|
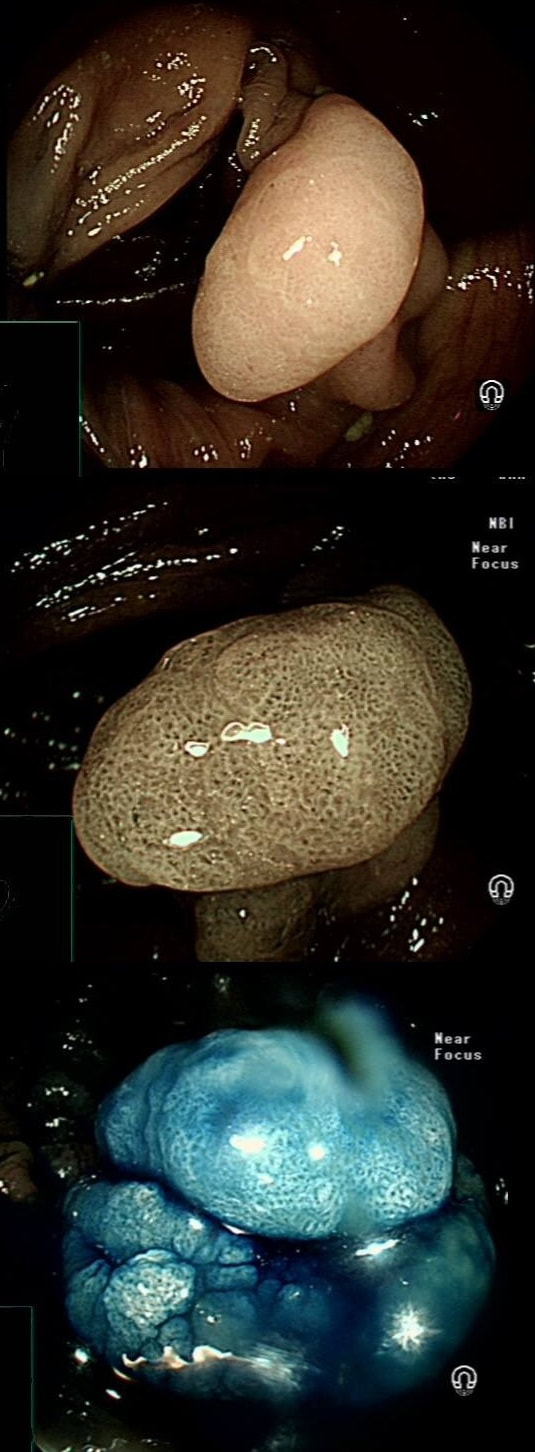
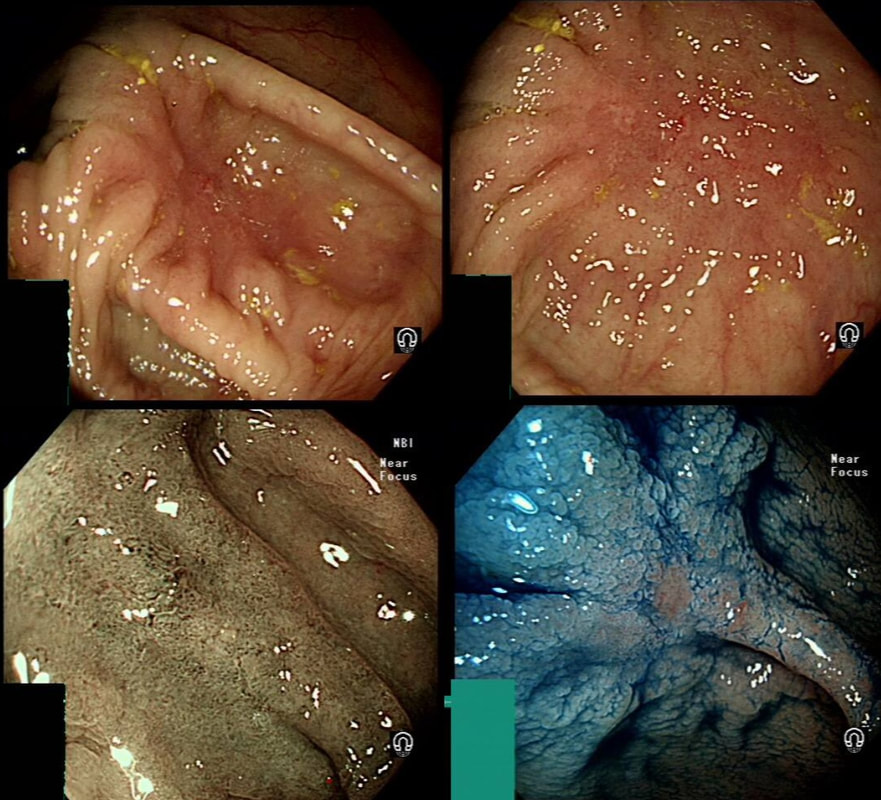
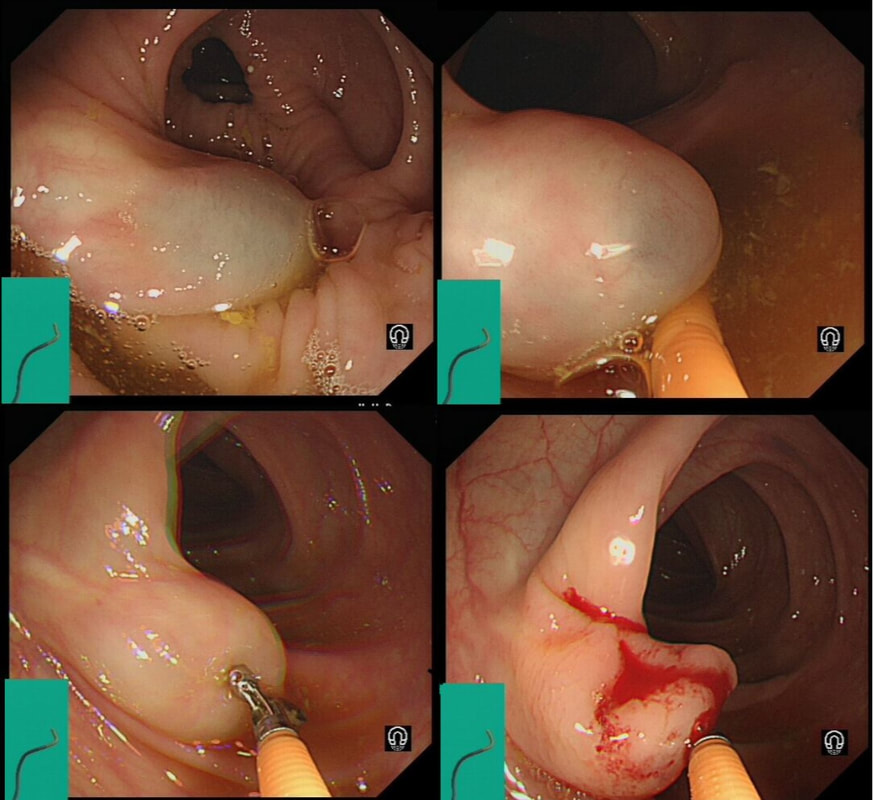
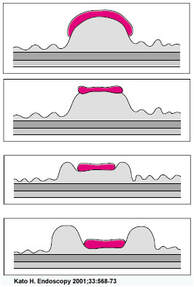
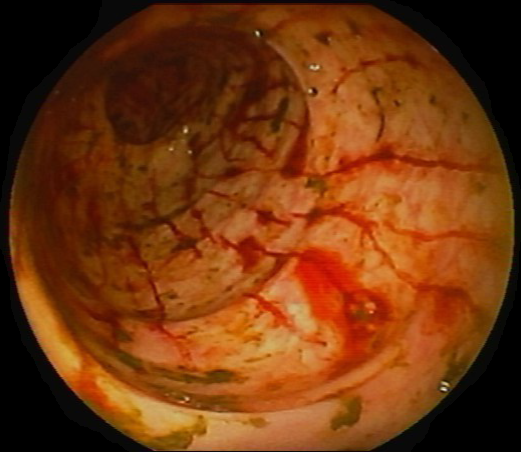
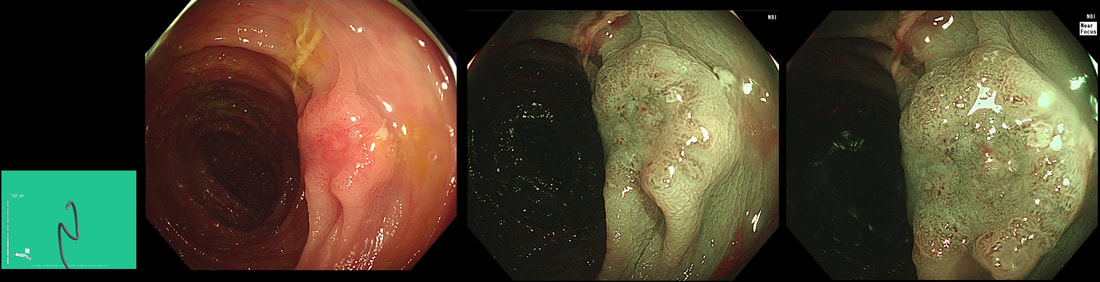
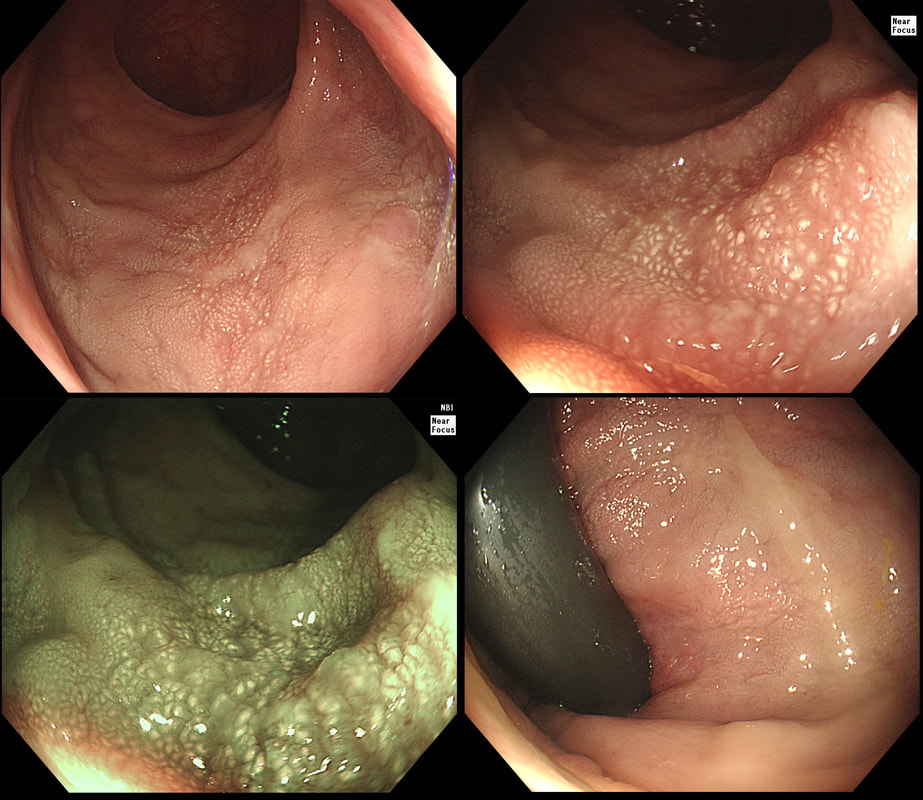
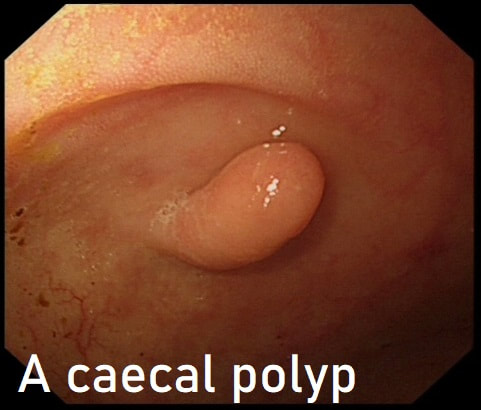
This polyp was found in the caecum.
WHAT'S THE LIKELY DIAGNOSIS?
■ Inverted appendix
The right shape but the wrong crypt pattern!
■ SSL
Absolutely, those crypts don't lie!
■ Adenomatous polyp
Wrong crypt pattern!
■ Likely underlying appendiceal tumour
Now, that's a wild guess!
explanation
I did hesitate for a moment as the lesion did, at first sight seem to arise from the caecal pole and was reminiscient of an inverted appendix (see below). However, on closer scrutiny, it has the typical wide-open crypts of a 'sessile serrated polyp' and wasn't actually that close the the caecal pole. It was removed without incidence after injecting some saline below.
