|
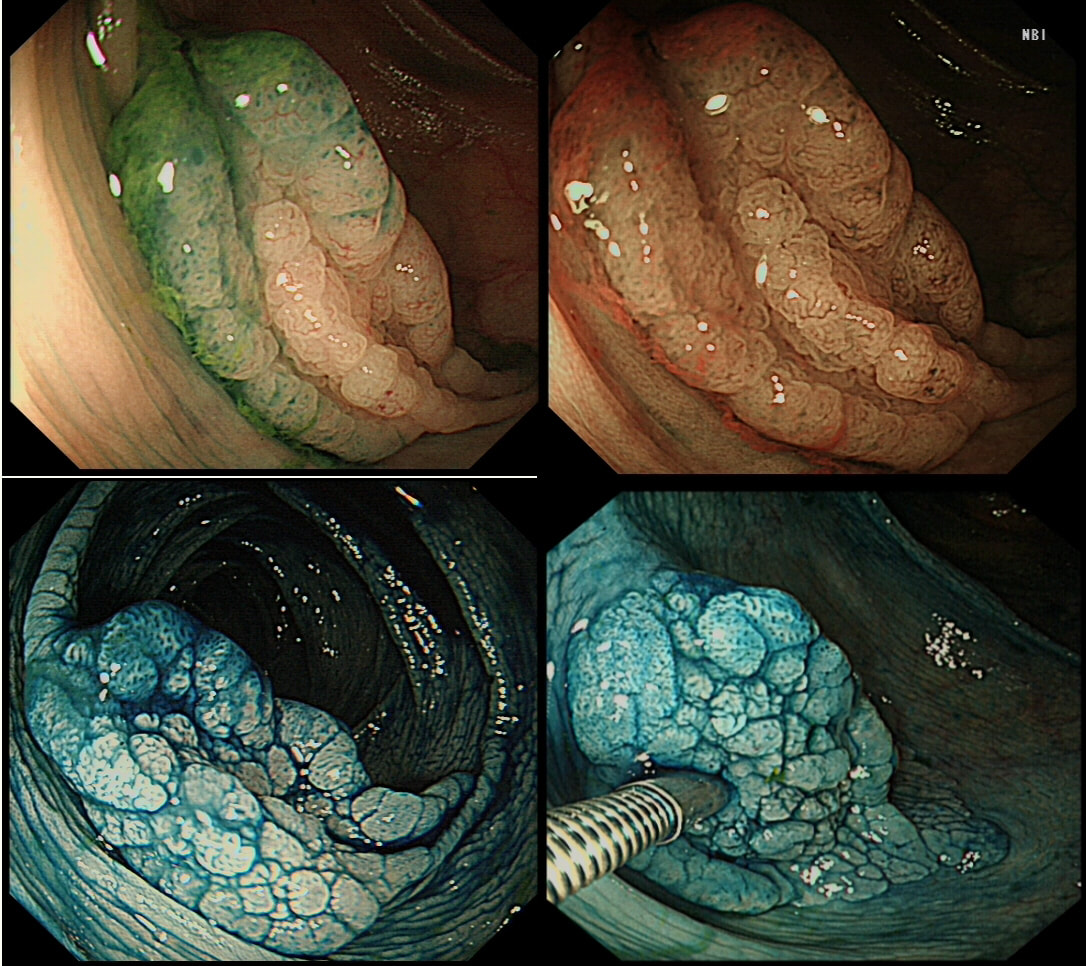
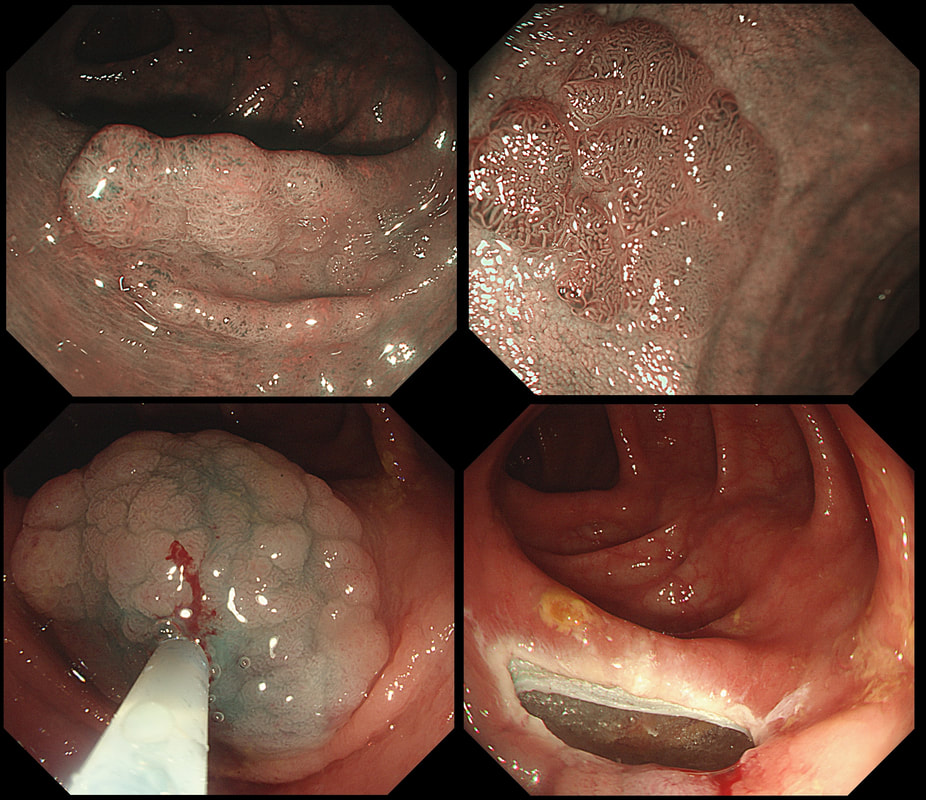
This lesion was found in the ascending colon and was subsequently referred for endoscopic resection.
