This is not an adenomatous crypt pattern!
■ Hamartomatous polyp
When in doubt, it's hamartomatous !
■ Malignant polyp
The crypt pattern is too organised and regular for cancer!
explanation
Sadly, an intimate knowledge of the Kudo crypt patterns doesn't help you here! Adenomatous polyps should be covered with one of the following;
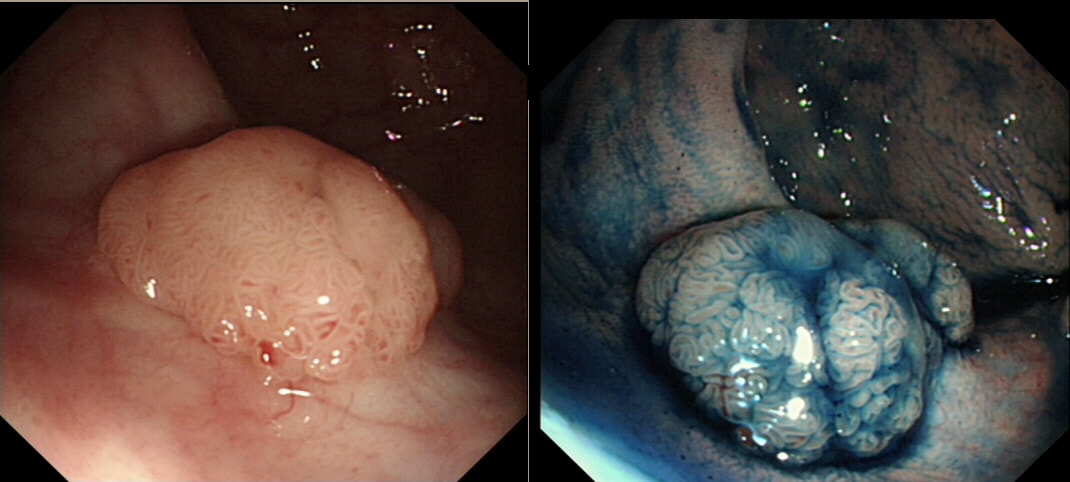
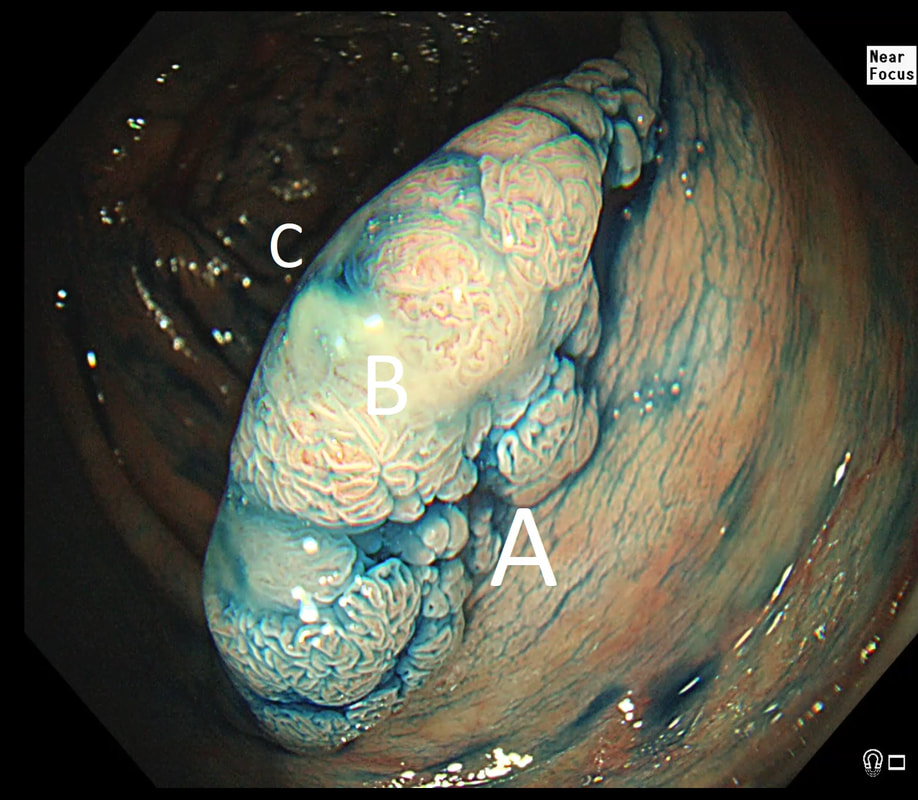
In this case, the closest match is of a serrated polyp, which have wide open crypts with a somewhat jagged outline. However, apart from the crypt openings, there is nothing on this which looks like a serrated polyp! If you can't see a crypt pattern, the lesion is likely to be malignant. Conversely, if the lesion does have crypts but still doesn't look familiar, its either a hamartomatous polyp or, perhaps less likely, a 'Traditional Serrated Adenoma'. This lesion turned out to be a hamartomatous polyp. Why a 60 year old man would grow a hamartomatous colonic polyp remains a mystery ! This is beautiful polyp, perched on a fold, was found at the junction between the caecum and the ascending colon. The video gives you a better idea of the size and extent of the lesion. WHERE WOULD YOU PLACE THE NEEDLE FOR THE BEST 'LIFT' ?
■ On the side facing you
A convenient choice but lift will not be optimal
■ Into the apex of the polyp
That would be my choice!
■ Just behind the lesion
Better than into the fold facing you
explanation
I have no qualms about injecting straight into the middle of lesion provided that I'm sure that it's benign. The lovely gyrate pattern of this polyp tells you that it's a TVA, likely to harbour no more than LGD. Injecting into the fold facing you is likely not to raise the 'blind side' of the polyp which extends down the back of the fold and onto the 'flat' beyond. Conversely, injecting into the back of the lesion would lift the back end but probably not the front. My choice was to inject into the apex (see video below) which resulted in a lovely lift. But, why not inject in two places? Because you should try to avoid injecting into more than one place (if it can be avoided). If you have made more than one hole in the epithelium you may will find that your injection leaks out through the previous hole and that the elevation is less effective. Of course when removing large, flat lesions multiple injection sites can't be avoide, unless you are removing the lesion by ESD of course.