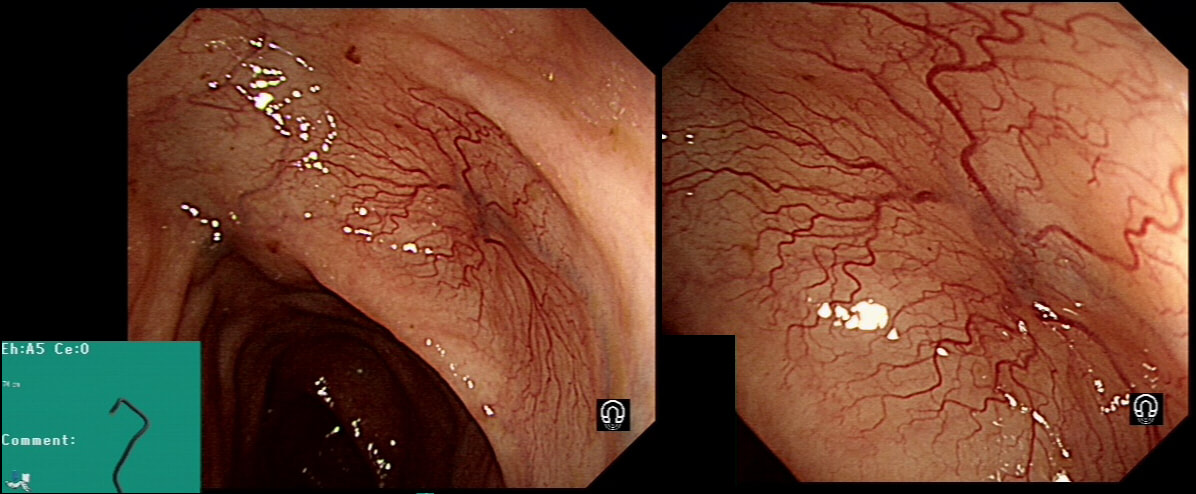
This lesion was found in the transverse colon of a patient with a change in bowel habit
HOW WILL YOU MANAGE THIS?
■ I will ignore it
Brilliant, so would I !!!
■ Apply APC in the centre
Yes but only if patient has an IDA !
■ Apply APC from the periphery and working inwards
Advocated by some but life's too short!
■ Inject saline below and then apply APC
Overkill I think ...
■ Apply a clip to the centre
Doesn't work!
explanation
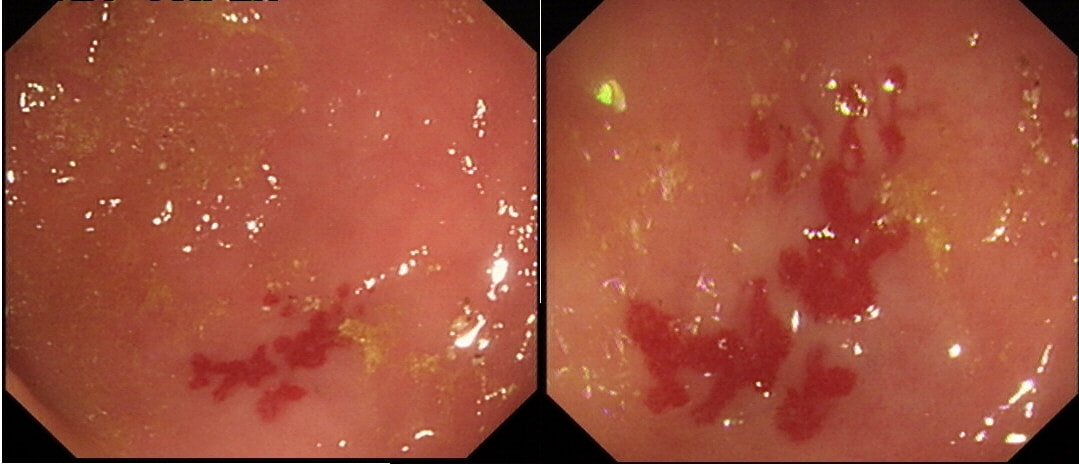
Angioectasia (formerly called 'angiodysplasia but of course these are not 'dysplastic'/ pre-malignant) are an acquired vascular malformation associated with advancing age, aortic stenosis, Von Willebrand Disease and chronic kidney disease. If a patient asks me, I usually put it more diplomatically as 'these come with maturity'. Seems kinder!
They are supposedly a little more common on the right than the left side. IF the patient is anaemic, APC is the treatment of choice. I have tried clips but they don't work. Be careful though as APC has been linked with delayed perforation in up to 1% of cases [Olmos J A. Dis Colon Rectum. 2006;49:1507–16]. I turn the APC down to 30 watts only and set the machine to 'pulsed APC'. Even so, please be sensible and stop after a maximum of a few seconds of heat! Some first raise the lesion with saline but first I don't think that this is necessary unless you are careless with the APC and secondly, I runs the risk of the underlying central, feeding vessel not actually getting ablated. Of course you could also use a 'coagulation grasper' if you are willing to pay €250 for the same outcome. Alternatively, you may consider a 'hot biopsy forceps' but of course these are now 'banned' and could land you in deep trouble if there is problem. Must admit that I use them instead of coagulation graspers because I'm too stingy to pay the €250. The correct answer in this case is probably not to do anything as the patient is not anaemic (and presumably has never been anaemic in the past). Secondly, I would advocate that you apply heat to the centre of the lesion only as those spidery legs will then disappear as in the example below.
By the way, it's not described in literature but there are actually two types of angioectasia. There are the 'spider naevi like' angioectasia as in the case above but angioectasia more commonly appear as red blobs as in the example below. Must admit that I don't know why. Must have something to do with the aetiology ...
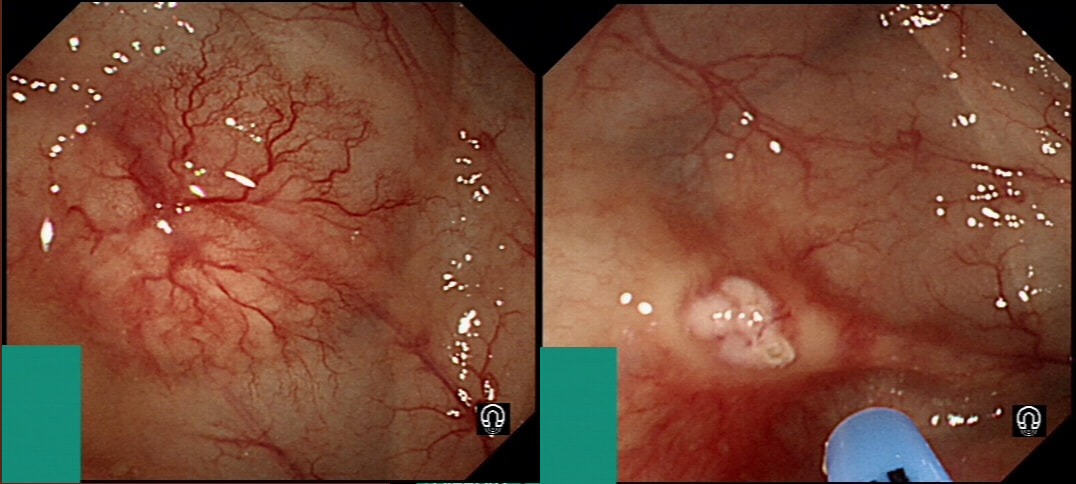
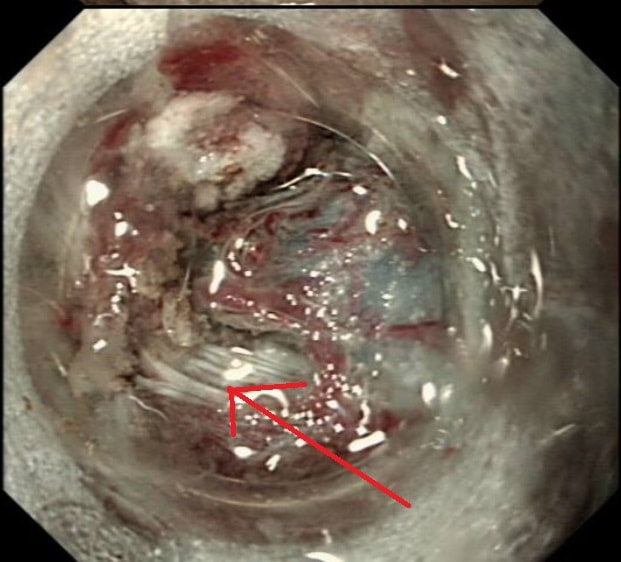
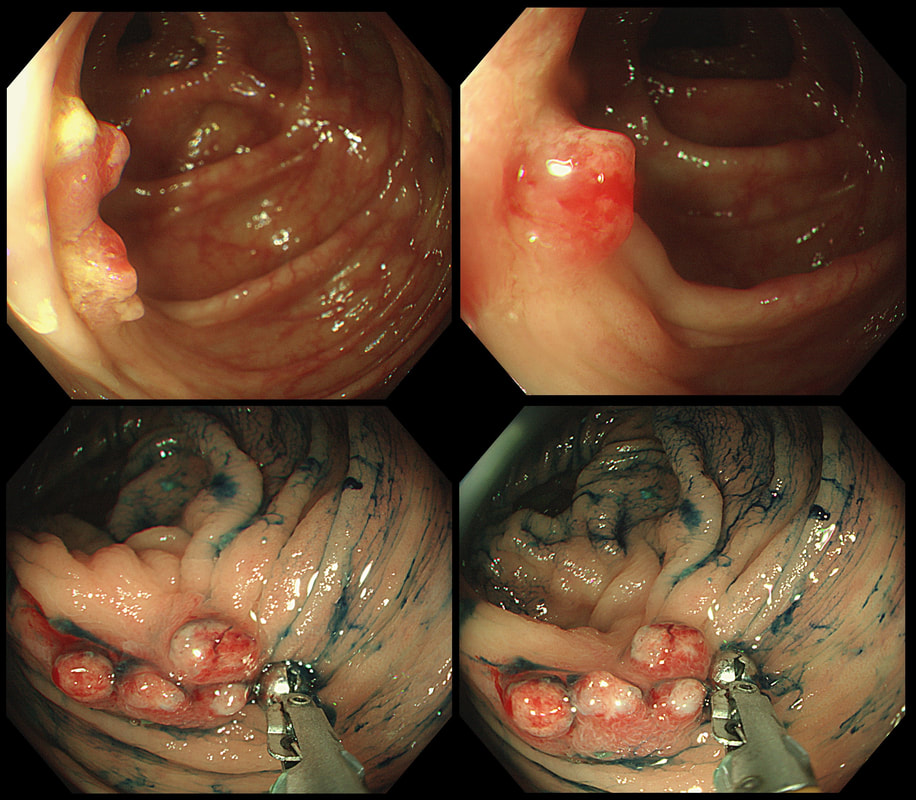
This red patch was noted in the transverse colon. You take samples and after the examination, the patient asks you what will happen next?
WHAT WILL YOU REPLY?
■ It's probably nothing
This is SOMETHING!
■ It will probably need to be resected endoscopically
Its a TA+HGD which had to be removed by ESD
■ It will probably need to be resected surgically
Now, that would be 'overkill' !
explanation
Six months ago a SSL+HGD had been removed from transverse colon. This is the EMR scar.
IS THERE A LOCAL RECURRENCE?
■ No, site looks OK
No it so doesn't !!!
■ Yes, looks like a local recurrence
In fact, it's a malignant local recurrence !
explanation
You will be surprised to hear that samples taken was reported as 'normal mucosa'!!! Fortunately, someone didn't believe this and organised an early follow up for further samples. This confirmed adenocarcinoma with LVI !!!
Bizarre how a lesion can seemingly turn cancerous after resection! Presumably what happened is that it was cancer all along but this wasn't recognised at the 'index histology'. The moral of the story? Don't take the histological diagnosis as the 'bottom line'. These guys are experts but at the mercy of accurate targeting of samples and they only have a few tiny scraps to look at. You on the other hand, can see the whole lesion. If histology, doesn't seem to correspond with your endoscopic resection, more tissue is needed. On the topic of endoscopic assessment, ALWAYS look at lesions in both anteversion and retroversion !
Just found another colonic lesion demonstrating the pillow sign.
WHAT IS THE DIAGNOSIS?
■ Lipoma
Doesn't it look translucent?
■ Lymphangioectactic cyst
Yes! Pillow sign + semi-translucent = cyst
■ Phlebectasia
A dilated vein? Nope!
■ Blue rubber naevus syndrome
The naevi are dark and firm!
■ Lymphangiosarcoma
Should then have a large solid component
explanation
Just when I'd say that all lesions exhibiting the 'pillow sign', I realised that the exception are 'lymphangioectatic cysts' (lymphangiomas) !
These are thought to be developmental malformations which can be found anywhere in or on the body. Of course they are entirely harmless. In fact the only harm they cause is by being poked about! There has been a reported case of peri-colic abscess formation following biopsy [Krishna SG, Endoscopy. 2012;44(Suppl 2 UCTN):E104–5]. Therefore, either biopsy or a 'de-roofing EMR' should probably be avoided. The best way to confirm the diagnosis, if it's in doubt is by EUS. It will show several cystic spaces confined to the submucosa and without any nearby nodes. There have been reports of lesions presenting with abdominal pain, and when pedunculated endoscopic resection may be possible [Case Rep Gastroenterol. 2017 Jan-Apr; 11(1): 178–183]. Must admit that I remain a little sceptical. Each case would have to be assessed on its own merit I guess !
This polyp was removed from the ascending colon. Histology confirmed that the lesion was a TA+LGD. However, it also mentions an underlying lipoma ...