WHAT IS YOUR ENDOSCOPIC DIAGNOSIS?
SSL
CORRECT!
TUBULAR ADENOMA
INCORRECT!
TUBULO-VILLOUS ADENOMA
INCORRECT!
VILLOUS ADENOMA
INCORRECT!
EXPLANATION
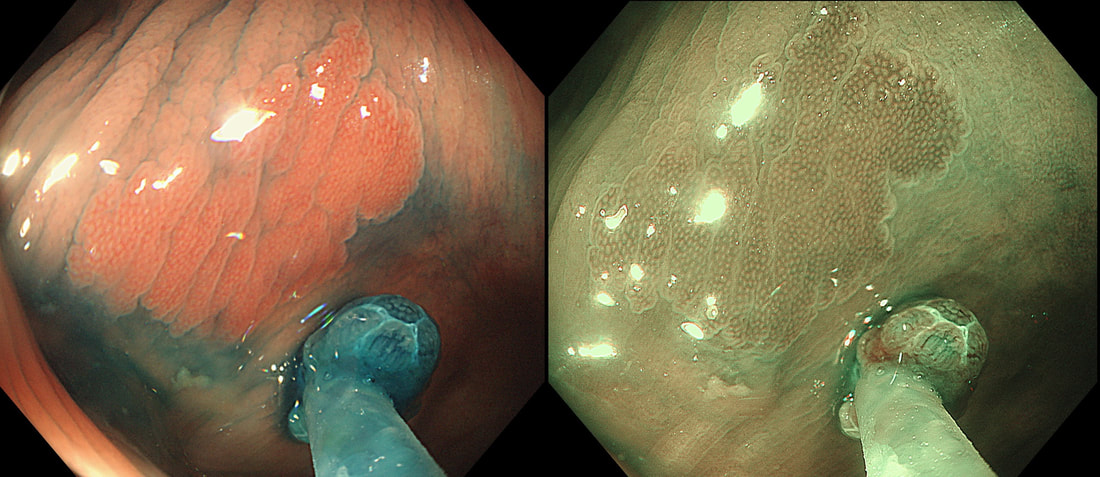
In the indigo carmine dye images, you can see some large, round/oval crypt openings. This was a Sessile Serrated Lesion which actually harboured HGD. 75% of our Facebook group members got the diagnosis right.
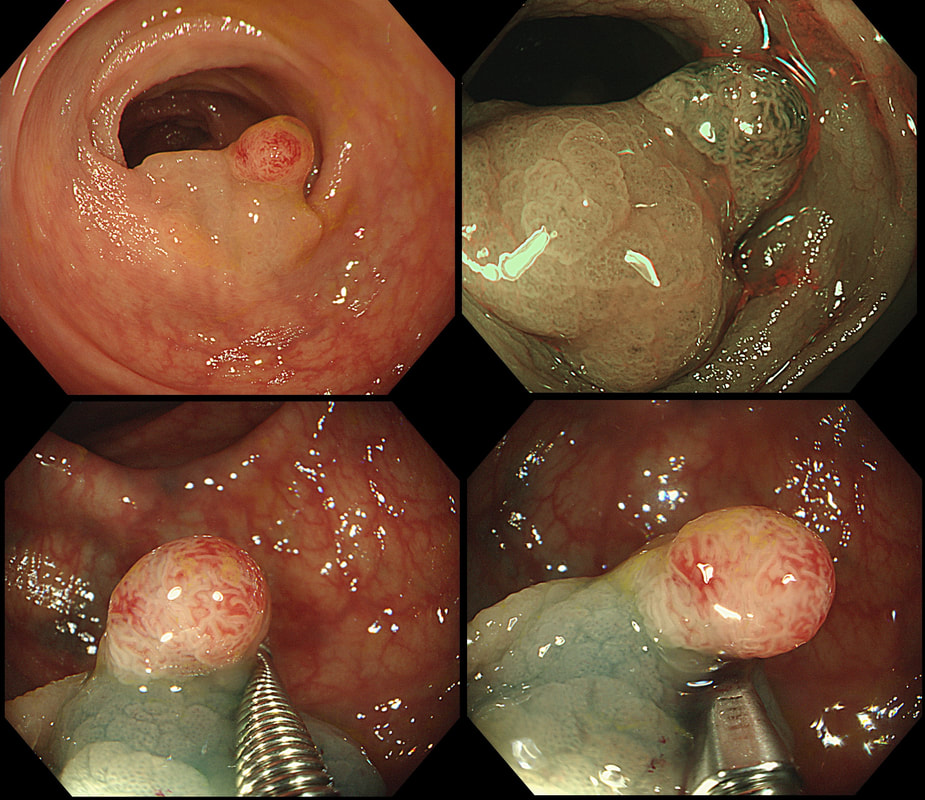
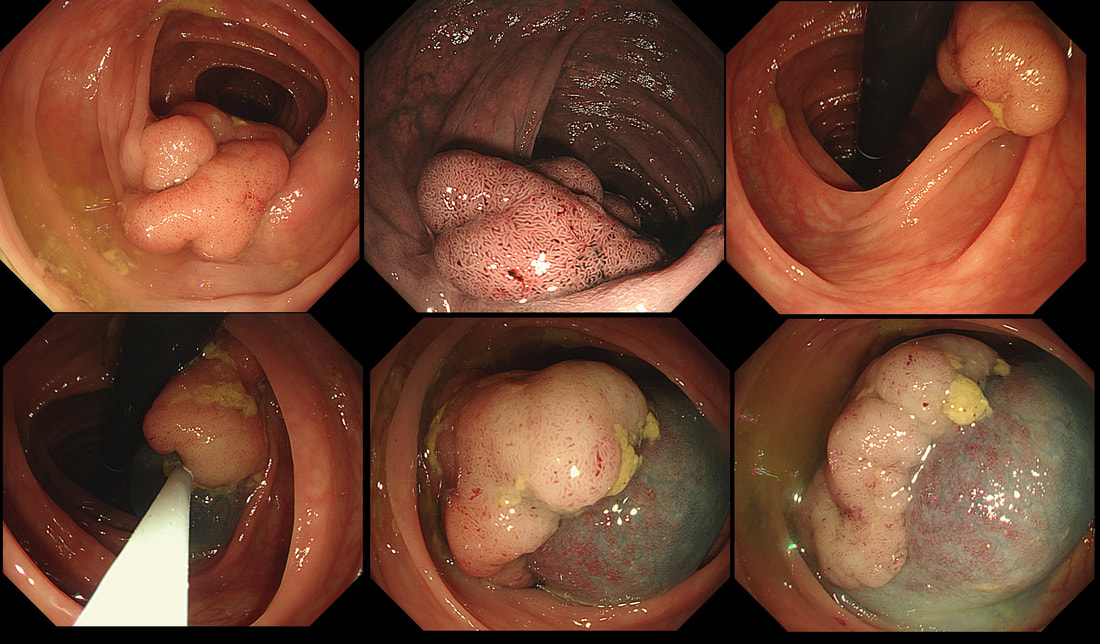
The confusing thing is that the growth pattern is that of a LST-G, which of course means that it should be a TVA! I wonder if there is any way I could have predicted the dysplasia? The fact that the lesion is rather "chunky" (but still soft) and not thin and flat, is the sign dysplasia within an SSL . The image caused some controversy back in 2014. Several endoscopists didn't believe that an endoscopic diagnosis could be made. In fact, some hinted that it would be inappropriate to second guess histology. After all, these are the guys with all the answers right? You should stand 'cap in hand' and wait patiently until they tell us what we have seen. It makes my blood boil ! Fortunately, endoscopy has moved on from those dark times. Now we recognise that we have a duty as endoscopists to contribute to the diagnosis. The diagnosis is based on an assessment of the entire lesion and it's degree of lifting. In contrast, our poor pathologists can only give an opinion on the square mm of tissue which we provide them with. That is why they need microscopes! By