■ Inflammed granulation tissue
Was my first too! But it doesn't quite look right ...
■ Adenomatous polyp
Adenomatous polyps are usually smooth topped
■ Malignant polyp
An intramucosal cancer arising from the anastomosis
explanation
This patient had in the past undergone a Billroth II operation. You can see that the nodule is arising from the surgical anastomosis. Distal gastrectomy is a well-known risk factor for developing an anastomotic cancer later [Sitarz R. World J Gastro. 2012;18(25):3201–6]. The risk of anastomotic cancer steadily increases after surgery. About 15 years after surgery, the risk exceeds that of the background population (age- and sex-matched). For this reason, surveillance has been suggested to start 15 yrs after surgery. The case for surveillance was strengthened by the fact that dysplasia can often be found in random biopsies from the anastomosis several years before cancer develops. In spite of this surveillance in not recommended. A surveillance study in Amsterdam traced 500 patients who had undergone a distal gastrectomy for benign disease and only detected 10 cancers (6 were in an early stage). Furthermore, there was no survival advantage in the screened group after 10 years follow up [J Clin Pathol. 1984;37:748–54]. Other studies have also put the cancer yield by surveillance at around 2% and concluded that regular surveillance could not be recommended [Am J Surg 1977;134:581-4], [ Lancet 1977;ii467-9] [ Scan J Gastro 1981;suppl 16:169-71]. Nevertheless, my practise is to always take do 'opportunistic screening' by obtaining 6 biopsies or so from the gastric side of the anastomosis when I come across a case. On first glance the nodule appears to be inflamed granulation tissue only. However, there is an odd cleft in the centre and the base from which it arises is also nodular. The polyp was removed and was confirmed as harbouring intramucosal cancer. Of course, after finding dysplasia (which often does not progress) or IMca, these patients should be offered surveillance.