WHICH STATEMENT DO YOU AGREE WITH?
■ There was no lipoma!
That's right!
■ Hmm, may have missed that?
You didn't miss anything!
■ Yes, this was noted at the time
The submucosa is often 'fatty' in the right hemicolon!
explanation
It's common for the submucosa on the right side of the colon to be laden with fat. In fact, you can see the fat globules in the bottom-right image where the lesion has been turned up-side-down. It can be a little alarming to see fat floating about in the mucosal defect after the resection . You may think that you have perforated but it's normal !
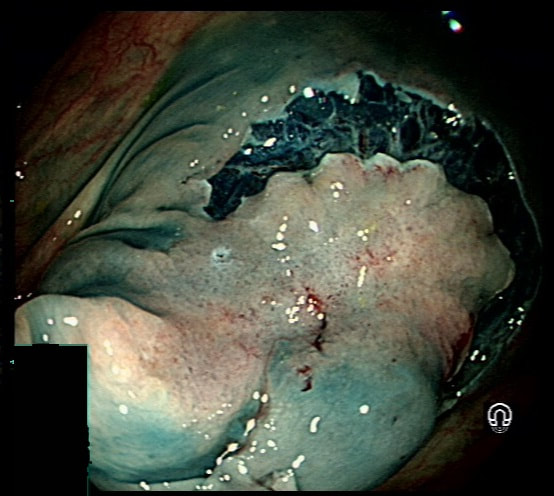
This 70 year old gentleman presented with PR bleeding and mucus. WHAT IS THE DIAGNOSIS?
■ Rectal prolapse
Nope, doesn't look like this!
■ Active proctitis
Seriously? How long have you been scoping?
■ Solitary rectal ulcer syndrome
SRUS can look almost like anything but not like this!
■ Rectal adenoma
Yes, to be specific it's a villous adenoma!
■ Rectal carcinoma
That polyp is big but not ugly!
explanation
Perhaps something of a 'noddy' question as any endoscopists worth his salt should recognise this as a large villous adenoma. However, many endoscopists would not see beyond the sheer size of the lesion and conclude that the lesion must be malignant SOMEWHERE! The truth is that it's precisely because the lesion is inherently innocent that it has grown to this size WITHOUT turning malignant! So how to deal with it? An endoscopic resection would include a circumferential resection for about 12cm, starting at the anal verge. There are several options; ■ Perhaps you could remove it in sections? For example, removing a quarter and then bring the patient back after a few month to do another section? I've tried it! Sadly, they grow back so quickly that by the time the patient returns, it has already re-grown to recover the previously cleared surface ! ■ Perhaps you could use APC, not to clear the lesion but to reduce the amount of blood-stained mucus it produces? I've tried that too!!! Unfortunately, APC tends to make the mucus production worse !!! ■ There is one more thing which I have learnt with large villous adenomas like this. They tend not to lift very well! As the lift is always 'shallow', you end up with a very narrow submucosal window to dissect through. Similarly, a piecemeal EMR is almost impossible unless the VA is much smaller. ■ The only thing which we haven't tried in Leeds is a joint effort whereby 2-3 endoscopists take it in turns to dissect the lesion over a 8-10 hour period, perhaps under GA. A daunting prospect and I doubt that it's actually feasible. Naturally, a TEMS procedure is similarly unlikely to succeed and the patient is probably most likely to wake up without a rectum at all. However, loosing the rectum to benign disease at the age of 75 would be a bitter pill. Perhaps that's why most choose to put up with the mucus ? 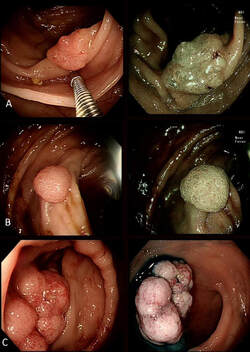
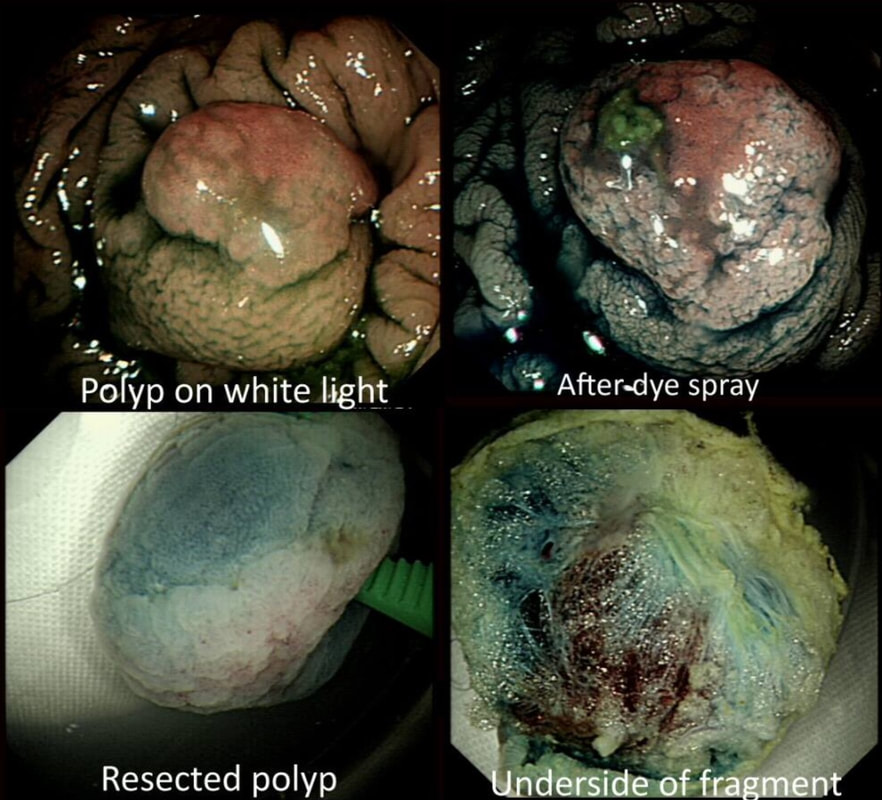
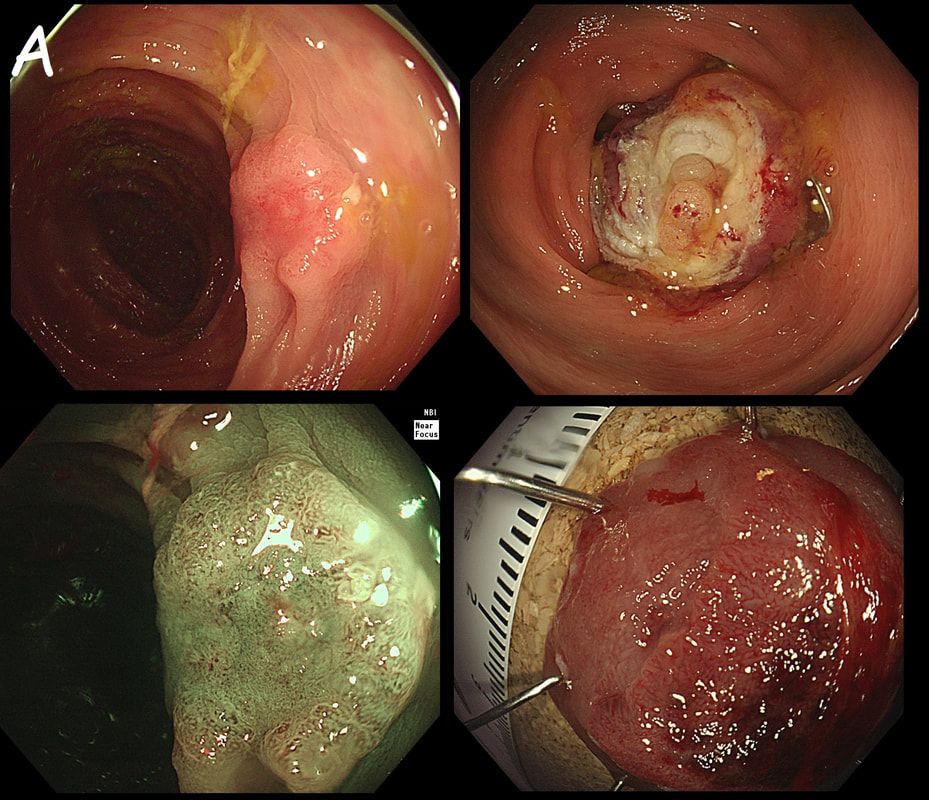
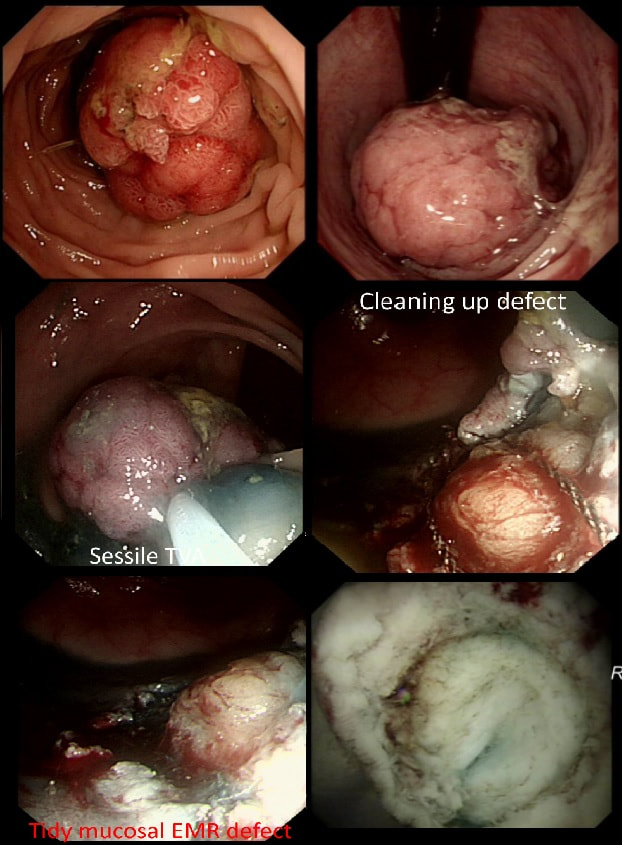
Three colonic polyps to choose between !
WHICH IS THE MOST SUSPICIOUS?
■ A
I agree! Looks like cancer!
■ B
Looks too pretty to be evil !
■ C
On size criteria, the risk is probably about 1:7
explanation
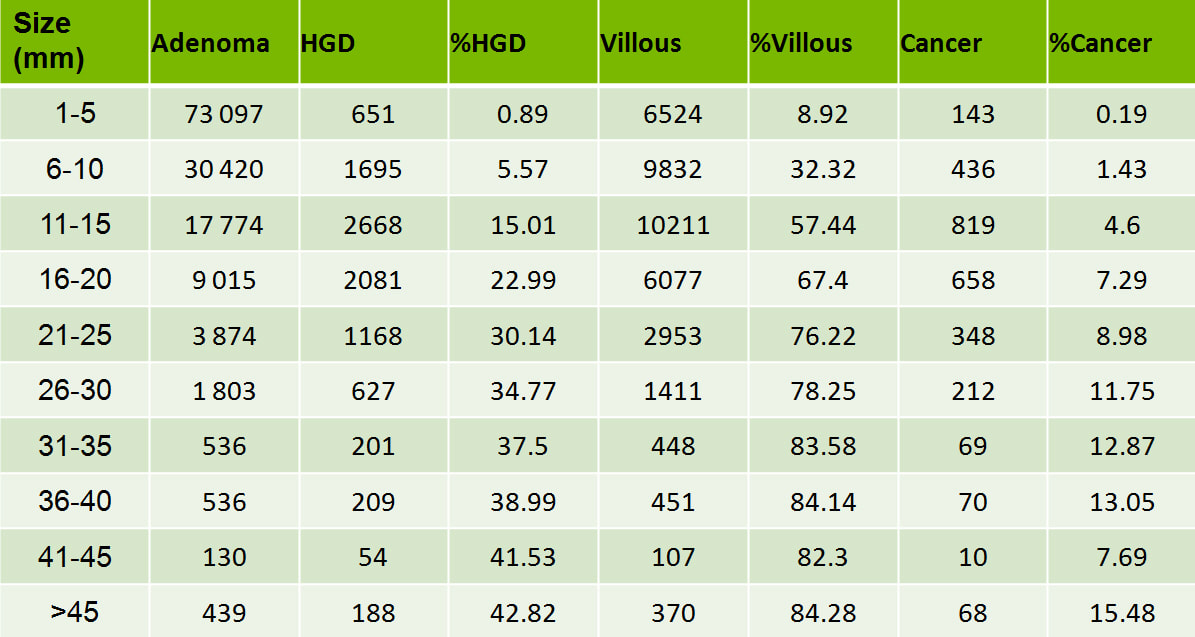
B has a lovely brain-like crypt pattern of a rather pretty TVA. Of course, C is difficult to assess as the surface crypt pattern may not reflect was is lurking deep inside. Data from the NHS Bowel Cancer Screening Programme (see table below) puts the risk of cancer in a >45 mm at 15%. For this reason, the polyp should be removed in as few fragments as possible.