recognising our responsibility, we accept a duty to develop expertise. Of course, this directly benefits patients as we are more likely than our pathologist to recognise an early malignant polyp and thus target samples to the area which is the most suspicious in appearance. Our pathologists also benefit from our diagnosis, although some endoscopists believe that we shouldn't tell them anything as it could bias their assessment ☺ . To conclude, the combined endoscopic and histological assessment is what is most likely to reach the correct diagnosis - a Team Effort !!! I was asked to remove this sigmoid lesion. Previous samples have revealed a TA+HGD. The dark colouration is due to a previous ink tattoo, placed rather too close to the lesion itself for my liking. SHOULD I ABORT OR ATTACK?
ABORT!
Correct!
ATTACK!
WRONG!
EXPLANATION
When this question was first published on our FB group, >95% correctly answered; Abort ! The complete absence of a crypt pattern means that this lesion should not be endoscopically resectable. The Japanese would say that there is evidence of 'massive invasion' (invasion beyond 2mm below the muscularis mucosa). Of course, I confirmed my impression with a 'test-lift', which of course failed. Subsequently, surgery confirmed that the lesion was T3 (invading through the muscle propria layer) but luckily no nodes were involved. The take-home message? If a 'test lift' had been done a the first examination, we could have saved a lot of time. A test-lift will not scupper a future endoscopic resection!
