|
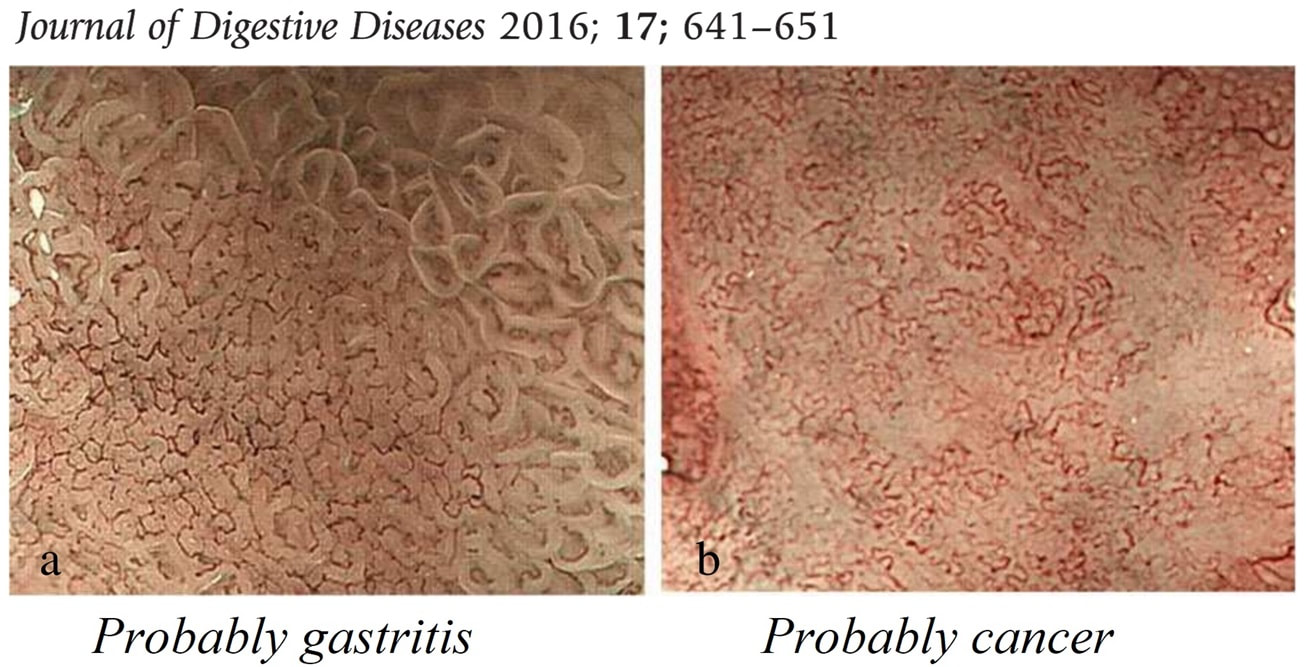
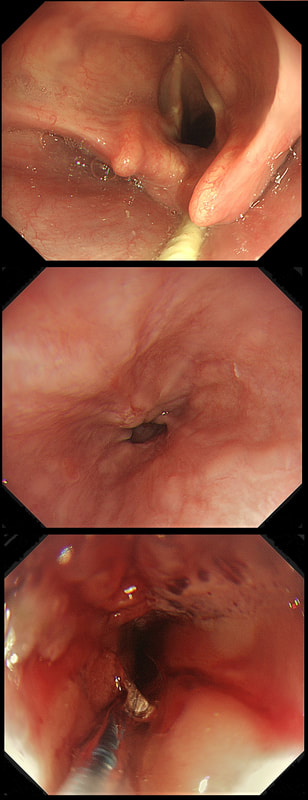
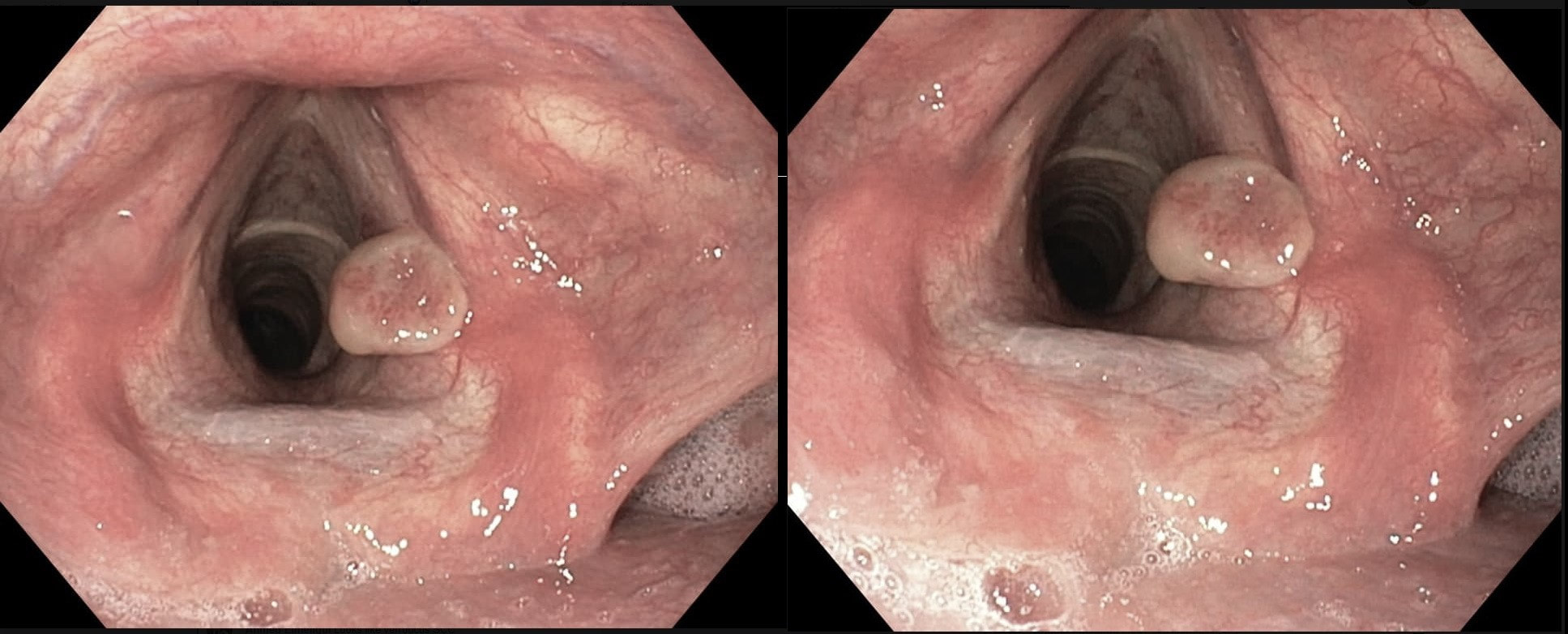
This patient is under surveillance after having had two previous EGC's. The stomach has been sprayed with indigo carmine dye when this lesion caught my eyes. WHAT IS THE LIKELY DIAGNOSIS?
■ Likely to be gastritis only
The non-staining is suspicious and rarely lie...
■ Likely to be a patch of intestinal metaplasia
There appears to be a crypt pattern in the centre but it can still be neoplastic!
■ Likely to be neoplastic
Non-staining, slightly raised and in an 'unstable stomach' - surely!
explanation
Clearly this is an unstable stomach having given rise to two previous EGC's. In patients attending for surveillance, I always spray indigo-carmine dye throughout the stomach using a spray catheter. I then look carefully at any spots where the dye seems to have slid off. This is such a 'lesion' which was confirmed as a subtle EGC. After my samples had confirmed my suspicion it was removed by ESD and was confirmed as HGD/IMca. I consider the both as the same histology as one pathologists HGD is another pathologists IMca. There was no LVI. However, I was surprised to find that one mucosal resection edge was involved by cancer. Looking back at the clip, I think that it's the 4 O'clock border which has an irregular extension which I failed to spot.
Most EGC's look like this, a shallow depression with a subtle elevated margin. I find the crypt pattern more difficult to interpret. However, as you know, the more destroyed it is, the more likely the lesion is to be poorly differentiated.
■ Inflammed granulation tissue
Was my first too! But it doesn't quite look right ...