However, A is the polyp which I think looks most suspicious. It has a suspicious central nodule and on the NBI image (top-right), I don't see an organised crypt pattern in parts of the polyp. This is a superficially invasive cancer which was just invading into the muscular mucosa (sm1 invasion)! it lifted well enough for a resection. Again single fragment resection would be paramount when removing this polyp. Below is a table which I received from Matt Rutter a few years ago. It links size with histology for polyps resected on the NHS Bowel Cancer Screening Programme
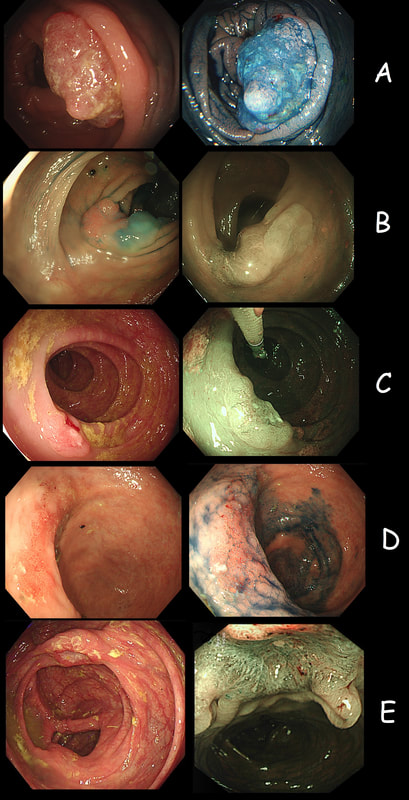
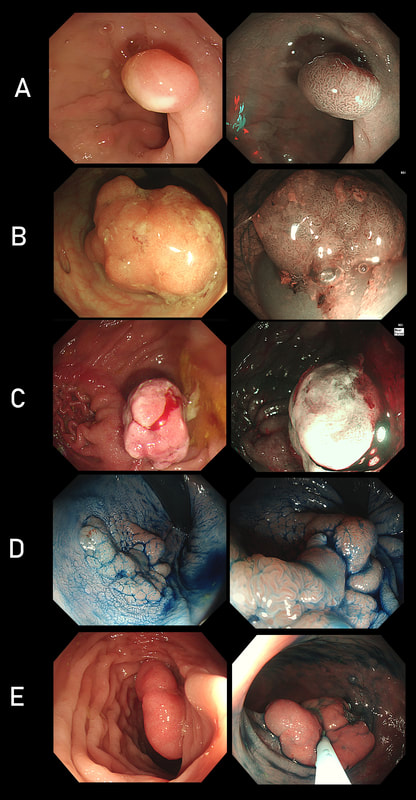
As you know, colonoscopy protects less well against future cancer in the right hemi-colon, than cancer elsewhere in the colorectum. Here are 5 lesions, all found in AC. Four are malignant and one isn't ...
WHICH ONE IS NOT A CANCER?
■ A
SM3 invading CRC with very little adenomatous tissue present
■ B
Cancer arising in an SSL !
■ C
A very superficial CRC removed by ESD
■ D
Yes! This is a 'caecal patch' in UC
■ E
Nope, its a mucinous carcinoma!
explanation
You can probably tell that the first lesion is a cancer because of it's nobbly surface and lack of crypt pattern in places. It was a sm1 invasion T1,N0 cancer (i.e. invading up to the muscularis propria layer but didn't invade into it).
The second lesion is a sessile serrated lesion with a small cancer at it's edge. The staging was identical to the first lesion (sm1 invasion T1,N0 cancer). The third lesion was a superficial carcinoma (sm1 invasion only) and the only in the series actually removed endoscopically. The fourth lesion is the 'odd one out'. It's actually a 'caecal patch' in a patient with ulcerative colitis, only affecting the rectum. The final lesion was a 'mucinous adenocarcinoma'. Interestingly, analysis of the initial biopsies reported 'TA+LGD'. Don't believe everything those pathologists tell you! Your eyes don't lie! There is a further interesting twist here. After another set of samples, got the pathologists to agree with our endoscopic diagnosis, we were surprised that the patient was turned down for surgery due to severe comorbidities. Rather begs the question what was the point in putting the 85 year old gentleman through two colonoscopies !!! Anyway, that was 4 years ago and he has still not developed any symptoms from his cancer!? A recent CT confirmed that it was slowly growing and was now annular but without any evidence of spread or obstruction. Interesting and clearly turning someone down for surgery is not necessarily a 'bad thing' ! Still leaves me wondering why they keep scanning the poor bloke though? Below I've attached a reminder about the 'Kikuchi staging' which your pathologists should use to stage superficial flat cancers.
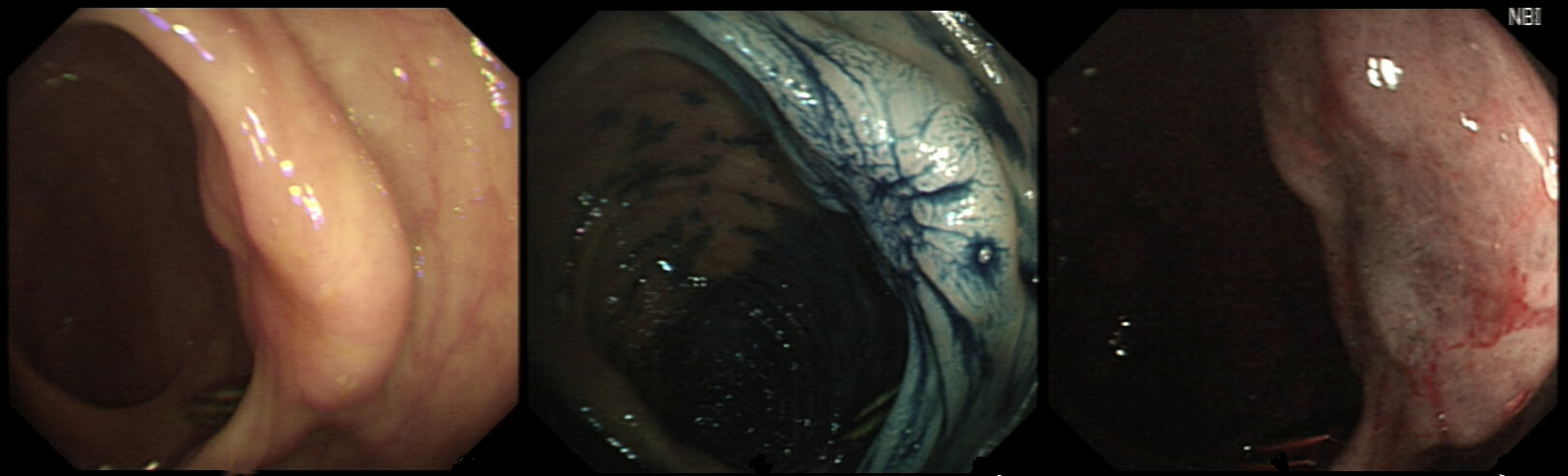
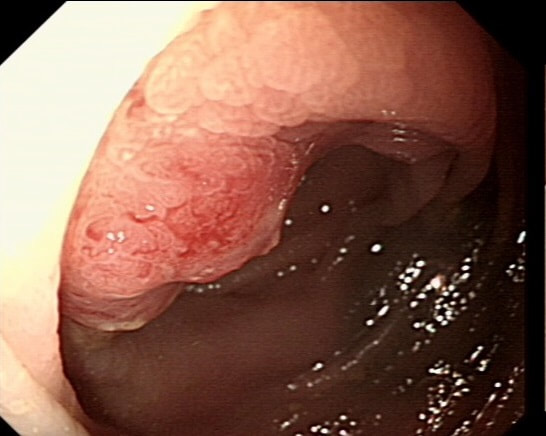
You are called into a neighbouring endoscopy room to advice on this lesion at the recto-sigmoid junction.
WHAT WOULD YOU DO NEXT?
■ Carefully assess and take samples
Yes but in what way would you 'more carefully' assess this?
■ Attempt to remove it now
Well, you would realise that it doesn't lift !
■ Attempt to remove it at a dedicated session
Noo! You missed it !
explanation
This lesion does look completely different when viewed in retroversion (image below). In retrovertion its obvious that it's malignant. Clearly the correct way ahead is to take some better targeted samples and refer to the next colorectal cancer meeting. If you hadn't assessed the lesion in retrovertion, samples would have revealed a TA+HGD only (this is actually what happened), delaying the correct management. The moral of the story is: always view all of the lesion before deciding on where samples should be taken from.
Aha! Are we not told that; "polyps which will be resected endoscopically shouldn't be sampled as it makes removal difficult"? The problem is that when patients have to wait long for their resection, it makes sense to take a few samples or at least do a 'test-lift'. Having to wait 3 month for a resection only to be told that the lesion is likely to be malignant is a disaster scenario. It's then better to exclude cancer as far as possible before asking the patient to wait months for the removal. Of course, its true that it can make a resection more difficult but it's hardly ever a showstopper !
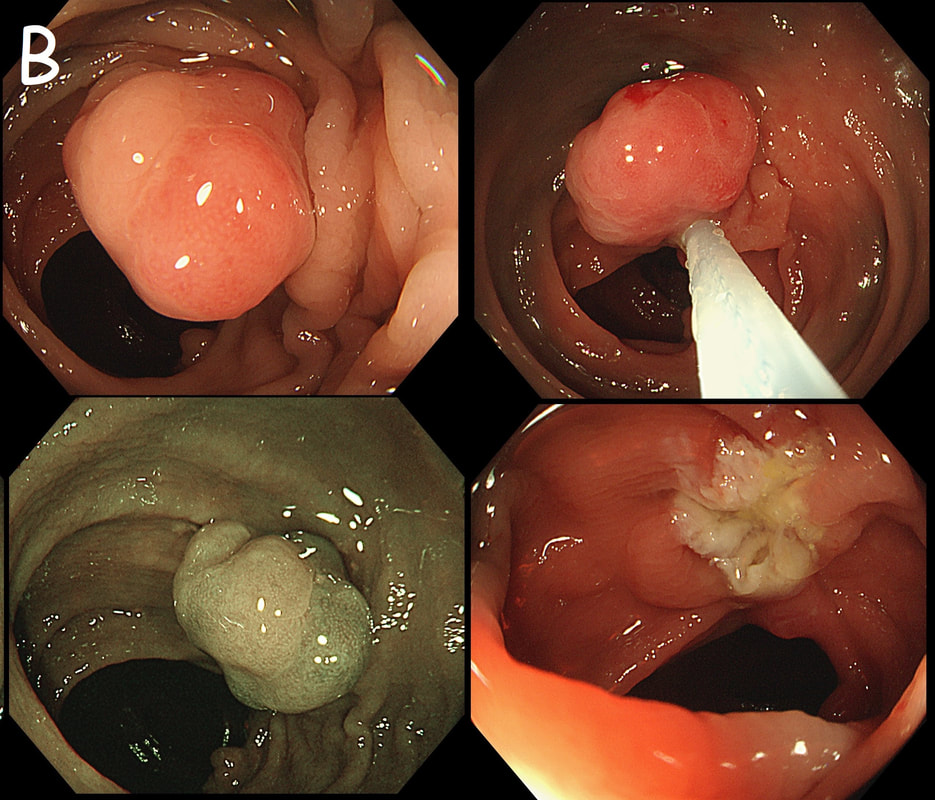
The lesion above is removed 'full thickness' and turns out to be an sigmoid carcinoma extending up to, but not involving the muscle propria layer (i.e. it's a T1 cancer with sm3 invasion). Histology reports clear margins but when the patient returns for a 'site-check' endoscopy, the nodule below in photograph B is found at the resection site ...
The mysterious nodule is removed and sent for analysis (Histology below as photograph C ) .
WHAT IS THE DIAGNOSIS?
■ Granulomatous/inflammatory reaction
Doesn't look like it!
■ Mucosal prolapse reaction
Yes, what else could it be!
■ Local recurrence of adenoma
Doesn't look adenomatous!
■ Local recurrence of carcinoma
Could be a concern, perhaps submucosal deposit but histology reported clear margins ...
Explanation
Endoscopically, this doesn't look like anything! It doesn't look either inflammatory or neoplastic (i.e. adenoma or cancer). Why then was it removed?! Endoscopically, this looked like normal mucosa but what then is it? To find out, the lesion was removed. I had my suspicions as to what was going on and took great care to close that tiny, unremarkable looking resection site with 4 clips.