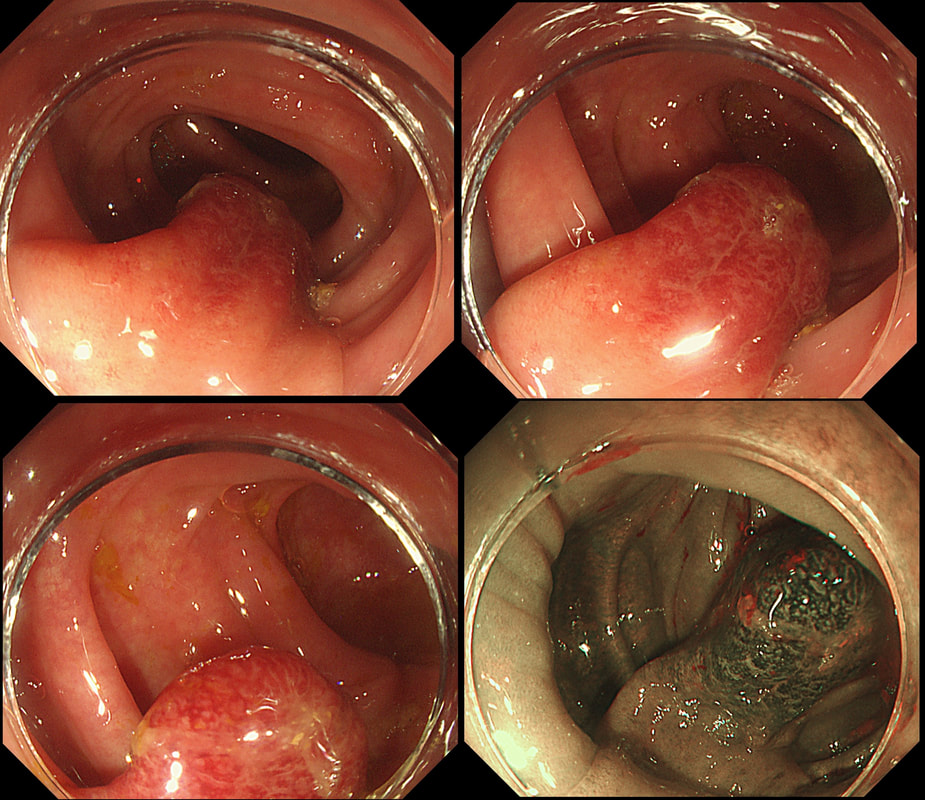
This polyp was found in a 30 year old patient with a family history of bowel cancer undergoing colonic surveillance because of a 'polyp syndrome'.
WHAT IS THE LIKELY POLYP SYNDROME?
a) Familial Adenomatous Polyposis (FAP)
INCORRECT!
b) Peutz-Jeghers syndrome
INCORRECT!
c) MUTYH associated polyposis (MAP)
INCORRECT!
d) Serrated Polyposis Syndrome)
CORRECT!
e) Lynch Syndrome (formerly HNPCC)
INCORRECT!
explanation
This proved to be a difficult question and only 15% of our facebook group got the correct answer of: Serrated Polyposis Syndrome !!!
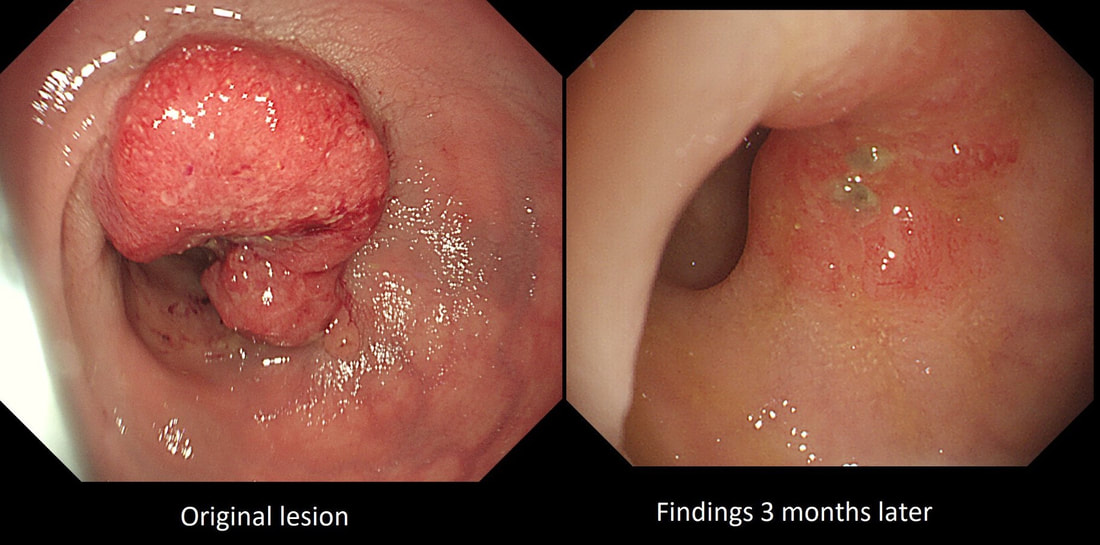
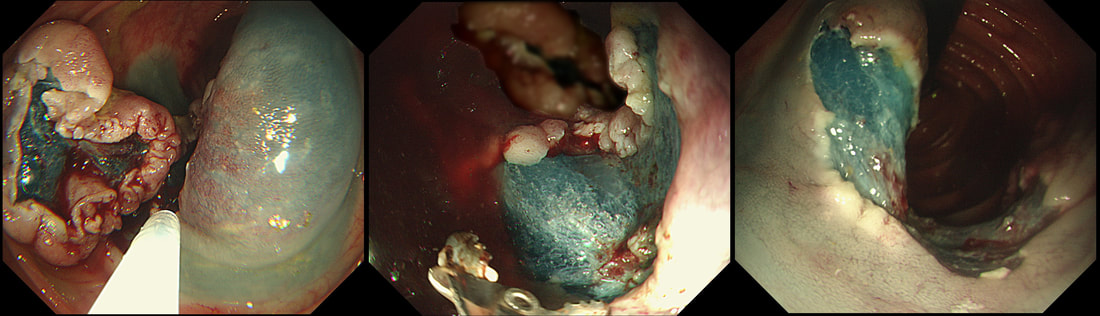
The odd thing about this polyp is that it appears to be a tubular adenoma (slit-like crypts) arising from a broad somewhat odd looking fold. The 'fold' is actually a larger hyperplastic polyp. Of course large hyperplastic polyps are now referred to as 'sessile serrated lesions' (SSL's). You are looking at an SSL, sprouting a TA !? A 'purist' histopathologist may refer to this entity as a 'mixed serrated, adenomatous polyp'. Do you know of a polyp syndrome where patients have a mixture of SSL's and adenomatous polyps? Of course, the serrated polyposis syndrome 'SPS' (previously called the sessile serrated polyposis syndrome). It's now realised that many patients with serrated polyposis syndrome actually have a mixture of serrated and adenomatous polyps. It's possible that these patients have a greater risk of developing cancer. The correct answer is therefore 'A' ! The BSG has published a 'position statement' on the topic of SSL's which is well worth reading. The BSG sensibly recommend annual surveillance until all serrated polyps above 5mm have been cleared, following which the surveillance interval may be reduced. This is because patients with SPS do not seem to have a higher risk of developing cancer than patients with Lynch syndrome. This high rectal lesion was treated with chemoradiotherapy 3 months ago (left image). On re-assessment of the site, there is some erythema remaining. WHAT IS THE LIKELY NEXT STEP? a) APC ablation b) EMR c) ESD d) Further radiotherapy e) Surgery explanation