■ Adenomatous polyp
Adenomatous polyps are usually smooth topped
■ Malignant polyp
An intramucosal cancer arising from the anastomosis
explanation
This patient had in the past undergone a Billroth II operation. You can see that the nodule is arising from the surgical anastomosis. Distal gastrectomy is a well-known risk factor for developing an anastomotic cancer later [Sitarz R. World J Gastro. 2012;18(25):3201–6]. The risk of anastomotic cancer steadily increases after surgery. About 15 years after surgery, the risk exceeds that of the background population (age- and sex-matched). For this reason, surveillance has been suggested to start 15 yrs after surgery. The case for surveillance was strengthened by the fact that dysplasia can often be found in random biopsies from the anastomosis several years before cancer develops. In spite of this surveillance in not recommended. A surveillance study in Amsterdam traced 500 patients who had undergone a distal gastrectomy for benign disease and only detected 10 cancers (6 were in an early stage). Furthermore, there was no survival advantage in the screened group after 10 years follow up [J Clin Pathol. 1984;37:748–54]. Other studies have also put the cancer yield by surveillance at around 2% and concluded that regular surveillance could not be recommended [Am J Surg 1977;134:581-4], [ Lancet 1977;ii467-9] [ Scan J Gastro 1981;suppl 16:169-71]. Nevertheless, my practise is to always take do 'opportunistic screening' by obtaining 6 biopsies or so from the gastric side of the anastomosis when I come across a case. On first glance the nodule appears to be inflamed granulation tissue only. However, there is an odd cleft in the centre and the base from which it arises is also nodular. The polyp was removed and was confirmed as harbouring intramucosal cancer. Of course, after finding dysplasia (which often does not progress) or IMca, these patients should be offered surveillance.
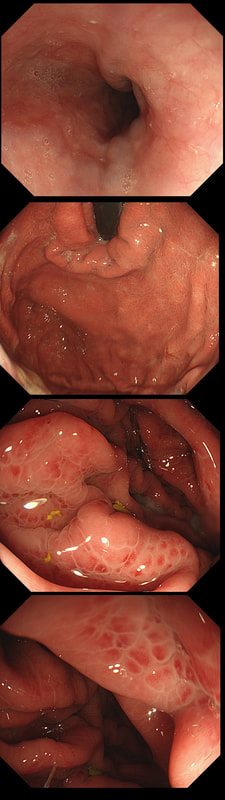
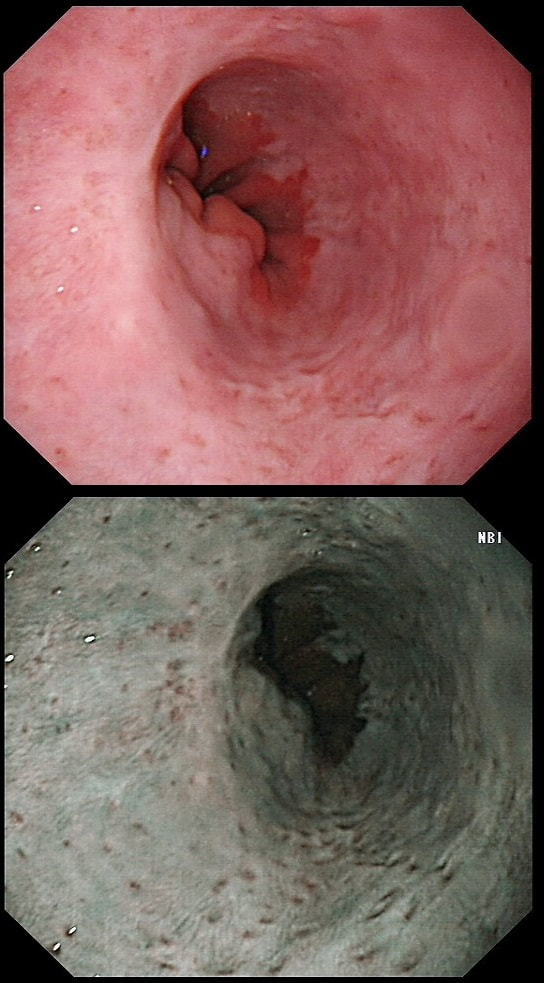
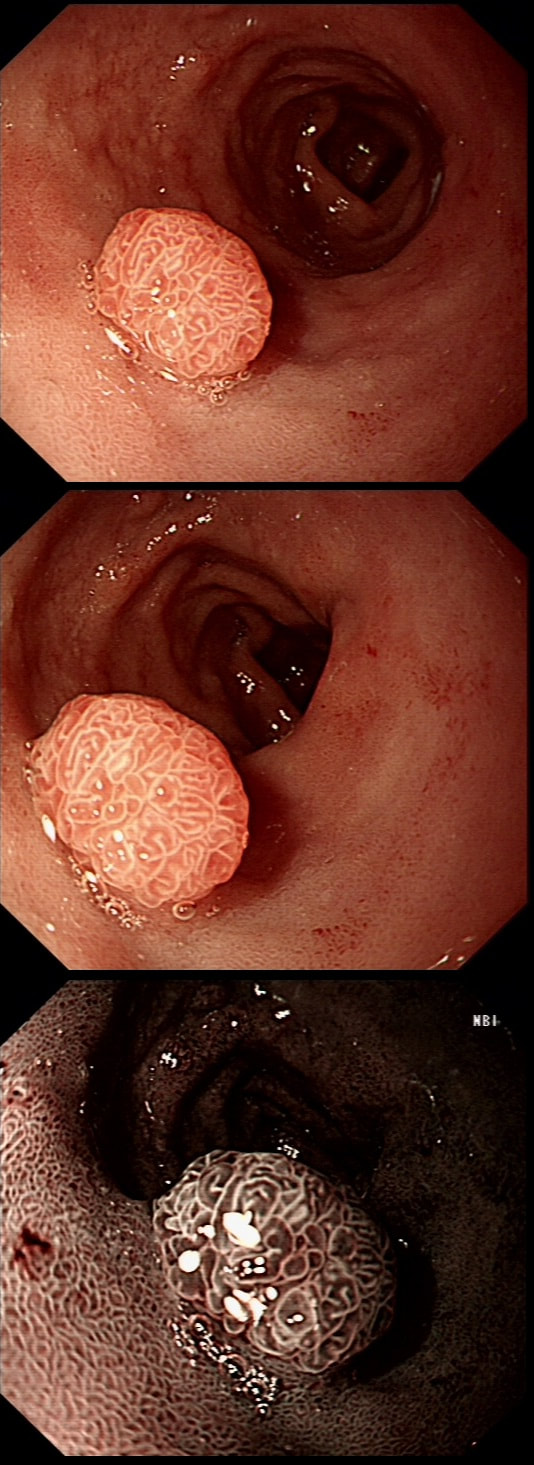
This was found at gastroscopy in a middle aged patient with anaemia
WHICH STATEMENT IS CORRECT?
■ The severity is related to the severity of the underlying disease
This is generally thought to be the case!
■ Its generally more marked in the distal vs the proximal stomach
It's the other way around actually
■ Beta blockers don't help
Beta blockers do reduce gastric perfusion
■ The condition is irreversible
A reduction in portal pressure improves the condition
explanation
Congestion secondary to portal hypertension is thought to be the primary cause of Portal Hypertensive Gastropathy. However, other factors may also play a part such as mucosal protective mechanisms, inflammatory response, local vascular tone, hepatic function, gastric mucosal perfusion, endotoxin, and gastric sucrose permeability, have been suggested to influence the development of Portal Hypertensive Gastropathy. However, because Portal Hypertensive Gastropathy is a dynamic condition which improves after liver transplantation or after TIPS, suggests that it IS portal hypertension which is the primary driver.
There is a correlation between Portal Hypertensive Gastropathy and Child-Pugh stage, HVPG (hepatic vein pressure gradient), MELD score, albumin level, bilirubin, platelet count, INR and even survival [Bang CS. BMC Gastro 2016;16(93] You are right, there is also a small oesophageal varix visible!
This is an odd looking, almost 'nodular' antral gastritis. You may be surprised that this appearance is typical for a certain type of gastritis ...
WHICH TYPE OF GASTRITIS?
■ Lymphocytic gastritis
Mucosa is usually ulcerated in LyC gastritis
■ Eosinophilic gastritis
You are correct!
■ H.pylori associated gastritis
Can look weird but not classically nodular
■ Collagenous gastritis
Is endoscopically invisible!
■ CMV gastritis
Usually ulcerative
explanation
This nodular appearance is typical of an 'eosinophilic gastritis'. In textbooks you also find that lymphocytic gastritis can give rise to this appearance. However, in my experience LyC gastritis is usually intensely red with superficial linear ulceration. See my example below. CMV gastritis look similar but the ulcers are usually round rather than linear.