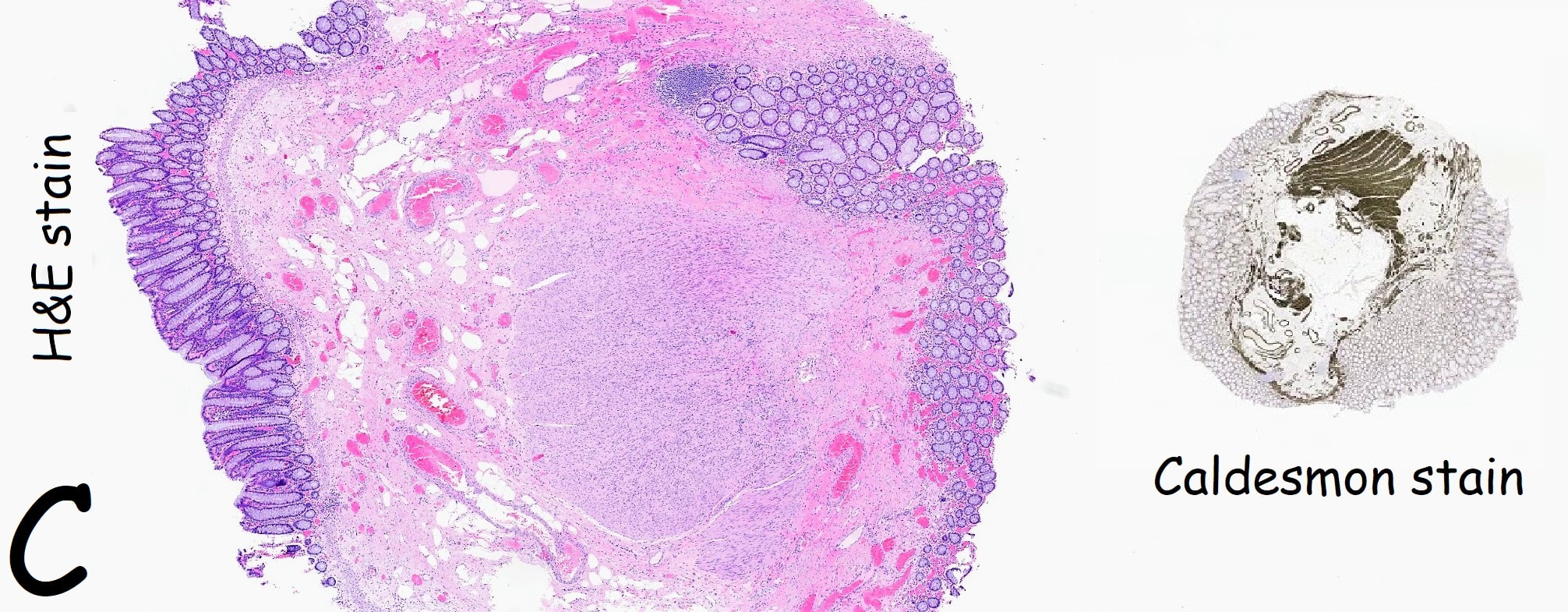
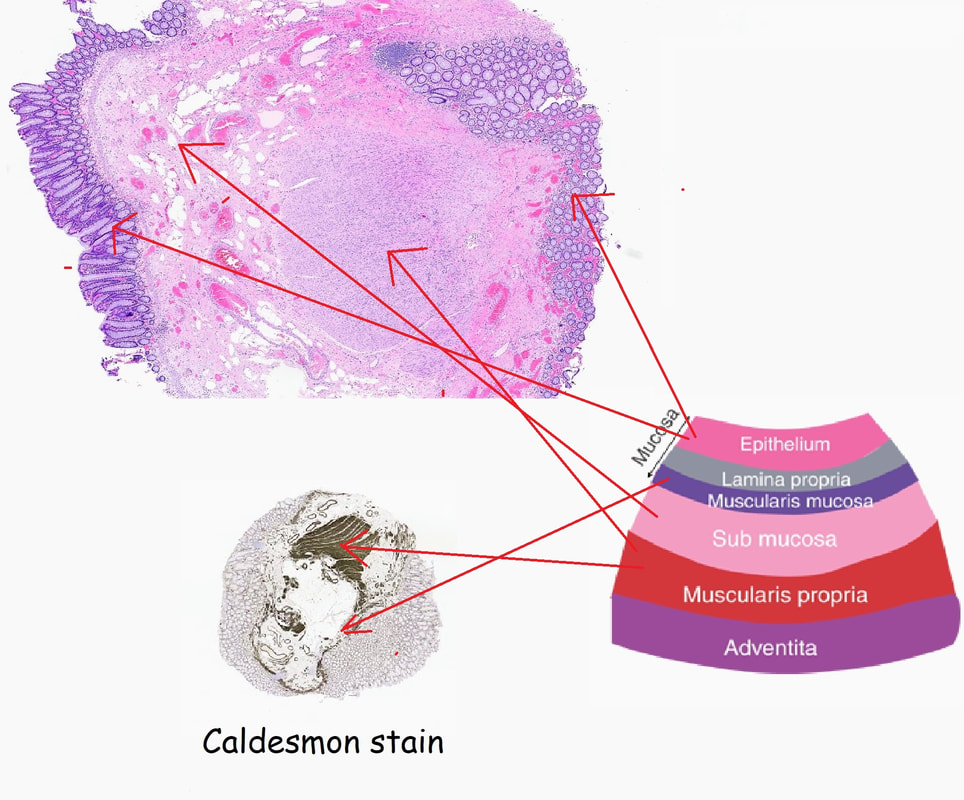
Histology did confirm that this is a full thickness mucosal prolapse at the FTR site. Of course, the point is that if you don't recognise this and resect it, you will end up with a delayed perforation !!! I've attached an annotated histology slide below. As a professional you should understand histology. By the way, the 'Caldesmon stain', shows up muscle. That large grey blob is of muscle propria in the centre of the 'polyp' and the thin grey layer around the periphery, just below the surface epithelium (the polyp is cut in a horizontal manner), is 'muscularis mucosa'.
Flat polyp found in the ascending colon of an elderly patient with anaemia
WHAT DO YOU THINK TO THE LIFT?
■ Good - Attack!
Hmm, you are an optimist here!
■ Borderline - proceed with caution !
Yes, or bail out unless you are sure of what you are doing !
■ Poor - Abort !
There is no shame in bringing this back to the Cancer group to think again!
explanation
Histology confirmed that the centre was a poorly differentiated cancer which was 'probably completely removed but only with a <100 micron clear deep margin. As I keep saying, don't expect there to be "entirely good news" after an endoscopic resection of a colonic cancer.
So what happened? In spite of the original surgical misgivings the 80 year old patient subsequently underwent a right hemicolectomy which did confirm that the lesion had been completely removed by the earlier ESD. Would a full thickness resection had made any difference? Of course not! The centre of that cancer would still have been poorly differentiated!!!
These linear lesions were seen within the left side colon of a lady undergoing colonoscopy because of loose stool. The patient denies taking any medication.
WHAT IS THE LIKELY DIAGNOSIS?
■ Post ischaemic colitis
Surprisingly (perhaps) not!
■ Post collagenous colitis
Well Done!
■ Post Crohn's colitis
Perhaps surprisingly, not!
explanation
These linear scars have been described (GIE 2010;71(1):221) in patients with both types of 'microscopic colitis' (collagenous colitis and lymphocytic colitis). Presumably it's because the lamina propria can be fibrotic, which makes the mucosa "brittle" and prone to superficial cracks. When I see these fresh cracks (example below, courtesy of Dr Gareth Davies), in the right hemi-colon of a patient with microscopic colitis, I always thought that I have insufflated too hard. However, they can evidently happen spontaneously and then heal as linear scars. In this case, the subsequent histology confirmed our endoscopic diagnosis of a microscopic colitis.
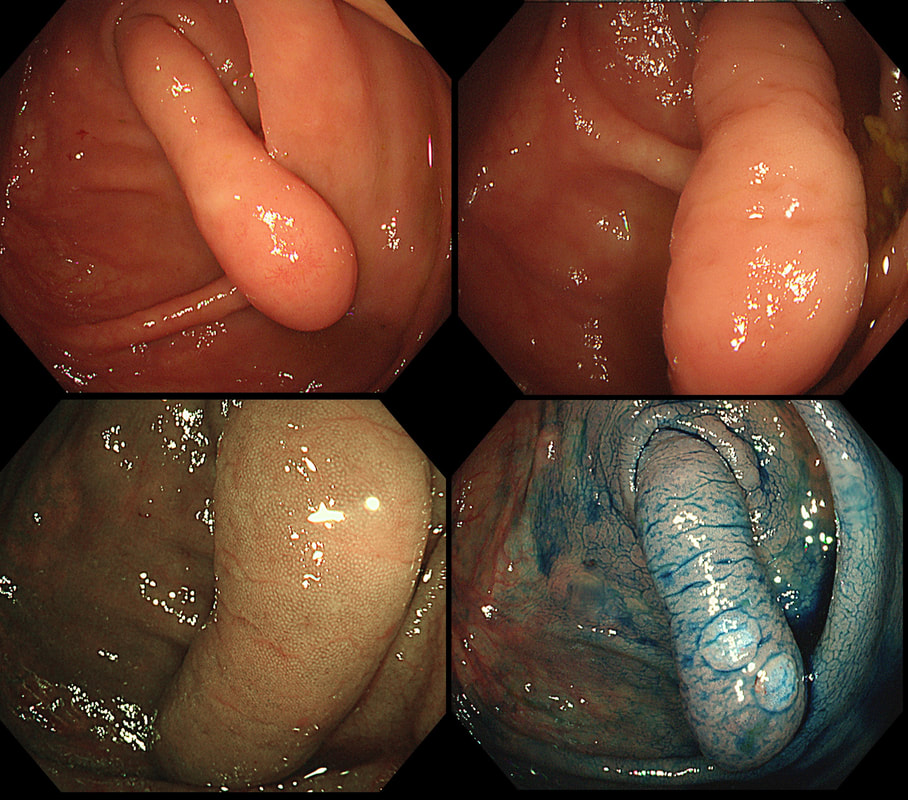
This was found at the caecal pole in an 75 yr old man. Two years ago, a small adenoma had been removed from this spot.
WHAT WOULD YOU DO NEXT?
■ Ignore the lesion
Perhaps reasonable if patient is unlikely to live more than a few years
■ Keep under surveillance
Reassuring to everyone but may seem ridicilous if patient is turned down for a resection at the age of 80, following 3 heart attacks and a stroke ...
■ Attempt another EMR
Very unlikely to succeed if original resection was done by someone with experience
■ Attempt a full thickness resection
Don't you need further information before deciding?
■ Refer surgically
Uncertain as not studies have compared endoscopic vs surgical approaches
explanation
The crux of the problem is two-fold: First, what is the risk that this lesion will turn cancerous and kill the patient and second what would be the risks of its' removal. • It's a little below 10mm in diameter and according to the data from the NHS BCSP, the risk of cancer within the lesion would be 1.5%. You could perhaps argue that it may be a little greater because it's a local recurrence. On the other hand the surface biopsies had been reassuring only showing a TA+LGD and this probably reduces the risk closer to 1%. The risk of the polyp turning cancerous in the future in the main depends on how long the patient lives. A Norwegian study by Eide T et.al. (Int J Cancer 1986;38;173–6) put the annual conversion rate to cancer for an ‘average’ colonic polyp at 1.25% at 5 years, 2.5% at 10 years and 5% at 20 years. A barium enema study (Stryker S. et.al. Gastroenterology 1987;93:1009–13) of polyps larger than 1cm, estimated the risk of cancer to be 2.5% at 5 years, 8% at 10 yrs and 24% at 20 years. There is also a CTC study of 306 polyps in which only 6% of polyps in 6-9mm range grew larger than 10mm at an average follow up of 2.3 yrs (Pickhart PJ et.al. Lancet Oncol. 2013;14(8):711–20). My own rule of thumb is that if the patient has a life-expectancy below 10 yrs, there may be little to gain by prophylactically removing lesions. This also fits with the latest UK postpolypectomy surveillance guideline which suggest that it should only be offered in patients with life-expectancies greater than 10 yrs (Rutter M et.al. Gut 2020;69:201-23). There are life expectancy calculators available online (just click the link). • As regards 'risks of removal', at one end of the spectrum you have the risks of an 'average colonic polypectomy' (for a subcentimetre polyp. I usually quote a ≤1:200 risk of late bleeding and ≤1:1000 risk of a perforation). Not all polyps are the same and at the other end of the risk spectrum, you have the risk linked with an 'ampullectomy/papillectomy'; 1:10 risk of acute pancreatitis for up to a few months after the procedure, 1:10-20 risk of late bleeding for up to 2 weeks after the procedure, 1:50 risk of a perforation, a 1:10 risk of late stenosis, 1:20 risk of acute cholangitis and a 1:200 risk of death ... • So what can a full thickness resection offer? A recent retrospective review by Ichkhanain Y et.al. from 18 centres (12 in the USA, 5 in Europe and 1 Canadian centre) reported on 66, young fit patients with smallish lesions (avg size 15mm). Surprisingly, 26 of the 66 lesions did not extend deep into the appendix lumen and arguably could have been removed by conventional EMR !? The authors don't elaborate and probably didn't have any further information than that 14/66 lesions had failed a prior attempt at conventional resection. 90% of patients were given prophylactic antibiotics and everyone was admitted after the procedure. Most (70%) of procedures were done under propofol anaesthesia but that is probably the normal approach in the US. The average procedure time was 1 hour. The overall chances of a completely successful endoscopic clearance was 75% (50/66). As regards complications, 17% developed appendicitis (10/58) and 6 pts these patients required surgery. I presume that the appendicitis was rather severe in these cases as patients staid in hospital for an average of 5 days following their surgery! The prophylactic antibiotics did not seem to make any difference to the risk of appendicitis. The success rate of 75% doesn't compare all that well to the reported success rates of ESD which was 95% but that study was small with only 34 cases (Jacob H et.al. Endoscopy 2016; 48: 829-36). • What can your surgeons offer? Insertion of a straight stapling device across the caecal pole whilst pulling on the appendix would probably be a quick surgical procedure without any risk of post procedural appendicitis and a 100% chance of complete clearance. Of course it would be more expensive and the usual hazards associated with a GA and surgery would be in play. By the way, Michael Bourke at Westmead Hospital in Australia writing in Gut 2021;70(2) points out that the surface area of the appendix constitute 7% of the caecal surface area. You will be familiar with a similarly sized triangle of mucosa between the two ends of the ileo-caecal valve and the appendix - the so called 'lawyers triangle' (for obvious reasons)? Michael highlights a study of 34 patients with the 'Serrated Polyposis Syndrome, serrated lesions was found in 23 and some were dysplastic (Pathology 2016;48:30–4). Unfortunately, the single most important fact is missing - what proportion of patients with 'Serrate Polyposis Syndrome' actually develop cancer of the appendix? To summarise, I don't think that it's possible to give a 'correct answer' for this question. The 'correct decision', would depend on the patients life-expectancy, the histology of the polyp (HGD would push you towards resection of course) and whether there is anything to suggest that the patient would not 'do well' if he/she developed severe appendicitis requiring emergency surgery afterwards.