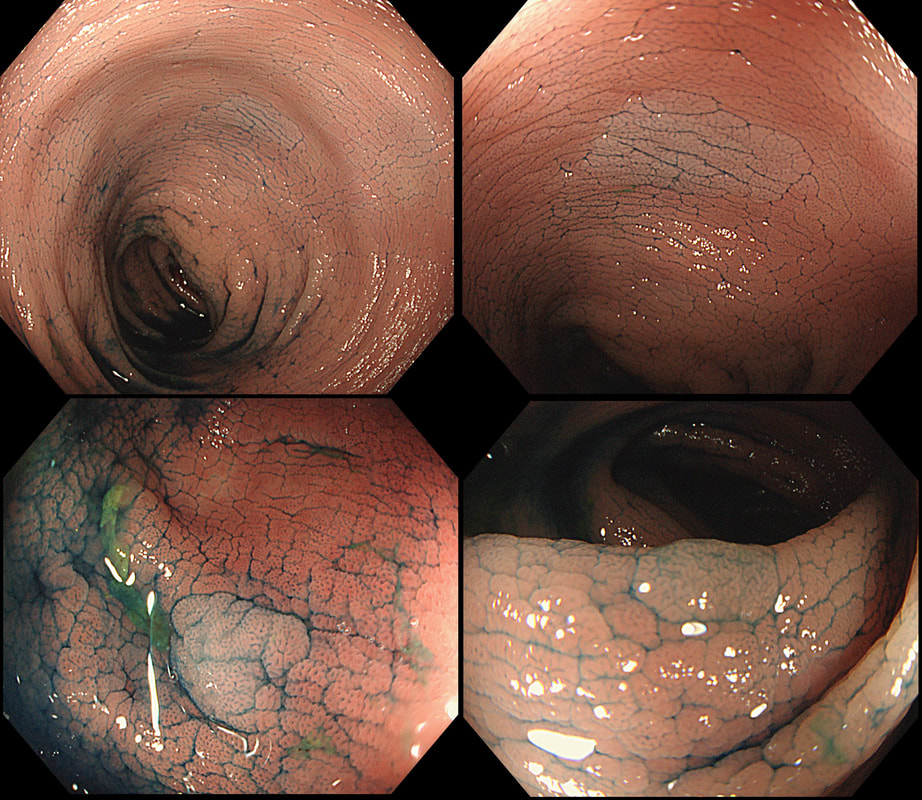
The initial staging of this rectal cancer was T2, N0. It took many years for Prof Angelita Habr-Gama (a famous colorectal surgeon from Sao Paulo, Brazil) to convince her fellow surgeons that neo-adjuvant chemoradiation for stage 0 rectal cancer could be as curative as surgery but without the need for a subsequent AP resection and a stoma. She produced a landmark paper; “Operative versus nonoperative treatment for stage 0 distal rectal cancer following chemoradiation therapy: long-term results,” [Ann Surg 2004;240(4):711-7] which proved the case. This particular patient had received chemoradiotherapy and the result has been good but NOT complete. Samples taken from that red area confirmed residual cancer. As it happens, the patient was not a surgical candidate and a brachytherapy boost was instead given. This was of course something of a trick question as both D and E are 'correct' next steps. This is the colon of a patient with long-standing colitis and PSC on annual surveillance. These were found after indigo carmine dye spray. WHAT IS YOUR ENDOSCOPIC DIAGNOSIS? a) patches of crypt distortion b) serrated lesions c) a flat adenomas Explanation
Actually, these are patches of 'crypt distortion'. Often seen after pan-colonic dye spray and VERY difficult to distinguish from serrated lesions. The crypt openings are a little larger than normal and strangely these seem to develop in patches which can be very large. In a case like this, I would take samples to reassure and alert subsequent surveillance colonoscopists to the presence of these odd lesions. In my experience they are usually seen in long standing colitis which has been troublesome in the past and I wouldn't be entirely surprised if future research find that these are more commonly found in patients who go on and develop flat dysplasia in the future. Below is a close-up of a lesion.