Anyway, finding more than 30 eosinophils per high power field (HPF) in at least five HPFs has been suggested as diagnostic. There is an argument that pathologists shouldn't make a diagnosis which doesn't change management. Not sure that I agree with this. After all, their job is to describe what they see! Nevertheless, it's probably prudent to ask about food or drug allergies, and consider if patient is atopic. A search for Helicobacters is probably also indicated. I would probably also consider taking further samples from the oesophagus as unrecognised and untreated eosinophilic oesophagitis can ultimately develop into a stricturing disease. Apart from this, it's difficult to know what to make of this diagnosis! Would you do a colonoscopy to chase after the possibility of asymptomatic IBD? This patient is attending for dilatation of a high oesophageal stricture but there is a surprise. WHAT IS THE CAUSE OF THE STRICTURING?
■ Corrosive ingestion
Yes, a likely cause of 2 high oesophageal strictures
■ Peptic ulceration
In the high oesophagus?!?
■ Lichen planus
Possible but never seen two !
■ Eosinophilic oesophagitis
They are not short and membrane-like!
■ SCC
The mucosa is unremarkable!!!
explanation
The 'surprise finding' is of course that there are TWO strictures some 2-3cm apart in the high oesophagus. All of the above could be the cause, I guess. Of course, the stricture does not look malignant. Eosinophilic oesophagitis gives smoother, long tapering strictures, not short membrane-like strictures like this. Peptic ulceration would be unlikely in the high oesophagus (unless there is a long stretch of Barrett's below). Lichen planus is a possibility but the fact that there are more than one strictures, makes corrosive ingestion the most likely diagnosis. Actually, the patient had been admitted after swallowing a lot of toilet cleaner a few months earlier.
in the images below you can see what her oesophagus looked like after that alkaline burn. I wonder if giving a reducing dose of steroid, like we do after large oesophageal EMR's could have prevented her stricture?
This patient has recently undergone a bone marrow transplant when she develops abdominal pain. PPI therapy has not improved her symptoms and she referred for a Gastroscopy
WHAT IS YOUR ENDOSCOPIC DIAGNOSIS?
■ CMV infection
A good guess although there are no ulcers?!
■ HSV infection
Never seen it! Does it really happen?
■ C.diff infection
Definitely doesn't happen in the stomach!
■ Acute GVHD
Actually, you are not wrong!
■ Checkpoint inhibitor gastritis
Not as crazy as you think!
explanation
Well this is a striking appearance! I'd never seen anything like it and tentatively suggested HSV gastritis on the histology request form. I didn't consider CMV because there was no mucosal ulceration.
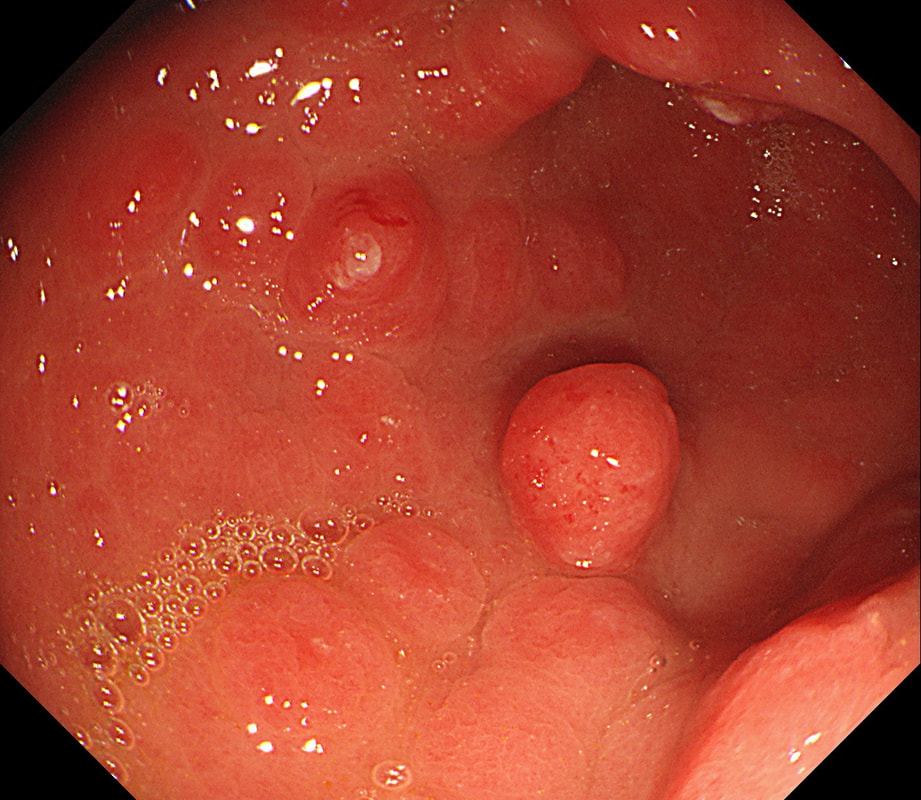
Histology confirmed finding lots of oedematous CMV infected cells with granulation tissue and there was no need for immunostaining for cytomegalovirus proteins. This is a case of CMV gastritis! Actually, there were also some features suggestive of acute GVHD and I can therefore not be entirely sure that these 'plaque-like appearance on the mucosa isn't due to the combined effect of CMV+GVHD+PPI! It's not uncommon, one study, reported that approximately one-quarter of patients who were unresponsive to first-line acute GVHD treatment, actually had coinfection with CMV [Bone Marrow Transplant. 47, 694–699]. Of course, immunosuppressants is the trigger for CMV reactivation but CMV gastritis can occur in apparently immunocompetent patients, although a link with occult cancer has been suggested [Scand J Gastroenterol 2011;46:1228–35]. My guess of HSV was of course unlikely as HSV infects squamous mucosa or the colon (surprisingly), not the stomach. In the oesophagus you may see vesicles and/or small ulcers (left when the vesicles rupture). If I had seen this appearance in the colon I would have called this 'pseudomembranous colitis but of course C.diff doesn't affect the stomach. Can you get gastritis with checkpoint inhibitors? Yes, you can! It can cause a severe, haemorrhagic gastritis as outlined in this article [Case Rep Oncol 2018;11(2):549–556]. There is more information on CMV gastritis at this site. These polyps were found in the stomach of a 40 year old man on a Barrett's surveillance programme WHAT WOULD YOU ADVICE?
■ We should eradicate your Hp
This patient is VERY unlikely to have H.pylori
■ We should remove these endoscopically
An unnecessary job unless one look wonky, like perhaps that single red one!
■ We should stop you PPI
But the patient has Barrett's ?!
■ We should stop you PPI
That's what I would do !
explanation
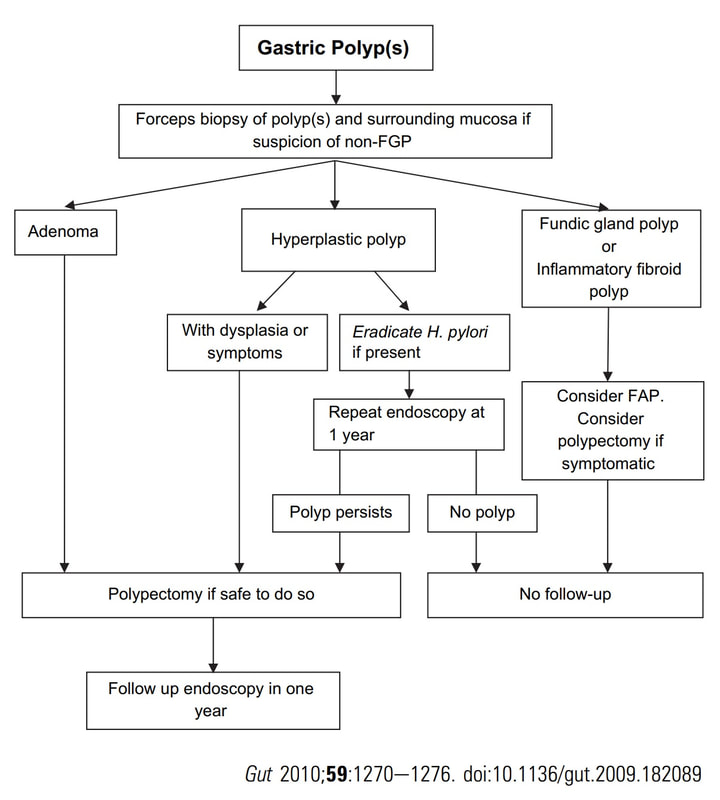
Of course, this is a Fundic Gland Polyps/Cystic Gland Polyps . They are usually multiple, somewhat transparent sessile polyps, usually in the 1-5mm in diameter and located in the body and fundus.