This was found at gastroscopy in a middle aged patient with anaemia
WHICH STATEMENT IS CORRECT?
■ The severity is related to the severity of the underlying disease
This is generally thought to be the case!
■ Its generally more marked in the distal vs the proximal stomach
It's the other way around actually
■ Beta blockers don't help
Beta blockers do reduce gastric perfusion
■ The condition is irreversible
A reduction in portal pressure improves the condition
explanation
Congestion secondary to portal hypertension is thought to be the primary cause of Portal Hypertensive Gastropathy. However, other factors may also play a part such as mucosal protective mechanisms, inflammatory response, local vascular tone, hepatic function, gastric mucosal perfusion, endotoxin, and gastric sucrose permeability, have been suggested to influence the development of Portal Hypertensive Gastropathy. However, because Portal Hypertensive Gastropathy is a dynamic condition which improves after liver transplantation or after TIPS, suggests that it IS portal hypertension which is the primary driver.
There is a correlation between Portal Hypertensive Gastropathy and Child-Pugh stage, HVPG (hepatic vein pressure gradient), MELD score, albumin level, bilirubin, platelet count, INR and even survival [Bang CS. BMC Gastro 2016;16(93] You are right, there is also a small oesophageal varix visible!
This is an odd looking, almost 'nodular' antral gastritis. You may be surprised that this appearance is typical for a certain type of gastritis ...
WHICH TYPE OF GASTRITIS?
■ Lymphocytic gastritis
Mucosa is usually ulcerated in LyC gastritis
■ Eosinophilic gastritis
You are correct!
■ H.pylori associated gastritis
Can look weird but not classically nodular
■ Collagenous gastritis
Is endoscopically invisible!
■ CMV gastritis
Usually ulcerative
explanation
This nodular appearance is typical of an 'eosinophilic gastritis'. In textbooks you also find that lymphocytic gastritis can give rise to this appearance. However, in my experience LyC gastritis is usually intensely red with superficial linear ulceration. See my example below. CMV gastritis look similar but the ulcers are usually round rather than linear.
Anyway, finding more than 30 eosinophils per high power field (HPF) in at least five HPFs has been suggested as diagnostic. There is an argument that pathologists shouldn't make a diagnosis which doesn't change management. Not sure that I agree with this. After all, their job is to describe what they see! Nevertheless, it's probably prudent to ask about food or drug allergies, and consider if patient is atopic. A search for Helicobacters is probably also indicated. I would probably also consider taking further samples from the oesophagus as unrecognised and untreated eosinophilic oesophagitis can ultimately develop into a stricturing disease. Apart from this, it's difficult to know what to make of this diagnosis! Would you do a colonoscopy to chase after the possibility of asymptomatic IBD?
This small polyp was removed from the transverse colon. H&E histology is attached
WHAT IS THE DIAGNOSIS?
■ Serrated polyp
Crypt pattern looks more adenomatous!
■ Adenomatous polyp
Slit-like crypts but that thick capillary doesn't fit!
■ Leiomyoma
You're good at histology!
■ Neuroendocrine tumour
That was my guess too but I was wrong!
■ Small CRC
This lesion is submucosal!
explanation
I was quite sure that this was a small NET. It's that vessel crawling up it's side which convinced me. The reason that I enclose the histology is because it's not a neuroendocrine tumour! Histology shows interlacing bundles of spindle-shaped, smooth muscle cells. with bland-looking nuclei probably arising from the muscularis mucosa just below the epithelium.