This 3cm polyp was removed from the sigmoid colon. After returning to the recovery area the patient is complaining of severe low abdominal pain.
WHAT WOULD YOU DO NOW?
■ Keep in 'recovery area' for observation
Not 'wrong' a such but you can do more!
■ Bring back into endoscopy room to apply more clips
Now is your chanse to do this before that peritonitis takes hold!
■ Organise a CT
In my experience, it often muddies the water and adds little useful information in this scenario
■ Admit, organise a CT and start AB's
Not 'wrong' but placing clips is more important than any of this
explanation
Large sessile colonic polyps are a particular challenge for endoscopists. That surface crypt pattern may not be an accurate reflection of what is hiding inside! When there is cancer hidden at the centre of the polyp, this will induce a fibrotic response (the pathologists call this a 'desmoplastic response') which can tether the polyp to the muscle propria layer. Large polyps being yanked about by peristalsis can also develop fibrosis beneath even if they are benign (example below of an entirely benign VA+TSA harbouring LGD only with dense fibrosis in its centre). Of course, non-lifting is usually obvious when the lesion is small or flat but can be impossible to see below a large sessile polyp.
The first sign that something is amiss is when in spite of 20-30 seconds of yellow pedal power (i.e. a blended cut/coag/pause diathermy cycle), the polyp is still in place! When this happened, I used to curse under my breath, re-set the diathermy and with a strong hand and a further 30 seconds of diathermy, I would usually 'win' the battle and the polyp would fall off. Unfortunately, this would often turn out to be a 'Pyrrhic victory'. There would either be quite alarming bleeding or a perforation. Nowadays, I believe that these large, sessile polyps are best removed by ESD. In a slow, controlled manner you can then dissect below the lesion. If you encounter fibrous tissue you can attempt to dissect around it, abandon or (if you've got the balls) attempt to continue the dissection in the same plane through the fibrous tissue. Of course, you can also deal with those chunky vessels, one-by-one in a more controlled manner. Back to the scenario in question. If I think that there could be any risk that I've perforated, I would bring the patient back into the procedure room to place LOTS OF CLIPS. In truth, I did immediately recognise the peri-colonic fat in the middle image on the right and closed the perforation without delay. That white base to the upturned polyp in the last image, is a sizeable chunk of muscle propria layer. Anyway, provided that there is no peritoneal contamination, the perforation has been thoroughly closed with a virtual 'suture line' of clips and the patient is young and 'sensible', I would observe them for another few hours in recovery and discharge provided that they remain free of abdominal pain. Beware of the patient who remains in pain but claims to be comfortable because he is desperate to get back home... Of course, the patient must understand my verbal and written information that there may have been a small perforation which I have closed with clips but that he/she must return to hospital if further pain develops. Of course, my standard post-resection information leaflet always advice patients to return to hospital if bleeding or pain develops up to 2 weeks after the resection. No antibiotics, no scans, no admission!?! Controversial of course and I must stress that this is my own management strategy. I believe that my way of dealing with micro-perforations is safer than admission (exposing the patient to all the in-hospital hazards of multi-resistant bugs, Covid-19 etc), reduces the risk of 'over-treatment' (CT scans after EMR's or ESDs' always look alarming to the radiologists with gas in the wall of the bowel and the peritoneal cavity which means that surgeons will be reaching for their knives) and cheaper (of course). By the way, I am no great fan of the over-the-scope (OVESCO) clips for three reasons; a) time is of the essence in perforations and with the OVESCO clip you loose time but having to withdraw the scope to attach the device and b) it is very cumbersome and it can be difficult to reach the lesion and c) you only get one chance to get the clip in the correct position. With 'normal clips, it doesn't matter if a clip goes on a little wonkily, you just place another one!
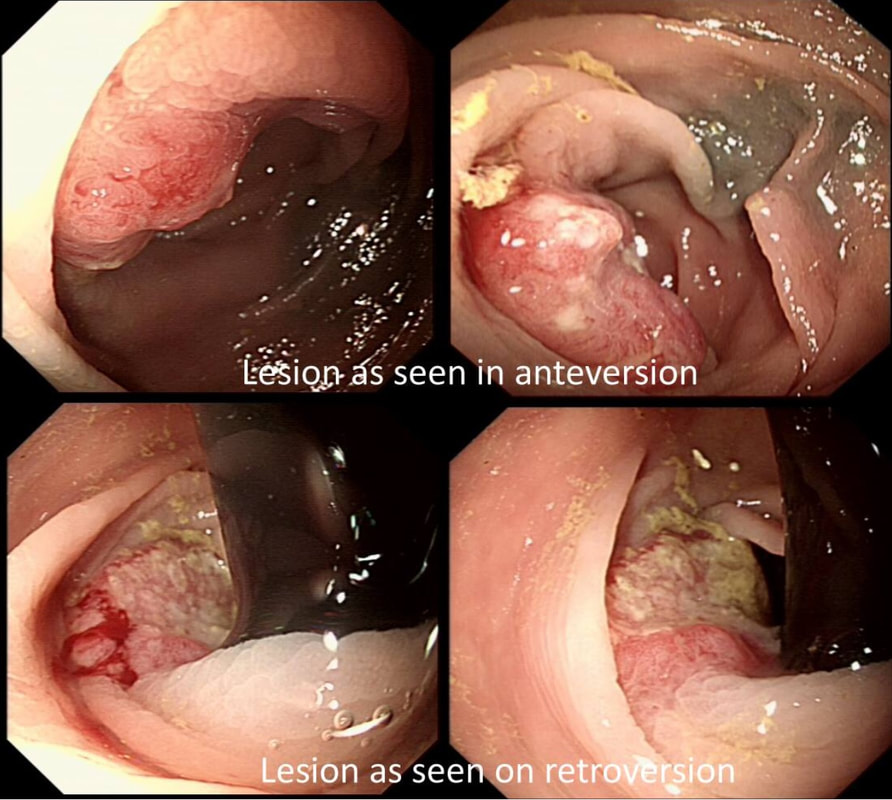
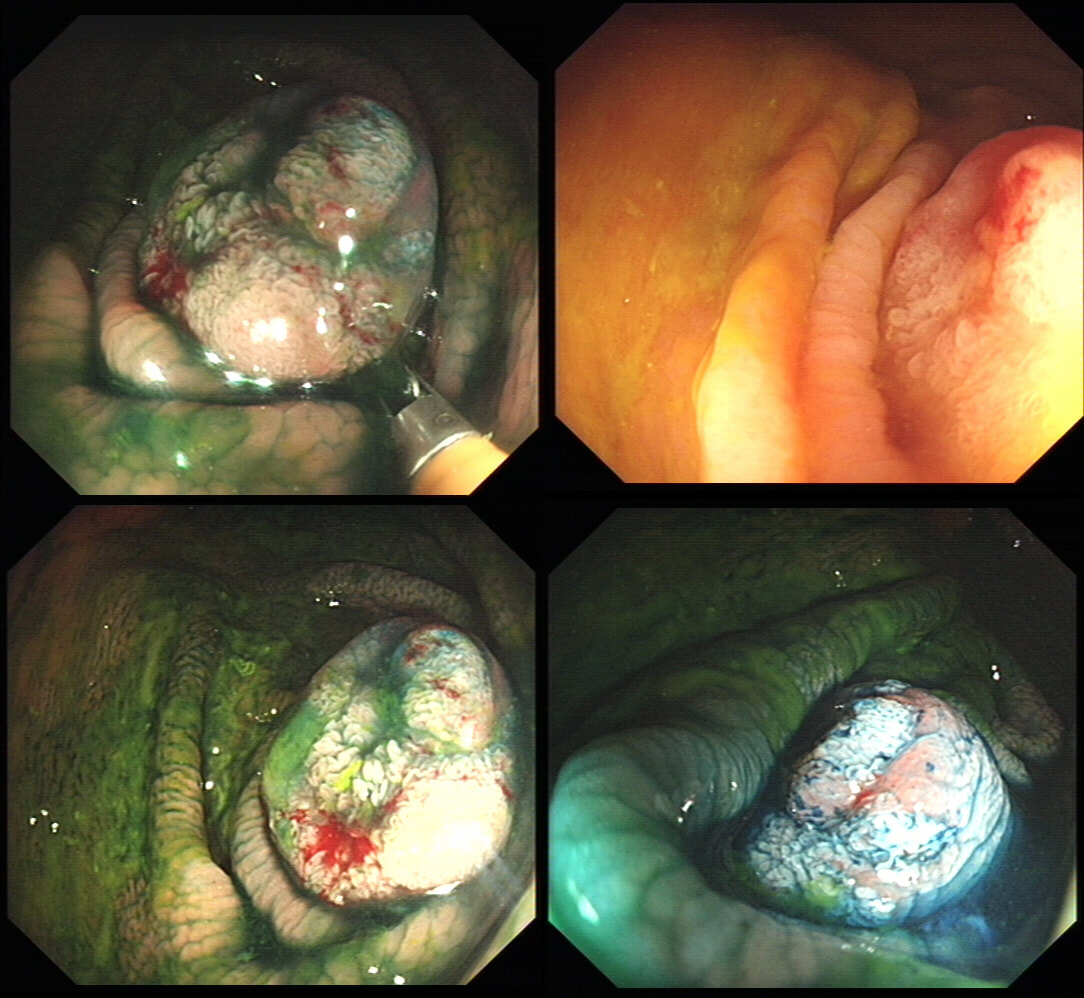
This 13mm colonic lesion has been referred for resection
WHAT IS YOUR PREDICTED HISTOLOGY?
■ Benign adenoma
No way! This looks like cancer!
■ SM1 cancer
This was our own diagnosis although the non-lifting was of a more advanced lesion!
■ SM3 cancer
Doesn't really look like it but this was the CORRECT answer!
■ T2 cancer
Wrong because the lesion should then have NO crypt pattern
explanation
Histology had 'informed us' that this was a TA+HGD. But of course, Endoscopically this IS A CANCER! 'Newbies to endoscopy' may wonder how I can be sure?
Simply, this is what early colorectal cancer looks like !!! Of course, some cancers are hidden deep within larger, sessile or pedunculated polyp and can not be glimpsed from any disturbance of the polyp surface. That is a separate issue and no amount of experience can help you with the chance finding of cancer buried deep within such a polyp. Attempting to find the words to describe the features which are 'malignant' is difficult. Firstly the lesion is firm! If you can reach it with a finger, you will find that it's hard to the touch. If you can poke it with the biopsy forceps you will find that it moves like a solid disc of firm tissue. It's also the elevated margin surrounding an angrily red centre which has a different crypt pattern is another feature. Actually, there does appear to be some sort of crypt pattern in the centre. Endoscopically we had diagnosed sm1 invasion (i.e. invading into the top 1mm of the submucosal layer). Surprisingly, it didn't lift at all and because the patient was not a surgical candidate, we offered a 'full thickness resection' (images below). Rather surprisingly, the final diagnosis was of sm3 invasion with LVI! With time you will find that removing cancers endoscopically never leaves you with absolute 'peace of mind'. There is always some adverse feature to keep you awake at night!
This patient has undergone a right hemicolectomy for cancer some 2 years earlier. He now attends for his first "post - surgery" colonic surveillance examination. This was found at the anastomosis.
WHAT IS YOUR DIAGNOSIS?
■ Granulation tissue
You ARE Right!!!
■ Adenomatous change
INCORRECT
■ Suspicious for tumour seeding
Looks odd but not because it's cancer!
explanation
There is no discernible crypt pattern, not because of cancer, but because these polyps are composed purely of granulation tissue! I also thought that it looked a little odd and took some samples (which I don't usually do when my endoscopic diagnosis is of "granulation tissue" at an anastomosis). Here is another example of granulation tissue, this time at a colonic EMR site, which looks worrying to the unwary ☺!