Histologically there are cystically dilated glands lined by gastric body type mucosa. Of course, there is no need to sample these. A study patients with FGPs reported that they can be diagnosed with a high degree of accuracy based on endoscopic appearance alone (J Clin Gastroenterol 2003;36:399-402). The sporadic polyps are caused by activating mutations of the beta-catenin gene. Not sure how an APC gene mutation (you may remember are involved in FAP), could give rise to FGP’s though! Furthermore, I don't know how the use of PPI's can be linked to the development of FGP's. Presumably its something to do with the prolonged hypergastrinaemia and enterochromaffin cell-like (ECL) hyperplasia? I have seen a couple of cases of cancer developing within a FGP. In both cases the patient had FAP and the polyps were markedly larger than all the surrounding cystic fundic polyps. There have been case reports of dysplasia arising within sporadic fundic gland polyps. A few series have reported a <1% risk of sporadic FGPs harbouring dysplasia (Am J Surg Pathol 1998;22:293-298, Eur J Gastroenterol Hepatol 2003;15:1153-6, Endoscopy 1995;27;32-37). I must admit that I don't believe the figure of <1% risk of dysplasia in a sporadic FGP. I suspect that these series consist of rather selected cases. Of course by only selecting polyps 10mm or larger, you would be filtering away 99% of the FGP's, leaving only the 'far above average risk lesions' behind. Nevertheless, the BSG recommend sampling some of them to confirm the diagnosis but not to set out on any attempt to remove them. Of course, this advice is different to the advice for Hyperplastic polyps which ARE usually linked with H.pylori and also a greater risk of developing cancer. The BSG advice is summarised in the algorithm below. Sporadic FGPs are not associated with either H.pylori gastritis or atrophic gastritis. In fact, it has been proposed that H. pylori infection may have an inhibitory effect on the development of FGPs. Of course, you wouldn't stop the PPI in a patient with Barrett's. Actually there is no evidence that patients with unstable Barrett's are any more or less likely to progress to cancer if they stop their PPI. However, there must be a point (I presume) when the use of PPI is beneficial in the 'evolution' of Barrett's. Are patients on a PPI less likely to develop Barrett's or to develop damage the stem cell DNA? Not sure! The BSG do recommend a colonoscopy in patients below the age of 40 yrs. to rule out the possiblity of FAP. However, as the average age of colorectal cancer in patients with 'attenuated FAP' is 55 yrs, I would personally recommend a colonoscopy in patients above the age of 40 yrs. This patient is undergoing a set of oesophageal dilatations of a radiotherapy induced stricture. Three set of samples, a total of 18 biopsies have been obtained from the stricture which have all been reassuring. A CT has reported: "Distal oesophageal mural thickening extends throughout the length of the previously demonstrated bulky oesophageal tumour. No focal abnormality to suggest recurrent disease and presumably appearances are all radiotherapy related". In the video clip, the stricture is seen to be dilated to 20mm. WHAT WOULD YOU DO NEXT
■ Organise the next dilatation in a weeks time
She WILL need more dilatations but you've missed something important!
■ Take another set of samples from that stricture
It does look concerning still but there is something even more concerning here!
■ Take another set of samples from the more proximal oesophagus
You've spotted it!
■ Take samples at the distal oesophagus (below the stricture)
That odd, fleshy lump at 6 O'clock below the stricture has been sampled already. More samples is never wrong but there is something even more important to sample first!
explanation
On intubation, I glimpsed something just below the upper oesophageal sphincter. I deliberately didn't dwell on it in the video, simulating the previous intubations when a synchronous lesion had been missed. In the explanatory clip below, you get a better view of her second SCC. CT staged the second lesion as no worse than T2, N0.
The moral of the story? It's when we are distracted that we run the risk of missing things! In this case, the previous endoscopists had approached the endoscopy as a case of dilatation and switched off their eyes! This is the GOJ of a 60 yr old woman with dyspepsia. WHAT IS YOUR DIAGNOSIS?
■ Reflux oesophagitis
No this is a nodule not an ulcer!
■ Hyperplastic/reactive nodule
Hyperplastic nodules have villous surface!
■ Unremarkable Barrett's
A nodule within Barrett's is always suspicious!
■ Adenocarcinoma
Absolutely!
■ Squamous cell carcinoma
No abnormal vessel pattern within nearby squamous mucosa!
Explanation
Actually, this patient was on a Barrett's surveillance programme. Five years earlier a small IMca had been removed following which annual surveillance had been reassuring. However, then this lesion was found and referred for removal.
Histological assessment of the resection fragment reported early invasion into the submucosa (which is 'allowed' provided that the depth of invasion is less than 500 microns (0.5mm). There was no LVI but unfortunately, there was poor differentiation. Of course, the 'worst' feature to find is probably LVI. Poor differentiation is worrying but less worrying than LVI. The reason for this is that 'poor differentiation' is a rather poorly defined entity. For example, how many crypts should be involved to call something 'poor differentiation' rather than 'focally poor differentiation'? Anyway, the patient was young and ultimately underwent an Ivor-Lewis which confirmed that the EMR had been curative with only some HGD remaining within the residual short stretch of Barrett's. We urgently need a prospective study following this type of Barrett's patient closely over time with regular EUS and CT to see if we can detect the small number of patients who subsequently turn out to have lymphnode involvement
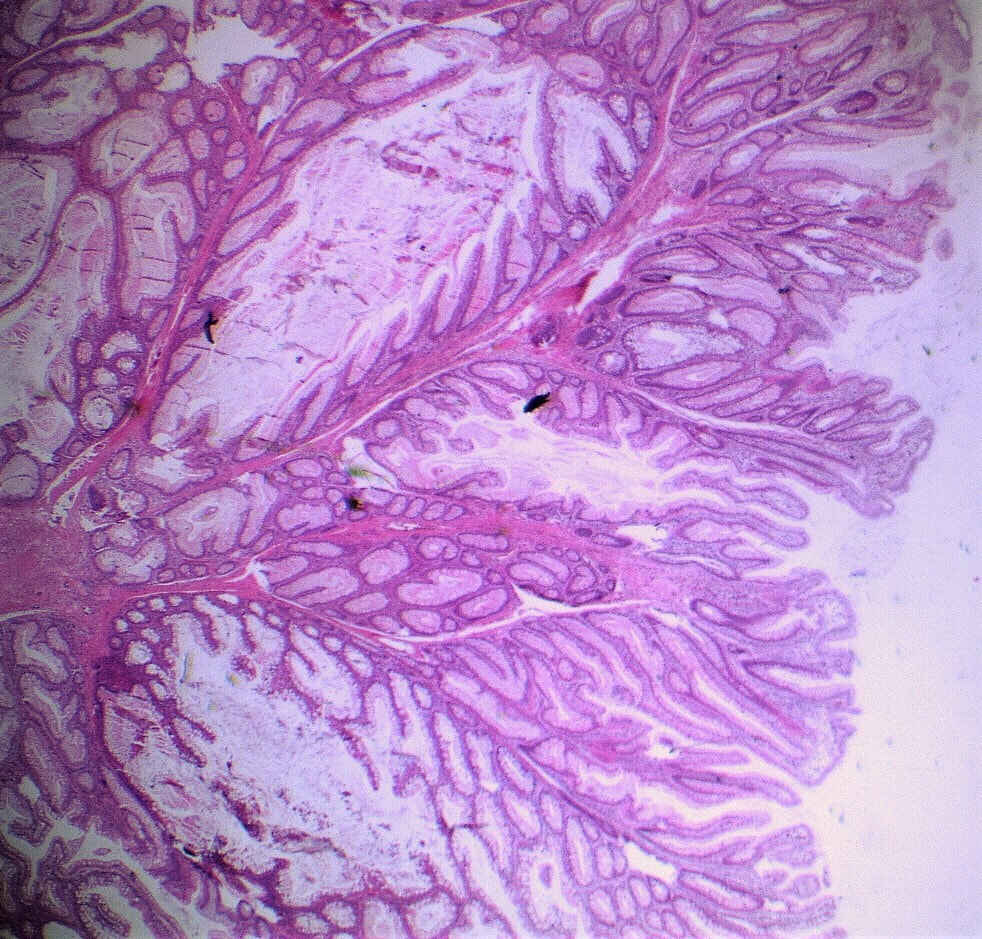
A middle-aged woman presented with loose stool and weight loss. Initially, she refused an endoscopy and a diagnosis of coeliac disease was based a high tissue transglutaminase (tTG) antibody titre. She was started on a gluten-free diet (GFD) but her symptoms remained despite adherence to the GFD. After several months she agrees to undergo an endoscopy. The images show the endoscopic view of the duodenal mucosa and the corresponding histology slide stained with haemotoxylin and eosin (H&E).