It's a leiomyoma, which are very uncommon in the colon but of course very common in the oesophagus. Endoscopically, it's usually not difficult to tell the two apart. NET's often have a dip in the centre and/or sizeable vessels crawling up their sides. Colonic leiomyomas are usually covered with entirely normal looking epithelium. With immunohistochemistry it's easy to tell the two apart. A GIST (thought to arise from the 'interstitial cells of Cajar') usually stain with c-kit (CD117) while a leiomyoma is (thought to originate from smooth muscle cells) stain positive for smooth muscle actin or desmin, but not for c-kit. Of course leiomyomas are subepithelial lesions, covered with a normal mucosa. The differential diagnosis of submucosal swellings in the colon include:
This patient is attending for dilatation of a high oesophageal stricture but there is a surprise. WHAT IS THE CAUSE OF THE STRICTURING?
■ Corrosive ingestion
Yes, a likely cause of 2 high oesophageal strictures
■ Peptic ulceration
In the high oesophagus?!?
■ Lichen planus
Possible but never seen two !
■ Eosinophilic oesophagitis
They are not short and membrane-like!
■ SCC
The mucosa is unremarkable!!!
explanation
The 'surprise finding' is of course that there are TWO strictures some 2-3cm apart in the high oesophagus. All of the above could be the cause, I guess. Of course, the stricture does not look malignant. Eosinophilic oesophagitis gives smoother, long tapering strictures, not short membrane-like strictures like this. Peptic ulceration would be unlikely in the high oesophagus (unless there is a long stretch of Barrett's below). Lichen planus is a possibility but the fact that there are more than one strictures, makes corrosive ingestion the most likely diagnosis. Actually, the patient had been admitted after swallowing a lot of toilet cleaner a few months earlier.
in the images below you can see what her oesophagus looked like after that alkaline burn. I wonder if giving a reducing dose of steroid, like we do after large oesophageal EMR's could have prevented her stricture? This sessile polyp was found in the caecum, close to the appendiceal orrifice WHAT IS YOUR DIAGNOSIS?
■ Mixed adenomatous & serrated polyp
I don't see a mixture of crypt patterns!
■ SSL
Absolutely!
■ TSA
No, TSA's look like a cross between TVA & VA!
■ Adenomatous polyp
Perhaps the crypts look a little slit-like but it's covered with mucus...
explanation
Well, perhaps the crypt openings look a little slit-like but the lesion is partially covered with that mucus typical of an SSL. By the way, couldn't it be a hyperplastic polyp? I think that pathologists have given up even attempting to tell the two apart! My own rule of thumb is that anything which is ≥10mm, I call an SSL and remove. By the way, SSL's usually lift very well. In this particular case, the lift is a little sub-standard for an SSL, presumably because it's situated very close to the appendix orifice which anchors it down. A word of caution! I've had several 'post polypectomy syndrome' cases after removing large SSL's. In these cases, the lift was excellent and I asked for the LARGE snare to remove the lesion en-bloc. However, in both cases, I found that the snare was taking quite a long time to cut through. Perhaps because of that fatty reaction in the stroma below which Neil Shepherd talked about in the Podcast. To avoid any risk of the 'post polypectomy syndrome', you should ask your assistant to close the snare as quickly and hard as possible. Don't worry about bleeding. These lesions are never supplied by any significant vessels. Oh yes! And place clips !!!
This patient has recently undergone a bone marrow transplant when she develops abdominal pain. PPI therapy has not improved her symptoms and she referred for a Gastroscopy
WHAT IS YOUR ENDOSCOPIC DIAGNOSIS?
■ CMV infection
A good guess although there are no ulcers?!
■ HSV infection
Never seen it! Does it really happen?
■ C.diff infection
Definitely doesn't happen in the stomach!
■ Acute GVHD
Actually, you are not wrong!
■ Checkpoint inhibitor gastritis
Not as crazy as you think!
explanation
Well this is a striking appearance! I'd never seen anything like it and tentatively suggested HSV gastritis on the histology request form. I didn't consider CMV because there was no mucosal ulceration.