This lesion was found in the descending colon. It's lifted with our standard EMR solution (volpex, adrenaline and indigo carmine dye). HOW WOULD YOU NOW BEST APPROACH THE RESECTION? a) the lift is poor, the lesion appears tethered and I would refer for surgery b) this is a benign polyp and a piecemeal resection using a 15mm snare would be best c) the polyp may be malignant and a large, floppy snare would be best to remove the lesion in as few pieces as possible d) preferable for 'single fragment resection' but a stiff, large snare would be preferable explanation
The lifting is a little 'lob sided' but that is common and not necessarily a 'showstopper'. A polyp of around 3-4cm would have a 1:7 risk or so of harbouring cancer. For this reason, removing the lesion either single fragment or in as few pieces as possible, would be preferable.
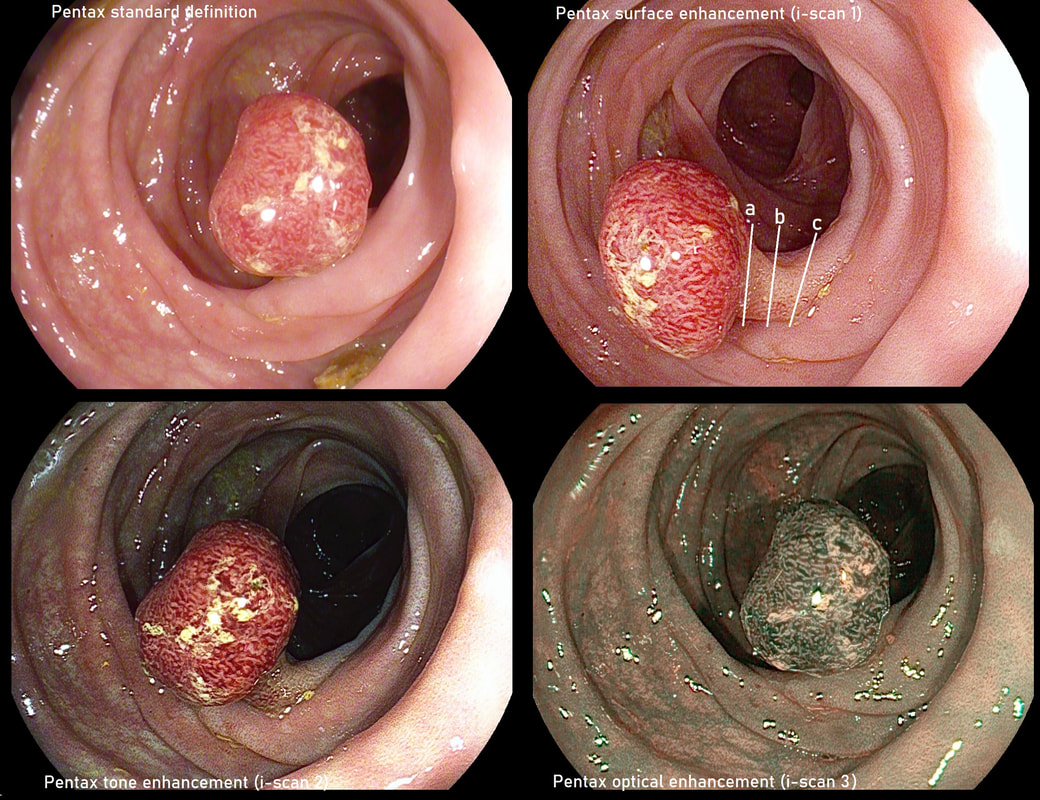
It's worth highlighting that when you have a sessile polyp like this, the surface crypt pattern may not accurately reflect what is happening deeper inside the polyp. So how could this polyp be removed in as few pieces as possible? Of course and ESD is the obvious answer. But it's not possible or appropriate to offer this to every patient. Another possibility would be to do a 'pre-cut' EMR, when you cut a grove around the polyp before resecting it. An 'underwater resection' would be a third possibility However, the simplest way to remove this as a central main fragment and then some small side-fragments would be with a stiff snare. A stiff snare allows you to push down hard on the polyp and get it off in as few pieces as possible. Of course there is a far higher than average risk of perforating with a stiff, large snare. Use it with caution and be prepared for trouble. A pre-cut around the polyp could also help in achieving a single fragment resection. Ultimately, this lesion was resected as a main central fragment and several small side-fragments (image below). The ultimate histology was of a tubulovillous adenoma (TVA) harbouring low grade dysplasia (LGD). A small colonic TA+LGD as visualised by Pentax's 4 different optical modes. WHAT SIZE SNARE TO USE? a) 10mm b) 15mm c) 20mm d) 25mm WHERE TO PLACE THE SNARE (top right image)? a) at 'a' b) at 'b' c) at 'c' explanation
Even with a small polypectomy such as this there are things to discuss. FIRST, select a snare which fits nicely around the polyp. In this case, a 10mm snare is probably optimal. A snare which fits snuggly, is less likely to capture a fold of mucosa somewhere behind the polyp. In this case, you could probably see if this is happening but particularly in an angulated and spastic sigmoid, capturing too much mucosa can be an issue. Of course, if I have already used a larger snare elsewhere in the colon, I would consider using that snare again. I do try to keep costs down if possible 😳 ! SECOND, place the snare in the middle of the stalk (at b). Giving a small amount of stalk for the pathologists to confirm that the polyp has been completely removed will avoid a subsequent 'site-check'. Conversely, placing the snare too close to the base may cause two problems: a) more heat damage is imparted onto the nearby colonic wall and b) you no longer have a stump to re-capture if there is bleeding. Which leads me on to the THIRD point; 'haemostasis'. Bleeding is the main concern for us all when we do polypectomy, EMR and even ESD. My approach is as follows: ■ Anticipate bleeding! It's more likely with large polyps or polyps with thick stalks. Consider what you will do if there is bleeding. For a stalked polyp, the best option is to re-snare the polyp. For a large sessile polyp, a haemostatic forceps is usually the best bet. Ask your assistant to have the forceps immediately available (still in it's sterile packaging). By the way, I usually use a 'hot biopsy forceps' because they cost £10 compared to the £200 for a fancy haemostatic forceps. ■ Prevention. Most admit that I never place a clip on the stalk before cutting it. This is because the current can easily travel down the clips tail and cause a burn at some unexpected place. However, I often pre-inject a thick stalks and large pedunculated polyps with dilute adrenaline. It will buy me a valuable minute of 'gentle oozing' from the vessel before the floodgates open. At ESD, you will of course coagulate the vessels as they appear in your dissection field. A large sessile 4cm polyp can be removed by single fragment EMR (there are snares large enough) but you will of course have to deal with several bleeding vessels once