WHAT WOULD YOU DO NEXT?
■ Refer the patient for a dietary review
You are assuming that the pt has Coeliac disease...
■ Request a clonal analysis of the intraepithelial lymphocytes
But is the diagnosis of Coeliac correct? Look at the histology again!
■ Refer for HLA DQ2/DQ8 testing
But the diagnosis has been made on the biopsy...
■ Refer the patient for capsule endoscopy
Tissue was the issue and you've got that already!
■ Add prednisolone to the dietary restrictions
Congratulations you spotted the collagen band?
explanation
Initially, the patient appeared to have classic coeliac disease, with diarrhoea and weight loss together with a positive serology. The endoscopy was crucial as the duodenal biopsies identified the presence of a thick band-like deposit of collagen just below the duodenal epithelium. On the basis of this finding, collagenous sprue was diagnosed.
Collagenous sprue was first described in 1947,1 but it was not until 1970 that Weinstein et al. introduced it as a diagnostic term to the medical nomenclature.2 Collagenous sprue is more frequent in females and in individuals who have other autoimmune diseases.3 It is now recognised that collagenous sprue shares similar clinical features with coeliac disease, such as chronic diarrhoea, anaemia and weight loss. In addition, the endoscopic and histological features of both diseases are similar, with an atrophic and scalloped duodenal mucosa. However, the histological hallmark of collagenous sprue is the presence of a thick subepithelial collagen band. Such collagen bands may also be found in collagenous gastritis and collagenous colitis.4 It has been proposed that collagenous sprue may be a heterogenous condition of collagenous gastroenteritides, including conditions such as collagenous colitis and coeliac disease.5,6 Unlike coeliac disease, in collagenous sprue, the typical histological changes may also be found in the stomach and colon.7 Furthermore, greater numbers of IgG4 plasma cells have been reported in the duodenal mucosa of patients with collagenous sprue when compared with the numbers in patients with coeliac disease, duodenitis or normal duodenal mucosa.8 As in our case, patients with collagenous colitis usually also have positive coeliac serology. For this reason, some believe that the collagen band is simply a marker of particularly severe coeliac disease, which may also be associated with ulceration, perforation and T-cell or B-cell lymphoma.9 An interesting report of paraneoplastic collagenous colitis was reported by Freeman et.al.10 In this case a patient with collagen deposits in both the small and large intestine was also found to have a coincidental colon cancer. After surgery, both the malabsorption and the histopathological changes completely resolved! For this reason, a search for underlying malignant disease should be considered. The response to a GFD is usually disappointing and for this reason, the outlook used to be grave for patients with collagenous sprue. However, remission of the condition has been reported with courses of corticosteroids11 or immunosuppressive agents such as infliximab.12 References
This patient presented with an emergency GI bleed. WHAT IS THE DIAGNOSIS?
■ Benign gastric ulcer
Hmm, multiple GU's ?!
■ Ulcerated GIST
Multiple ulcerated GIST's. Really?
■ Adenocarcinoma
Multifocal adenocarcinoma ?!
■ Lymphoma
Too crater-like !!!
■ Melanoma
Got it!
explanation
There are actually several odd lesions in the stomach. Apart from the 'multi-focality', the lesions look a little like craters on the moon. Of course, this is what metastatic disease looks like. The dark colour clinches the diagnosis of melanomatous deposits. Here is a link to Sanjeev's and Ashok's case published in Digestive and Liver Disease [2020;52(12):1512] which is an even better demonstration of that dark pigmentation. Malignant melanoma has a peculiar predilection for the gastrointestinal tract. Melanoma can spread anywhere and is the most common metastatic tumor of the GI tract. More often the haematogenous spread is to the small bowel or colorectum. Gastric metastases are uncommon. It’s one of the more common findings in the small bowel of younger patients with anaemia. In an old post mortem series, the prevalence of GI metastasis in patients with melanoma was only 0.9% in 1000 melanoma patients (Arch Surg 1964:88:969-973). However, when you look at cases with advanced disease, up to 50% will have GI metastases (Cancer 1964;17:1323-39). As one may expect, the most common presentation is with anaemia or obstructive symptoms. Interestingly, surgical resection has been linked with a longer survival! Of course, its important to distinguish between a primary GI mucosal melanoma and metastatic melanoma. The criteria for a diagnosis of primary intestinal melanoma include 1) no evidence of concurrent melanoma or atypical melanocytic lesion of the skin, 2) absence of extra-intestinal metastatic spread of melanoma, and 3) presence of intramucosal lesions in the overlying or adjacent intestinal epithelium In a small series, the 5 year survival was 38% (Arch Surg 1996;131 :975-980). Without surgical intervention, the prognosis used to be grim and Amer et al. (Gynecol Obstet 1979;149:687-92) reported a mean survival time of 9.7 months, whilst others have reported an even shorter median survival of 4.7 months. Nevertheless, it is perhaps likely that case selection is the reason for the apparent survival benefit of surgery. The new checkpoint inhibitors and other immunotherapies are revolutionising the treatment of advanced stage disease.