Histology confirmed finding lots of oedematous CMV infected cells with granulation tissue and there was no need for immunostaining for cytomegalovirus proteins. This is a case of CMV gastritis! Actually, there were also some features suggestive of acute GVHD and I can therefore not be entirely sure that these 'plaque-like appearance on the mucosa isn't due to the combined effect of CMV+GVHD+PPI! It's not uncommon, one study, reported that approximately one-quarter of patients who were unresponsive to first-line acute GVHD treatment, actually had coinfection with CMV [Bone Marrow Transplant. 47, 694–699]. Of course, immunosuppressants is the trigger for CMV reactivation but CMV gastritis can occur in apparently immunocompetent patients, although a link with occult cancer has been suggested [Scand J Gastroenterol 2011;46:1228–35]. My guess of HSV was of course unlikely as HSV infects squamous mucosa or the colon (surprisingly), not the stomach. In the oesophagus you may see vesicles and/or small ulcers (left when the vesicles rupture). If I had seen this appearance in the colon I would have called this 'pseudomembranous colitis but of course C.diff doesn't affect the stomach. Can you get gastritis with checkpoint inhibitors? Yes, you can! It can cause a severe, haemorrhagic gastritis as outlined in this article [Case Rep Oncol 2018;11(2):549–556]. There is more information on CMV gastritis at this site. These polyps were found in the stomach of a 40 year old man on a Barrett's surveillance programme WHAT WOULD YOU ADVICE?
■ We should eradicate your Hp
This patient is VERY unlikely to have H.pylori
■ We should remove these endoscopically
An unnecessary job unless one look wonky, like perhaps that single red one!
■ We should stop you PPI
But the patient has Barrett's ?!
■ We should stop you PPI
That's what I would do !
explanation
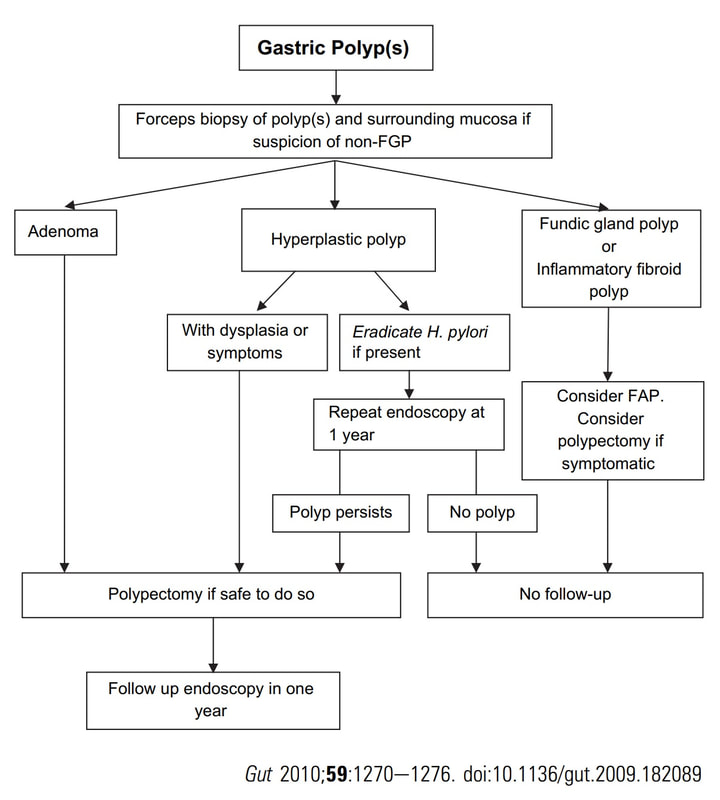
Of course, this is a Fundic Gland Polyps/Cystic Gland Polyps . They are usually multiple, somewhat transparent sessile polyps, usually in the 1-5mm in diameter and located in the body and fundus.