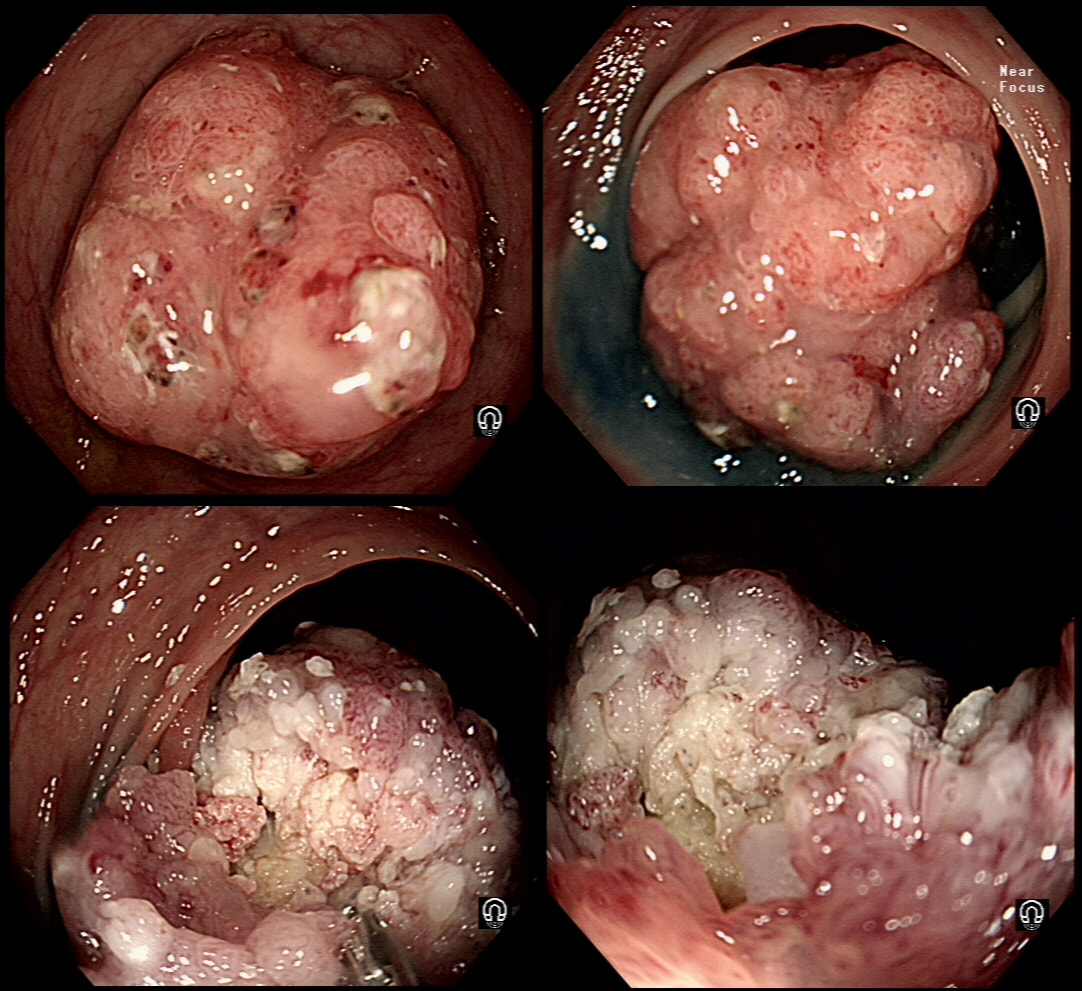
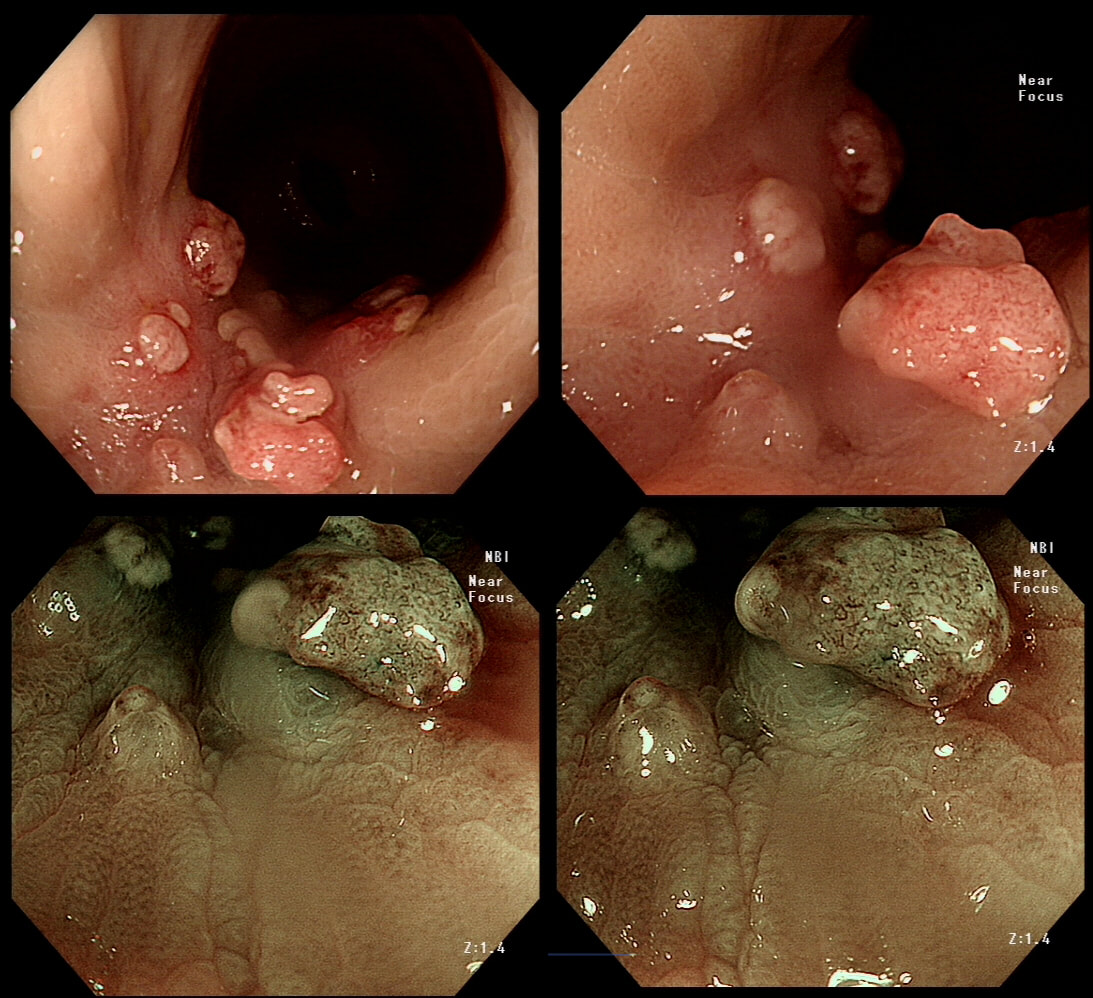
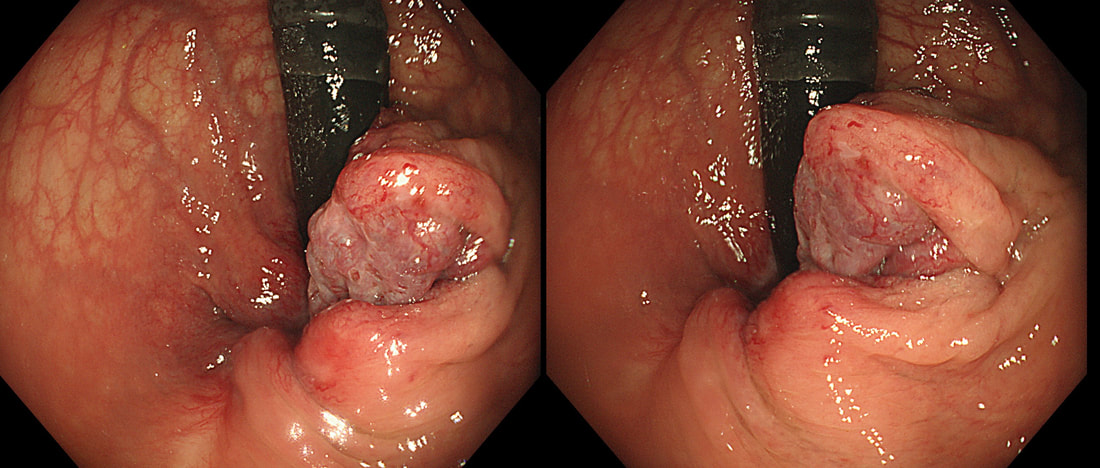
A beautiful polyp removed from the ascending colon. WHAT IS THE LIKELY DIAGNOSIS?
■ Hyperplastic polyp
CORRECT, or rather more precisely an 'inflammatory polyp'
■ Adenomatous polyp
INCORRECT
■ Malignant polyp
INCORRECT!
explanation
This is a typical inflammatory polyp! The pathologists call these inflammatory/hyperplastic polyps. However, for clarity I wish that they would simply call them 'inflammatory polyps'. After all, pure 'hyperplastic polyps' are actually a different entity in the colon consisting of mutated epithelium. In contrast, 'inflammatory polyps' like this one consists of granulation tissue! As you probably know, the white spots are superficial caps of fibrinopurulent ulcer slough overlying small spots of ulceration. It remains a mystery why such a spectacular lump of granulation tissue developed in this particular patient. There was no history of NSAID use but the patient did have 'neurofibromatosis' and the lesion was found at a surveillance examination.
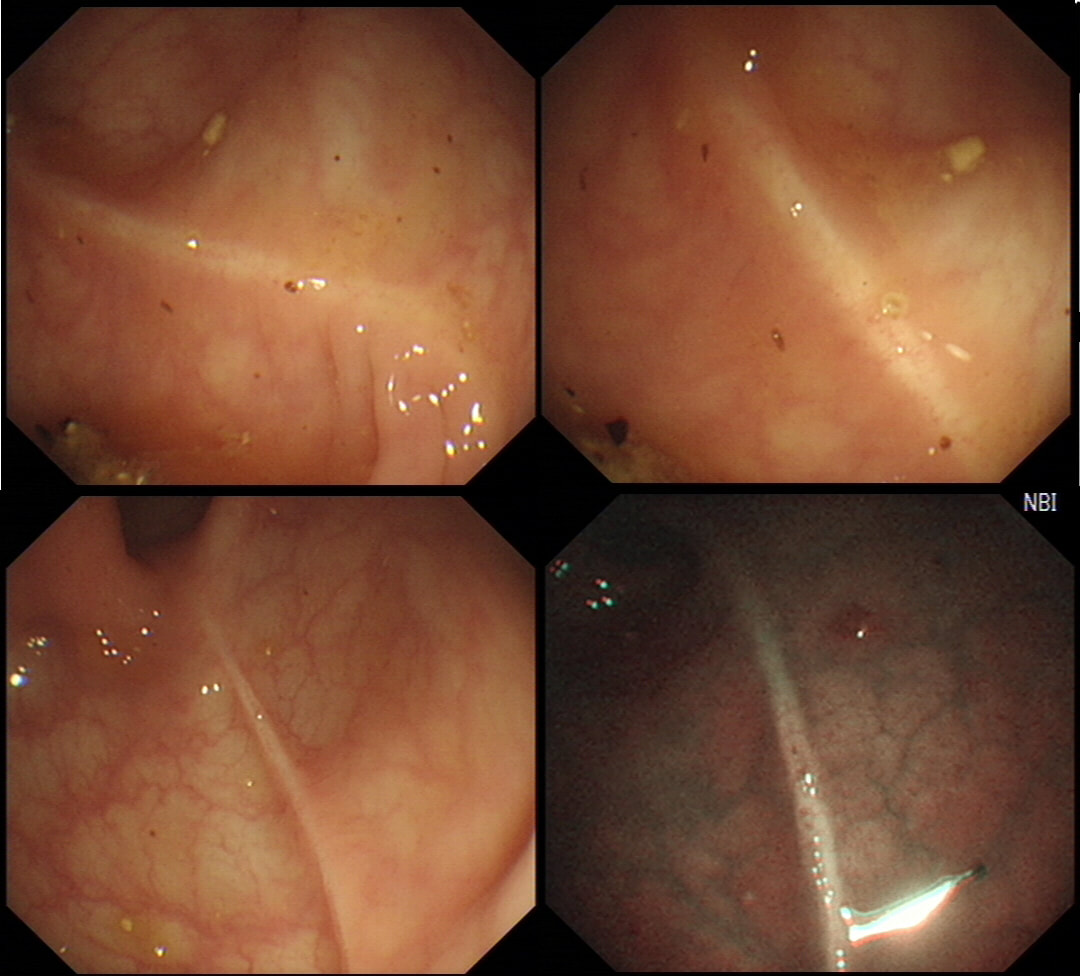
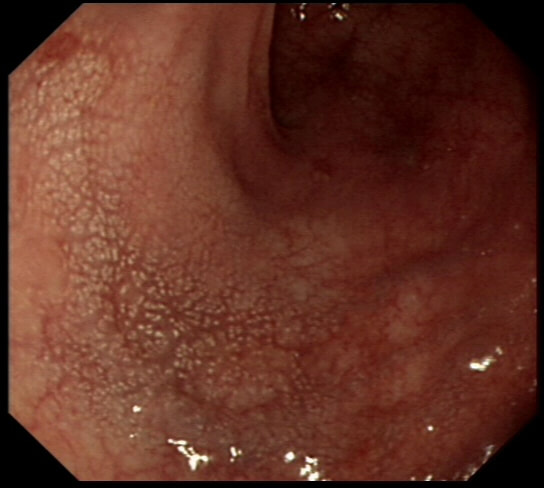
This is the colonic mucosa close to a sigmoid polyp.