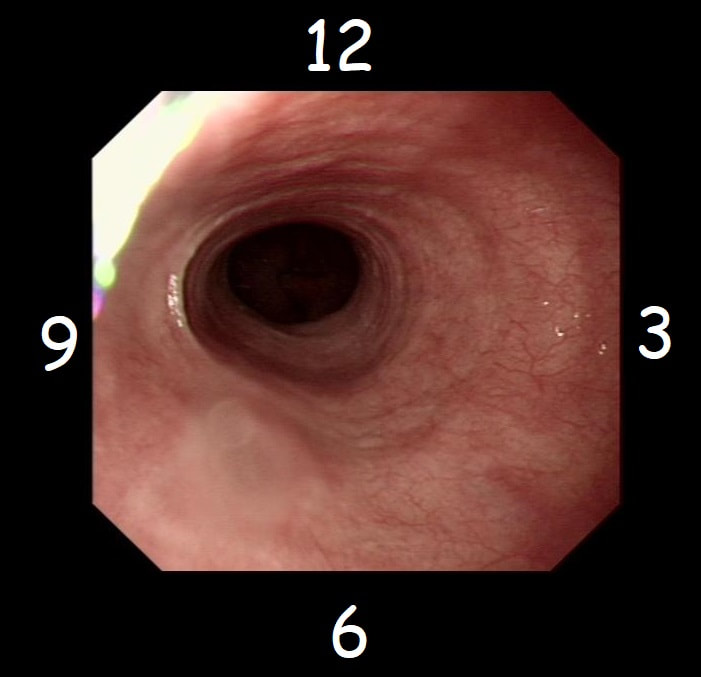
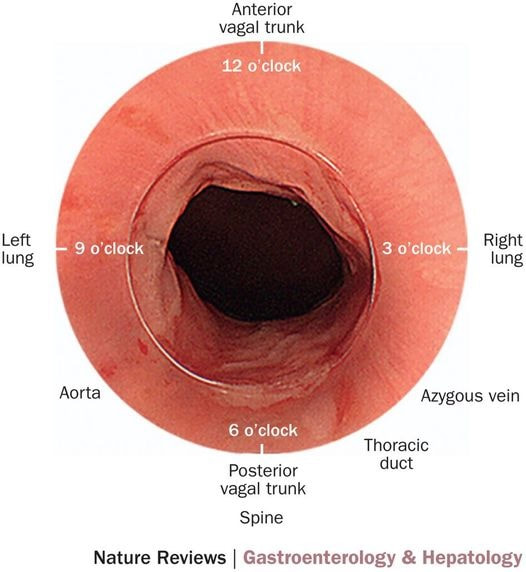
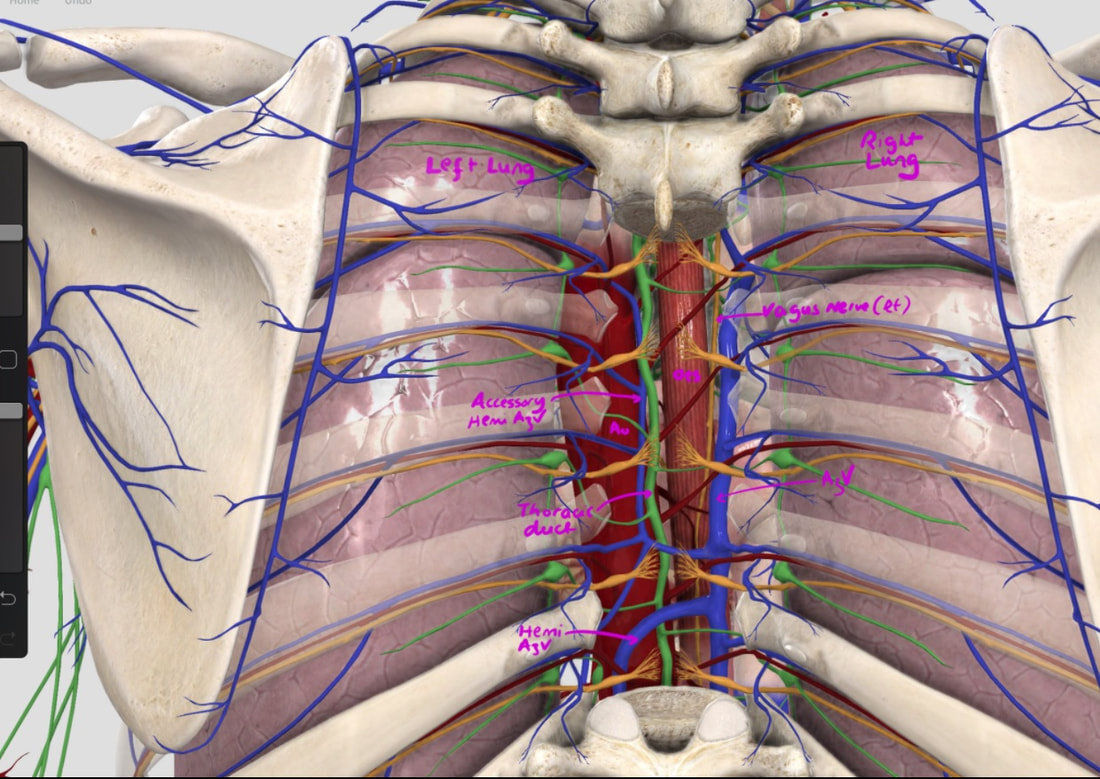
With POEM, knowing the landmarks outside of the oesophagus has become something which endoscopists are expected to be familiar with. Here is a quick self-test!
AT WHICH POSITION OF THE 'CLOCKFACE' DO YOU FIND THE:
■ Spine
6 o'clock
■ Anterior vagal trunk
12 o'clock
■ Posterior vagal trunk
6 o'clock
■ Thoracic duct
5 o'clock
■ Left lung
9 o'clock
■ Aorta
7-8 o'clock
■ Azygous vein
4 o'clock - runs in the posterior mediastinum, on the right side of thoracic vertebral column. It drains into the SVC, and returns blood from the posterior walls of the thorax and the abdomen.
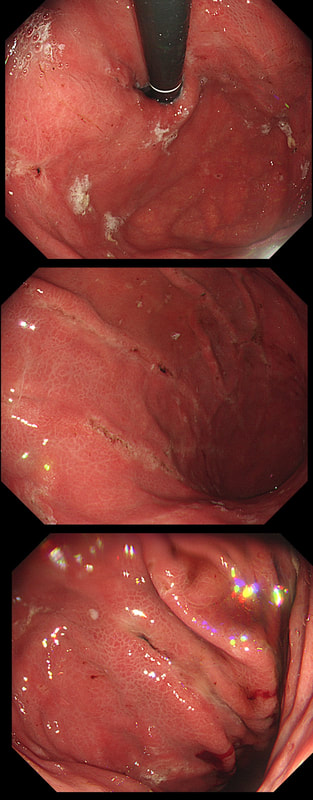
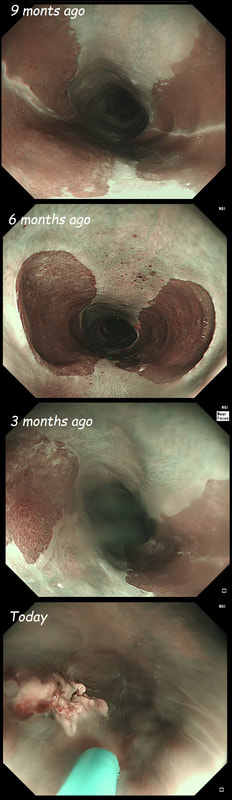
This 50 year old lady presents with dysphagia and a distal oesophageal stricture is confirmed. Intubation is a little tricky due to a pharyngeal pouch but we managed to intubate the oesophagus after the successful passage of a guidewire. After you have obtained a set of samples (last photo), the patient asks what will happen next.
WHAT IS YOUR REPLY?
■ Too early to tell, we will await analysis of the samples first
It's not too early to tell - the signs are there!
■ Whilst we await histology, I will request at CT
Good man! And organise a cancer MDT discussion!
■ The analysis will probably be reassuring and in all likelihood a dilatation is next
Sadly, this is unlikely !
■ The pouch is more likely to be the main problem than the stricture
No, that's not it!
explanation
The stricture doesn't look quite right does it? Somewhat 'nobbly' and without associated reflux oesophagitis. This stricture looks malignant ! There is one more concerning feature which you may have missed...
The vocal cord on the left-hand side is weaker than the right! On direct questioning, the patient confirmed that her voice had recently changed and become more hoarse! This patient had an advanced oesophageal cancer with metastases to the high lymphnodes on the left causing a recurrent laryngeal nerve paralysis! Out of the four options, organising a staging CT and referral to the UGI cancer MDT is the correct course of action! This patient has been referred for an oesophageal dilatation after biopsies have been reassuring WHAT WOULD YOU DO?
■ Abort and take more samples
You are a wise as a fox!
■ Dilate to 12mm
Hmm, cautious but not cautious enought !
■ Dilate to 15mm
Seems reasonable but you've missed something!
■ Dilate to 18mm
You are clearly brave but look again!
■ Dilate to 20mm
If this stricture was benign, I would also aim for 20mm but it isn't!
explanation
Of course this stricture doesn't look right! There is a peculiar plaque-like area in the 10 O'clock position. The dilatation was cancelled and another set of samples were requested together with a CT. Samples indicated that this was a case of poorly differentiated intramucosal cancer and an EMR was organised. Have a look at the clip below. Clearly this lesion is firmly tethered to underlying structures and is beyond endoscopic cure. Some options may pop into your head such as injecting some fluid below the lesion or using the 'pull-within-the-snare' EMR technique. The problem is that these will not change the basic fact that endoscopically the lesion is beyond endoscopic cure! You run the risk of 'muddying the waters' with scrappy histology reporting 'intramucosal cancer at least' and with uncertain margins. Far better is to recognised the endoscopic irresectability of the lesion which provides a clear steer towards the 'next treatment level'. In this case the patient wasn't a surgical candidate and was offered chemo-radiotherapy (CRT).
The squamous portion of this oesophagus looks very unusual !
WHAT IS THE LIKELY REASON FOR THIS APPEARANCE?
■ Previous reflux disease
Nope, that's not it!
■ Previous EMR
Gives a pale scar, usually covered with squamous mucosa
■ Previous RFA
Absolutely. You've also seen it?
■ Previous radiotherapy
Gives telangioectatic vessels!
explanation
This patient had undergone RFA, leaving the squamous mucosa looking a little odd. It's actually the openings of the 'oesophageal glands' which you can see. In my experience, there is no point in taking any samples as histology will be reported as normal. In most cases, Barrett's returns at the gastro-oesophageal junction. The return is slow and subtle and for this reason you should take samples from what appears to be the 'columnar side' of the squamo-columnar junction. As intestinal metaplasia (Goblet cells to you and me) are common at the cardia, the pathologists will not be able to tell you if the Barrett's is returning. However, they CAN tell you if the epithelium look dysplastic again!
In a minority of cases, perhaps 1:4 the recurrence happens below the squamous mucosa which can be very hard to see. Of course, the only way to deal with 'buried Barrett's', is to remove the patch by EMR!