Histologically there are cystically dilated glands lined by gastric body type mucosa. Of course, there is no need to sample these. A study patients with FGPs reported that they can be diagnosed with a high degree of accuracy based on endoscopic appearance alone (J Clin Gastroenterol 2003;36:399-402). The sporadic polyps are caused by activating mutations of the beta-catenin gene. Not sure how an APC gene mutation (you may remember are involved in FAP), could give rise to FGP’s though! Furthermore, I don't know how the use of PPI's can be linked to the development of FGP's. Presumably its something to do with the prolonged hypergastrinaemia and enterochromaffin cell-like (ECL) hyperplasia? I have seen a couple of cases of cancer developing within a FGP. In both cases the patient had FAP and the polyps were markedly larger than all the surrounding cystic fundic polyps. There have been case reports of dysplasia arising within sporadic fundic gland polyps. A few series have reported a <1% risk of sporadic FGPs harbouring dysplasia (Am J Surg Pathol 1998;22:293-298, Eur J Gastroenterol Hepatol 2003;15:1153-6, Endoscopy 1995;27;32-37). I must admit that I don't believe the figure of <1% risk of dysplasia in a sporadic FGP. I suspect that these series consist of rather selected cases. Of course by only selecting polyps 10mm or larger, you would be filtering away 99% of the FGP's, leaving only the 'far above average risk lesions' behind. Nevertheless, the BSG recommend sampling some of them to confirm the diagnosis but not to set out on any attempt to remove them. Of course, this advice is different to the advice for Hyperplastic polyps which ARE usually linked with H.pylori and also a greater risk of developing cancer. The BSG advice is summarised in the algorithm below. Sporadic FGPs are not associated with either H.pylori gastritis or atrophic gastritis. In fact, it has been proposed that H. pylori infection may have an inhibitory effect on the development of FGPs. Of course, you wouldn't stop the PPI in a patient with Barrett's. Actually there is no evidence that patients with unstable Barrett's are any more or less likely to progress to cancer if they stop their PPI. However, there must be a point (I presume) when the use of PPI is beneficial in the 'evolution' of Barrett's. Are patients on a PPI less likely to develop Barrett's or to develop damage the stem cell DNA? Not sure! The BSG do recommend a colonoscopy in patients below the age of 40 yrs. to rule out the possiblity of FAP. However, as the average age of colorectal cancer in patients with 'attenuated FAP' is 55 yrs, I would personally recommend a colonoscopy in patients above the age of 40 yrs. This 80 yr old man was first referred for a CT to investigate his PR bleeding and mucus. The CT reported that the rectum appeared thickened and a Flex Sig is organised WHAT IS YOUR DIAGNOSIS?
■ Active proctitis with inflammatory polyp
That was my diagnosis and I was half correct!
■ Active proctitis with adenomatous polyp
Very good call!!! Better than mine if fact!
■ Circumferential polyp with dominant nodule
Apologies for the indigo carmine dye which has confused you!
■ Circumferential polyp with malignant change
The blue dye is confusing and the proctitis does look weird in places
explanation
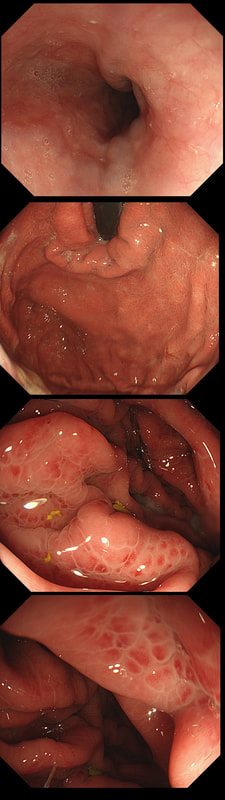
Yes, it does seem odd to refer for a CT rather than a Flex Sig? Perhaps this was because the patient was 80 yrs old or because of our endoscopy waiting list. Anyway, I was half correct. I diagnosed an active colitis and histology confirmed a: transmucosal inflammation with crypt abscess and focal epithelial loss (tiny, tiny ulcers). We all know that active colitis is characterised by an dull erythema, bleeding and erosions/ulcers. The best endoscopic scoring system is currently the Ulcerative Colitis Endoscopic Index of Severity (UCEIS) which scores these three parameters on a scale of 0 to 3. There are other scoring systems of course such as the Mayo score, the Baron score, The Rachmilewitz Endoscopic Index, The Sutherland Index, The Matts Score, The Blackstone Index and the most recent addission, The Ulcerative Colitis Colonoscopic Index of Severity! Of course this tells you than none of them is the last word on the topic. For a good review of all endoscopic issues relating to colitis, please