WHAT IS THE SIGNIFICANCE?
■ It's of NO significance!
ABSOLUTELY CORRECT!
■ It indicates a nearby cancer
INCORRECT!
explanation
I don't apply any significance to this finding! Below is an image from the low rectum of a patient with SPECTACULAR 'chicken skin appearance' of the low rectal mucosa. However, there was no nearby lesion and histology reported: "These biopsies are all normal. There are no features to suggest a diagnosis of active idiopathic inflammatory bowel disease or microscopic colitis and there is no evidence of dysplasia or neoplasia"
It was first suggested that this finding indicate that the polyp is malignant and that the chicken skin appearance is caused by lymphatic infiltration by cancer. However, with time it became clear that this was rarely the case. More recently it was suggested that the finding was more common in polyps harbouring HGD (Intest Res. 2015;13(4):318–25). However, I am convinced that it's actually mucosal 'trauma' which causes this 'chicken skin' appearance. In the case below, it was perhaps from straining on the toilet. When you see this close to a nearby polyp, the trauma is probably from the colonic peristalsis which intermittently pulls on the polyp. Of course, larger polyps are likely to be yanked more violently by the peristalsis and the link is therefore between the size and the location of the polyp rather than the histology. It was therefore entirely predictable that the authors found a link between 'chicken skin mucosa' and pedunculated or sessile polyps rather than flat polyps and 2) that the sign was most common in the sigmoid and rectum (because these generate the most propulsive force)! This polyp was found in the transverse colon. It has not previously been sampled! WHAT IS THE LIKELY HISTOLOGY?
■ SSL
INCORRECT as the crypts are slit-like
■ Adenoma with LGD
INCORRECT as the central dip says otherwise
■ Adenoma with HGD
CORRECT!
■ Superficial cancer
INCORRECT because the lesion does lift
explanation
This is a rather interesting polyp. Well its starts off looking rather booring but after the submucosal injection it changes. Rather than a bland looking TA, it appears to be somewhat tethered in it's centre. I suspect that the subtle central dip would have been evident on retroflexion but retroflexion in the ascending proved impossible. Just look at how the scopeguide shows the shaft keeps buckling in the sigmoid and transverse, in spite of abdominal pressure to try to limit its movement. With suboptimal lift, you of course have three options: ESD, piecemeal EMR or choosing a more powerful, superstiff (but more dangerous) snare. I opted for the latter option (I usually do) and with some difficulty managed to capture the polyp single fragment. Arguably, as this lesion is a 'LST-D' type of lesion a piecemeal resection may have been less appropriate. The LST-D's are usually either HGD/IMca or superficially invasive cancer. The lift is good, telling you that it's not an invasive cancer after all. . On the other hand, a single fragment ESD may be linked with a 1:20 risk of a microperforation. In all honesty, probably not any different to the risk of perforation with a superstiff snare! This elderly patient with renal disease presented with loose motions and abdominal discomfort. A non-contrast CT had reported a sigmoid mass and a flexible sigmoidoscopy is organised. WHAT IS THE DIAGNOSIS?
■ Likely diverticular stricture
INCORRECT!
■ Stricture is probably malignant
INCORRECT
explanation
This is a benign entero-colic fistula. It's the explanation for the odd looking "double lumen". On FoE, 90% got this right. By the way, we had a similar case in Leeds a few years ago when the unwary endoscopist was puzzled when histology reported the presence of normal duodenal mucosa in what he had reported as "hepatic flexure" ☺ !
This is a 55 year old lady undergoing flexible sigmoidoscopy to investigate her PR bleeding. This lesion was found on retroverting the endoscope in the rectum.
WHAT IS THE MOST LIKELY DIAGNOSIS?
■ Fibro-epithelial polyp
INCORRECT
■ Inflammatory polyp
INCORRECT
■ Haemorrhoid
Yes!
■ Adenocarcinoma
INCORRECT!
■ Squamous cell carcinoma
INCORRECT
explanation
The three anal vascular cushions (at 3, 7 and 11 O’clock) help with maintaining continence.
When these cushions become abnormally enlarged, due to for example excessive straining (chronic constipation), age, or raised intra-abdominal pressure (such as pregnancy, chronic cough, or ascites), they become pathological. Haemorrhoids are classified as follows:
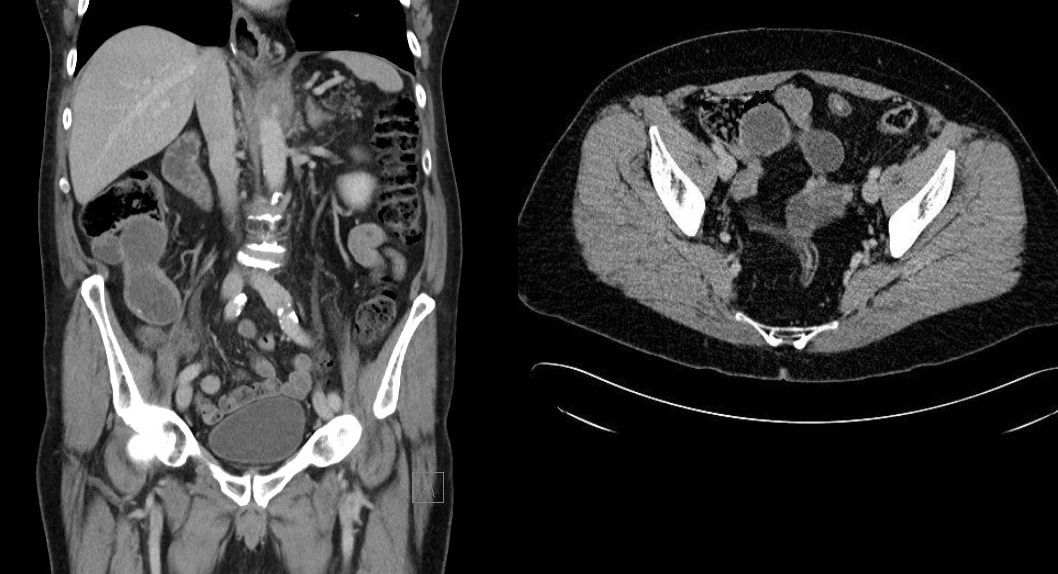
After a flexible sigmoidoscopy has excluded a low colonic lesion, most cases can be managed conservatively by reassurance, preventing constipation and topical analgesia. Alternatively, 1st and 2nd degree haemorrhoids can be treated with rubber-band ligation (RBL). A surgical haemorrhoidectomy may be indicated in the case of 3rd degree and 4th degree haemorrhoids. The serious complication of hemorrhoidectomy is anal stenosis which occurs after 2-4% of operations. However, I have the impression that this procedure is performed far less now than in the past but have not seen any data to actually confirm this impression. A 50 yr old patient complains of some low abdominal discomfort and undergoes a CT (image 1) following which a colonoscopy is organised to examine the caecum (video). WHAT IS THE MOST APPROPRIATE MANAGEMENT?
■ Biopsies only
THERE'S NO NEED FOR BIOPSIES
■ Snare Polypectomy
There is no need to remove this
■ EMR
There is no need for a polypectomy
■ ESD
INCORRECT!
■ Surgery
CORRECT, THE CT TELLS YOU THIS!
explanation
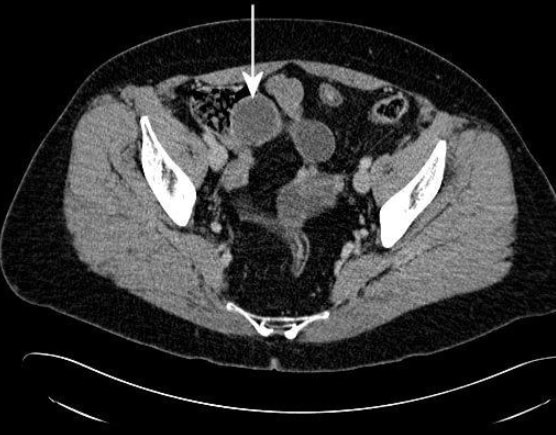
The CT shows a large appendiceal mucocoele (see arrow below) and of course therefore, this is simply a prolapsed appendix. More than 90% of our FoE group got this right.
Appendiceal mucocoeles are rare, incidence of 0.07 % - 0.3 % with a female predominance (M:F 1:4), and mean age at presentation of around 50 years. It most cases it is an incidental finding at for example ultrasound, laparoscopy or colonoscopy. The most likely aetiology is proximal obstruction of appendiceal lumen due to chronic inflammation and subsequent cicatricial contraction. Continued production of mucous from the distal appendiceal cells lead to large collection of mucus within the lumen. Noriko Suzuki told us the story of her as a junior endoscopist taking a biopsy from a mucocele, following which pus started coming out from the biopsy site! Following which the patients abdominal pain improved. She thought that she had treated the patients appendicitis but of course the abdominal pain recurred a few days later. Of course, an open appendicectomy is the treatment of choice. It has been proposed that a laparoscopic resection may be linked with a greater risk of iatrogenic rupture of the cyst. Leakage of the gelatinous material into the peritoneal cavity may lead to peritoneal dissemination of thick mucinous blobs causing recurrent intestinal obstructions, a condition called “Pseudomyxoma peritonei”. Pseudomyxoma peritonei carries a significant morbidity due to recurrent hospitalizations and a reported mortality of 50% in 5 years (Hinson 1998). There is a second reason to recommend surgery and this is the potential for malignant progression to “mucinous cystadeno-carcinoma”.
Here are five polyps in five patients.
CAN YOU TELL WHICH IMAGE IS SHOWING;
■ TA + LGD
Polyp A shows a small TA with LGD (most small polyps are TA's)
■ TA + HGD
Polyp E is a TA with HGD. All LST-NG's are TA's and many harbour HGD or even superficial invasive cancer. Recognise them and try to remove them single fragment or in as few fragments as possible.
■ TVA + LGD
Polyp D is a LST-G type of polyp. LST-G's are almost always TVA's with no more than LGD. When they 'turn' they usually develop a nodule at the site of cancer.
■ TVA + HGD
Polyp B is a sessile TVA with HGD. Of course the Devil has Horns! Furthermore, in large sessile polyps surface biopsies may not reveal what hides deeper inside ...
■ NON-NEOPLASTIC
Polyp C is an ugly little polyp without any crypts composed entirely of inflamed granulation tissue.
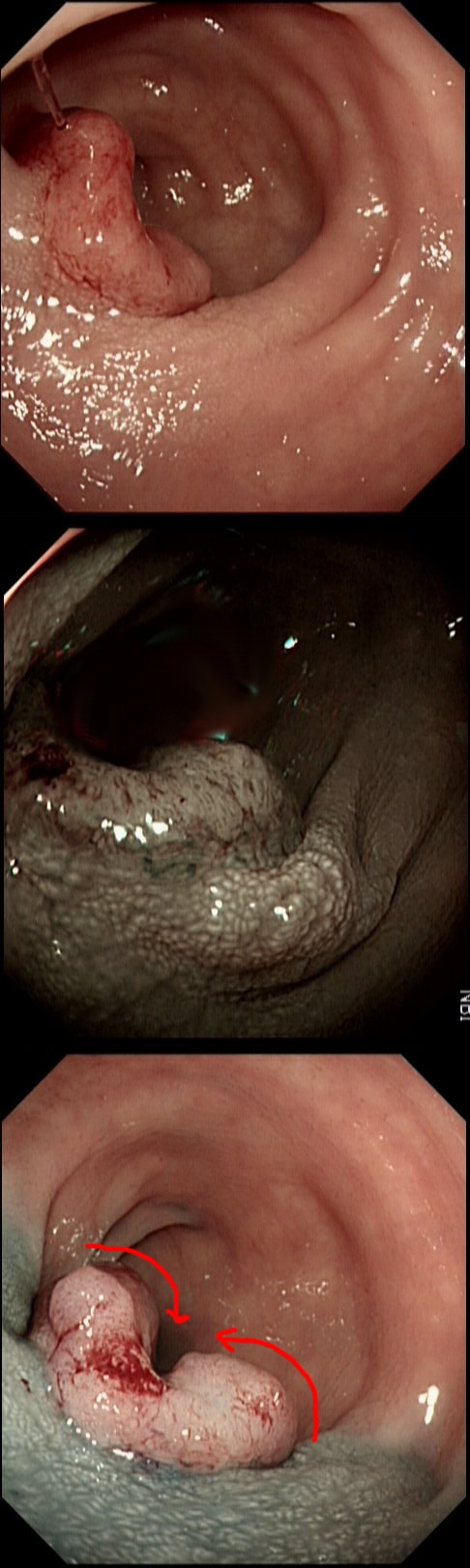
This polyp was seen in the caecum.
WHAT WOULD YOU DO NEXT?
■ Nothing!
CORRECT!
■ Take a biopsy
INCORRECT!
■ Snare polypectomy
INCORRECT!
■ EMR
INCORRECT!
■ Full thickness resection
CORRECT!
Explanation
Of course this is a normal inverted appendix and there is no need to do anything. However, occasionally there is a mass in the appendix pushing the root back into the caecum. Therefore it may be prudent to have a look at the 'apex' of the caecum to make sure that this is not indented in any way. If it was, or if there is another reason to be suspicious, a CT or an ultrasound would be the next step
|
Categories
All
|