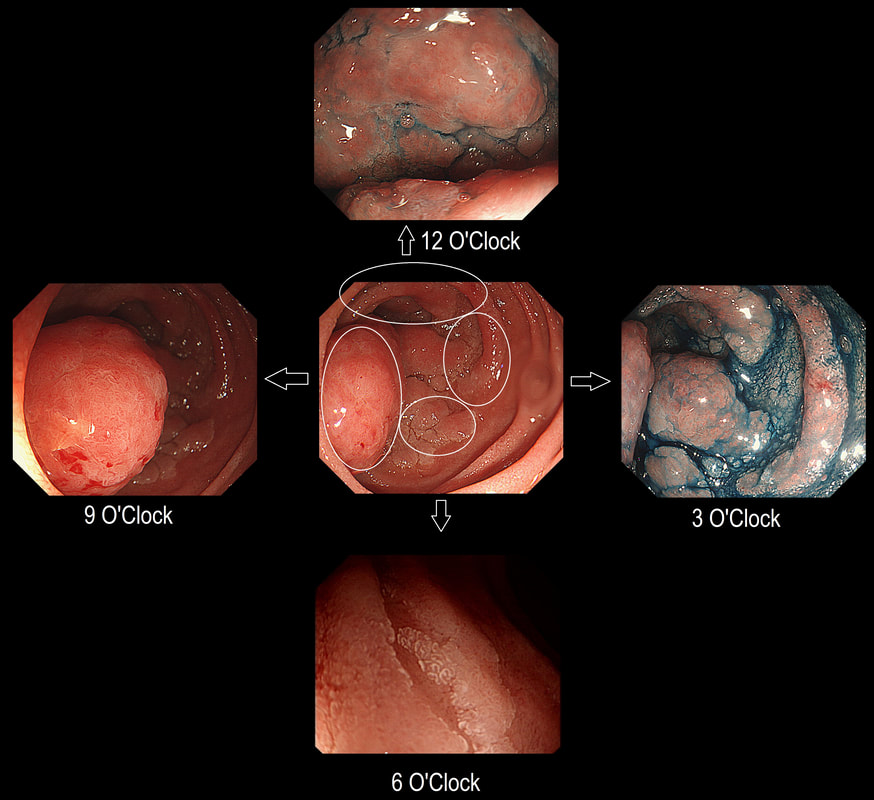
Here is a circumferential duodenal polyp situated in the second part of the duodenum. The patient is somewhat uncomfortable and your 'window of opportunity' is closing.
WHERE SHOULD YOU TARGET YOUR BIOPSY?
■ 12 O'clock
Looks amourphous and worrying but not the right answer
■ 3 O'clock
Wouldn't be my priority site
■ 6 O'clock
This is the least worrying spot!
■ 9 O'clock
Absolutely - for two reasons!
explanation
In general ampullary or peri-ampullary adenomas are far more likely to turn malignant than adenomas situated elsewhere in the duodenum. This is the first reason, why you should target your biopsy at 9 O'clock. There is a second reason and that is that the most 'chunky' polyp is at 9 O'clock. In my experience, the larger the 'polyp volume', the greater is the risk of malignant conversion.
So should we offer an ampullectomy? I would offer this to anyone with a life-expectancy of 10 years or more, I would consider (no promises) anyone with a life-expectancy of 5 years or more IF there is HGD on biopsies and there is no evidence of ductal obstruction (pt never had jaundice and there is no ductal dilatation or MRI or on EUS). Of course, an ampullectomy (probably better called a 'papillectomy') is dangerous but less dangerous than surgery. In Leeds we quote the following risks of a papillectomy: 1:10 risk of acute pancreatitis for up to a few months after the procedure, 1:10 risk of late bleeding for up to 2 weeks after the procedure, 1:50 risk of a perforation, a 1:10 risk of late papillary stenosis, 1:20 risk of acute cholangitis and a 1:200 risk of death Tiger country!!!
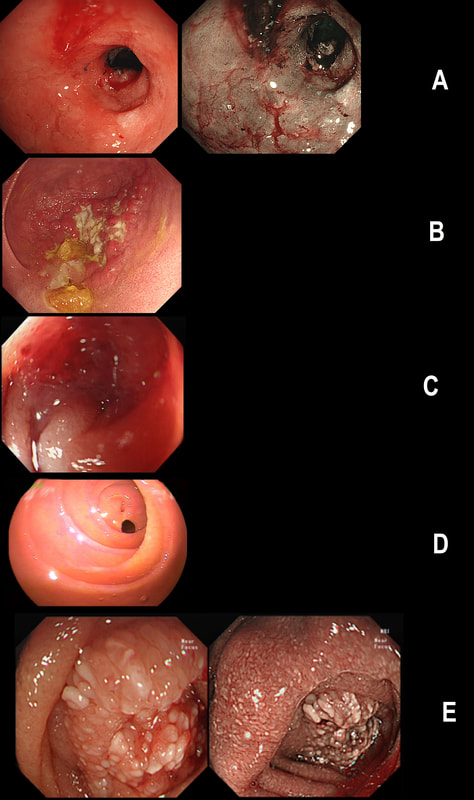
Just another piece of fun ☺! Here are 5 photographs of duodenal strictures, each of a different aetiology!
CAN YOU MATCH THE STRICTURE WITH THE PHOTO?
■ NSAID induced stricture =
Photograph D
■ Peptic stricture =
Photograph A
■ Chronic pancreatitis =
Photograph E
■ Chrohn's disease =
Photograph B
■ Duodenal adenocarcinoma =
Photograph C
EXPLANATION
You'll know that NSAID induced strictures are classically 'membrane-like' as in Photograph D.
The 'Peptic stricture' is shown in photograph A. The healed duodenal ulcer had been at 9 O'clock in that stricture. Chronic pancreatitis causes external compression of D1 or D2 usually with swollen villi. Of course there are two photographs to choose between - Photo C or E. Lets leave that one and see if the Crohn's stricture is easier to find. Of course, photograph B shows ulceration and inflammation and must be the CD stricture. That leaves the malignant stricture which could be photograph C or E. It's usually relatively easy to squeeze past the external compression caused by a swelling in the head of the pancreas. In contrast, malignant duodenal strictures are usually impassable. Which one looks the tightest? Photo C or E? Well, C is the malignant stricture which I had to dilate up to 10mm before I could take samples a little deeper into the stricture to confirm the diagnosis. Photograph E was a case of severe pancreatitis which we scoped as he had dropped his Hb (never found out why or how). This video clip requires no introduction. No other abnormality was found. WHERE IS THE LIKELY SITE OF THE PROBLEM?
■ in the portal vein
Then you would expect BOTH oesophageal and fundal varices!
■ In the hepatic vein
In Budd-Chiary, there are no varices!
■ In the splenic vein
Isolated splenic vein varices - absolutely correct!
explanation
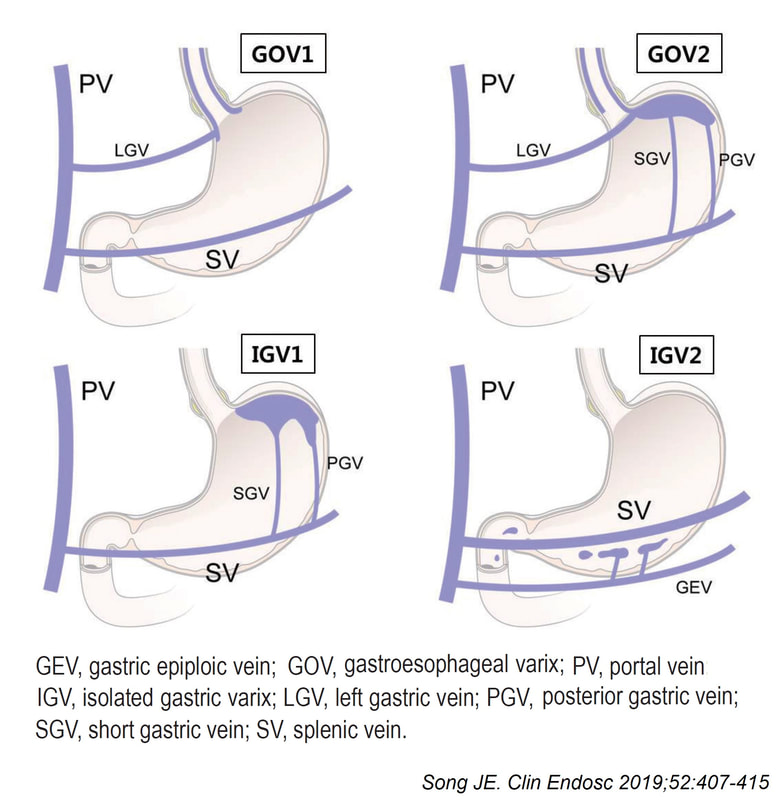
Quoting the 'Sarin Classification' of gastric varices, this is a case of an 'isolated fundal varix' (IGV1). In this case the underlying cause was splenic vein thrombosis following an admission for acute on chronic pancreatitis. The precise distribution of the varices helps you to pinpoint the location of the thrombus. The infographic below from Song et.al is brilliant:
Briefly, 1) oesophageal varices with or without fundal varices points to portal vein thrombosis 2) fundal varices ALONE points to splenic vein thrombosis 3) antral and/or duodenal varices points to a thrombus in the gastro-epiploic vein Brilliant !