check this article out; Annese V. European evidence based consensus for endoscopy in inflammatory bowel disease, J Crohn's Colitis 2013;7(12);982-1018 Anyway, I find it odd that other common features of an active colitis are not included in any of these scoring systems. This includes mucosal oedema, a villous epithelial surface (as seen in this video) and a mucopurulent discharge (which I regard as feature of a severe attack of colitis). These three features are prominent in this video and if not recognised as features of an active colitis, can be mistaken for a neoplastic process. I thought that the nodule would be inflammatory but it turned out to be a 'TSA' (traditional serrated adenoma), the most rare and elusive of all colorectal polyps. Well I blame the inflammation and that thin mucopurulent layer obscuring the crypt pattern. This polyp just distally to the caecum has been referred after an initial set of samples have indicated that it's a benign TVA harbouring no more than LGD. An unfortunate, fixed loop at the hepatic flexure means that I can't retrovert behind this polyp. In fact, it's difficult to get much closer to the lesion than this. WHAT STATEMENT DO YOU AGREE WITH?
■ The resection may be difficult but it can be done
I agree with that!
■ The location of the lesion and difficult colonic configuration probably makes a resection impossible
Of course the patient may have to travel to a nearby centre with the neccessary expertise!
■ All this is irrelevant as the polyp looks suspicious and would be best removed surgically
I disagree, it does look benign! But will of course have a 15% risk of harbouring cancer...
explanation
Actually, the lesion does look very benign. It wobbles about freely perched on top of the first fold distally to the caecum. Furthermore, it has a reassuring gyrate crypt pattern of a TVA. Naturally, any polyp of this size will have a 15% risk of actually containing an unexpected focus of cancer within. Because our therapeutic waiting list is in mess in the aftermath of Covid, I do encourage everyone to sample the sessile lesion even though you are planning to refer it for resection. Such sampling on the surface of large sessile lesions doesn't induce any fibrosis and provides some (limited of course) reassurance that the lesion isn't malignant. In this case, the position is very challenging but you can get close enough to stand a good chance of removing the lesion. That's actually what I did and I've just uploaded a video of the resection together with my face-to-camera annotation to highlight the main points. Ultimately, histology confirmed a TVA harbouring no more than LGD This patient is undergoing a set of oesophageal dilatations of a radiotherapy induced stricture. Three set of samples, a total of 18 biopsies have been obtained from the stricture which have all been reassuring. A CT has reported: "Distal oesophageal mural thickening extends throughout the length of the previously demonstrated bulky oesophageal tumour. No focal abnormality to suggest recurrent disease and presumably appearances are all radiotherapy related". In the video clip, the stricture is seen to be dilated to 20mm. WHAT WOULD YOU DO NEXT
■ Organise the next dilatation in a weeks time
She WILL need more dilatations but you've missed something important!
■ Take another set of samples from that stricture
It does look concerning still but there is something even more concerning here!
■ Take another set of samples from the more proximal oesophagus
You've spotted it!
■ Take samples at the distal oesophagus (below the stricture)
That odd, fleshy lump at 6 O'clock below the stricture has been sampled already. More samples is never wrong but there is something even more important to sample first!
explanation
On intubation, I glimpsed something just below the upper oesophageal sphincter. I deliberately didn't dwell on it in the video, simulating the previous intubations when a synchronous lesion had been missed. In the explanatory clip below, you get a better view of her second SCC. CT staged the second lesion as no worse than T2, N0.
The moral of the story? It's when we are distracted that we run the risk of missing things! In this case, the previous endoscopists had approached the endoscopy as a case of dilatation and switched off their eyes! |
Categories
All
|