the polyp has come off. Of course, at ESD you can deal with the vessels one-by-one and this may be preferable than going for the 'quick option' of an EMR. Under the heading "Prevention", I should also mention that I advice my assistant on how many seconds I think it will take to cut the polyp. I also tell them that once they see the stalk being cut by the snare, they should ease off and stop closing the handle. The 'inertia' in the system will cut the rest of the polyp at the appropriate speed. After all, the vessels are in the CENTRE of the stalk and it's the last few seconds of cutting which deals with them! You don't want to rush this bit!!! For this reason, I am not a fan of 'pre-coagulating' the stalk by pressing on the blue pedal first. This cooks the harmless outside of the stalk but leaves the important centre intact. If anything one should do the opposite! Actually, with modern diathermy you don't need to concern yourself as the clever machines deliver mainly 'cut' at the beginning (when the resistance in the electrical circuit is greater) and then moves to short pulses of 'coagulation' and 'pause' (to allow tissues to cool down) as the electrical resistance drops as the stalk is cut. ■ Clipping. This deserves it's own 'heading'. I almost always place clips unless it's cold snaring a small polyp in a patient who is not taking blood thinners. Of course, I work in a part of the world when I don't need to worry about whether the patient can actually afford the clips 🙂 . When you feel exhausted after a 60 minute resection, it's easy to leave the clips out. It would be a mistake! Clipping reduces the risk of both late bleeding, the post-polypectomy syndrome and late perforations! This polyp was found in the transverse colon in a patient with long-standing ulcerative colitis. WHAT WOULD YOU DO NEXT? a) retrieve the polyp and look for other polyps b) obtain 4 samples from the nearby mucosa c) apply APC to the EMR edges d) tidy the edges up with a cold snare e) apply clips Explanation
That hole isn't right! More often than not, this is how a perforation looks immediately after polypectomy. As a beginner you may think that you should be able to see the peritoneum but that would be the exception. Must admit that I was surprised to see this perforation. The lift seemed to be good and I didn't use a 'super-stiff' snare. Perhaps it had something to do with the fact that the patient had colitis? Because the 'cutting' should have taken 6-7 seconds but took about 12 seconds, I did expect 'trouble' and had started to suck air out of the colon before the snare cut through completely. Fortunately, the colonic lumen is not deflating as the perforation is close to the omental reflection. We will have a few minutes before the patient starts to become uncomfortable. Spend those minutes well !!! I applied 10 clips. The colon was clean and I was confident that there was no peritoneal contamination. Of course, this is one of the reasons why your polypectomy site should be as dry and clean as possible before you step on that yellow pedal! There was no pain and after a couple of hours in recovery and I discharged the patient with verbal and written instructions to return to A&E if pain developed later. The patient was fine. Can I reassure you that every patient will be fine after closing a perforation by clips? Of course not! You need to follow your instincts and your local protocols and of course be able to defend your decisions. Naturally, the particulars of the actual patient is very important. For example, an elderly patient or someone living alone should probably not be discharged. After all, sepsis can develop quickly and the patient may soon be in no fit shape to call for help. This polyp was found in the sigmoid colon. WHAT IS THE LIKELY AETIOLOGY OF THE POLYP? a) hyperplastic polyp b) adenomatous polyp c) malignant polyp Explanation
The sigmoid colon is the most 'powerful' part of the colon developing the force needed to go to the toilet. Presumably this is the reason why diverticular disease first develop in the sigmoid. The force can also create pseudo-polyps from patches of inflammation which I presume gets tugged along with each peristaltic wave. Even adenomatous polyps which develop here usually become traumatised. The end result is that this is the most difficult part of the colon to assess polyps. This particular lesion had been unchanged for several years but it spooked an endoscopist sufficiently to refer it for an EMR. Samples had indicated that it was a hyperplastic polyp only (correct answer was A). Fifteen years ago, the idea of subjecting the patient to an endoscopic resection for a lesion not recognised to be linked with cancer could have been criticised (if there was a complication). However, nowadays it is recognised that SSL's (hyperplastic polyps with L or T-shaped crypts etc) are 'fair game'. The truth is that it's probably impossible to tell a 'simple' hyperplastic polyp apart from a 'sessile serrated lesion' and pathologists are increasingly called all these 'serrated lesions'. For this reason, I go by the size. If the 'serrated lesion' is 10mm or larger, I usually remove it. In this particular 'polyp', most is actually just a swollen fold. The serrated area is situated at the very tip of the fold. When I see this, I don't go overboard placing the snare a long way down the 'pseudo-stalk'. If you did, you will find that it's taking a long time to cut through all that healthy sigmoid mucosal fold and you run the risk of a late perforation. Instead, I just caught the tip of the fold and ask my assistant to close the snare as quickly as possible whilst it was cut on a blend current (yellow pedal). Of course, you don't need to worry about large vessels in serrated polyps. This patient was brought down from the haematology ward with diarrhoea. The patient was immunosuppressed following a donor bone marrow transplant for myeloma. WHAT IS THE LIKELY DIAGNOSIS? a) Graft versus host disease b) An opportunistic infective colitis c) Checkpoint inhibitor colitis explanation
This is a case of acute Graft-versus-host disease (GVHD). Checkpoint inhibitor colitis gives a severe looking colitis whilst superficial ulceration is what should make you consider an opportunistic infective colitis. In the case of GVHD, there is usually very little to see. I knew that GVHD was due to T cells from the stem-cell donor which attack tissues in the recipient. However, I hadn't realised that it's the main cause of death after a stem cell transplant! This is bad news! The most common tissues affected by acute GVHD are the skin (widespread rash even appearing on the palms of the hands), liver (transaminitis) and the gastrointestinal tract. The horrendous secretory diarrhoea, classically developing in the weeks following a stem cell transplantation, is the most severe complication and linked with the 40% mortality rate of acute GVHD. There are a few take-home messages for endoscopists. ■ First, is that the mucosa may appear entirely normal but that samples nevertheless have to be taken as the diagnosis is clinical (of course) and histological (apoptosis of epithelial cells with loss of crypts). ■Secondly, endoscopic appearances are variable ranging from oedema (as in my video) to small intramucosal haemorrhages to erosions and ulceration. There is something called the 'Freiburg Classification' which basically orders this into a 4-level scale: 1 Normal mucosa 2 Patchy erythema 3 Aphthous ulcers and/or focal erosions 4 Confluent ulceration ■ Thirdly, 10 biopsies from the distal colon have the greatest chance of yielding the diagnosis. I take 3-4 biopsies from the rectum and sigmoid and if possible also the descending colon. ■ Fourth, because of the horrendous diarrhoea, there is no need to subject the poor patient to a phosphate enema before the flexible sigmoidoscopy. ■ Fifth, these patients often have low platelet counts and deranged clotting. Personally, I place an 11mm clip on each biopsy site to avoid the need for platelet transfusions and the need to correct the INR. However, this is a personal preference and for example the ASGE recommend bringing the platelet count up to above >50 × 109/L before biopsy (>20 for an endoscopy without biopsies). ■ Sixth, consider the possibility of superimposed infection, particularly if there is superficial ulceration as both CMV and HSV causes ulceration. Biopsies for viral cultures have to be placed in viral transport medium. Of course, the ward team should also have sent stool for viral PCR (adenovirus, astrovirus, rotavirus, noro-virus can all cause watery diarrhoea) as well as for bacterial pathogens such as C.diff and C. septicum as well as for parasites such as Giardia and Cryptosporidia Recently, I've had five referrals for GVHD. All patients were on high dose steroids (eg. 80mg of prednisolone) and in none of them did it seem to make any difference to their symptoms! All of the patients felt awful with abdominal pain, profuse diarrhoea and swollen limbs (from hypoalbuminaemia). Sadly, 2 out of 5 died within weeks of their flexible sigmoidoscopy. |
Categories
All
|