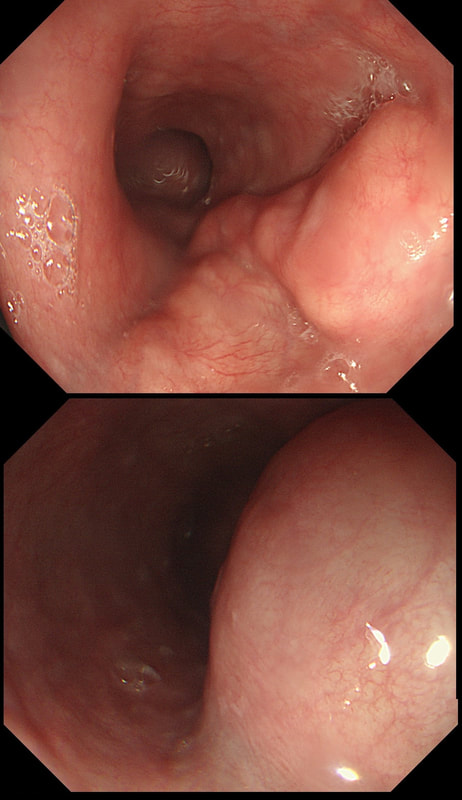
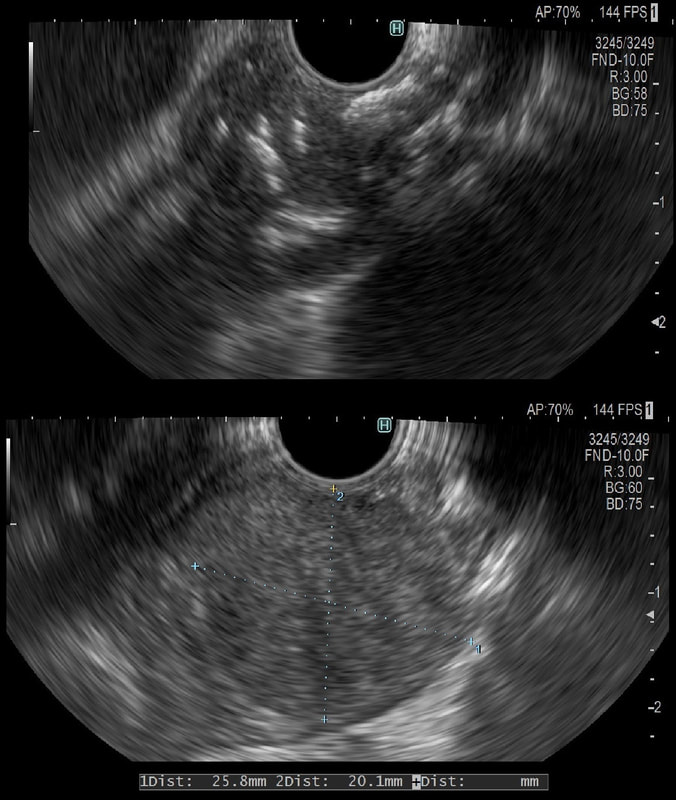
This was an incidental finding in the mid-oesophagus. Subsequently an EUS is organised and the lesion is ultimately removed (histology in the last image)
WHAT IS THE DIAGNOSIS?
■ Neuroendocrine tumour
I've never actually seen an oesophageal NET
■ Granular cell tumour
But it isn't a small white submucosal disc?!
■ Leiomyoma
A shrewd guess!
■ GIST
These are VERY rare in the oesophagus (but common in stomach)
■ Melanoma
Usually look brownish endoscopically!
explanation
Leiomyomas are the most common submucosal lesion found in the oesophagus. No idea why they are so rare in the stomach, where GIST's are the most common submucosal lesion. Correspondingly GIST's are very rare in the oesophagus.
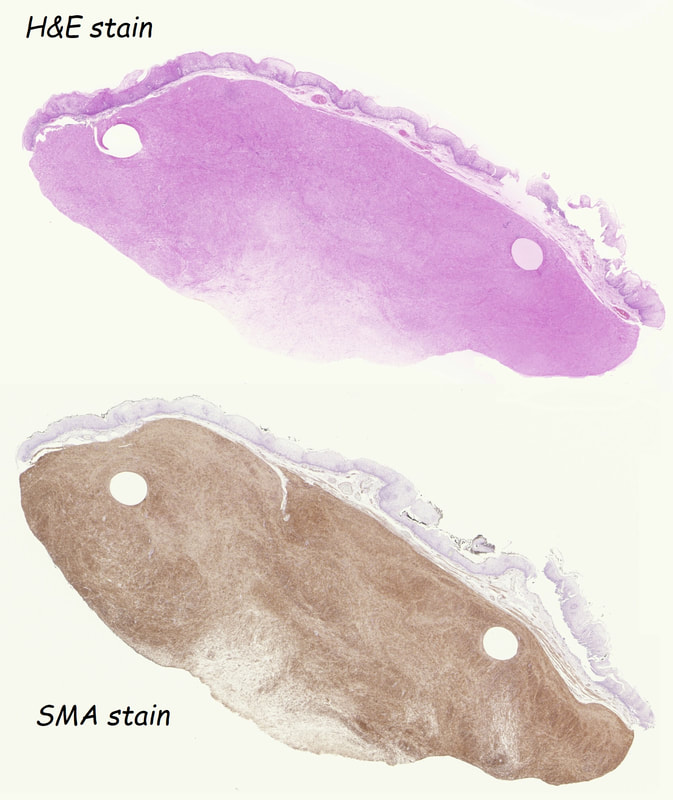
These usually arise from the circular muscle propria layer. On CT they are often calcified and there are of course no nearby nodes to suggest malignant disease. Indeed, malignant transformation is extremely rare and for this reason many centres would ignore a small submucosal lesion such as this, when found in an elderly person. Larger (around 5cm usually) and symptomatic lesions (which are more likely to be leiomyosarcomas) are enucleated surgically. "- They shell out like peas" according to one of our UGI surgeons. However, our local surgeons are also careful to point out that resection usually result in some damage to the muscle propria layer, which require repair as otherwise a diverticulum may develop. With endoscopists spending more time in the 'third oesophageal space' (i.e. the submucosal space), doing ESD's and POEM, the suggestion that we should also remove leiomyomas endoscopically has arisen. May seem like a good idea but I would advocate caution for the simple reason that endoscopically, we are unable to repair the defect we cause in the muscle propria layer ... Oh, how about that 'SMA stain' used in the histology. This is 'Smooth Muscle Actin' stain which does exactly what it says on the tin ...
This patient originally presented with a variceal bleed and was managed on ITU. Investigations confirmed alcoholic liver disease and the patient is entered onto a variceal ablation programme. At the first outpatient endoscopy, this is noted
WHAT IS THE LIKELY DIAGNOSIS?
■ Vocal cord granuloma
Looks like granulomatous tissue!
■ Mucus retention cyst
It doesn't look cystic !
■ SCC
Unlikely as wasn't noted at intubation
explanation
This is a vocal cord granuloma likely caused by irritation from the ET tube during the recent admission. Other causes include 'voice abuse', severe cough, vomiting, habitual throat cleaning, and the use of irritants such as tobacco and alcohol. Mucus cysts rarely develop on a vocal cord and look cystic rather than as granulomatous tissue as in this case.
Initially, management is supportive as these often shrink within 6-12 weeks. Surgery or laser ablation is reserved for large lesions but is a little fraught is these tend to recur unless to underlying cause is addressed. Singers should be advised to warm up the voice before singing (or speaking), resting the voice in anticipation of future speaking obligations and to avoid singing at times of an upper respiratory infection. This excellent photograph of a vocal cord granuloma was actually supplied by Dr Muslim Alkafaji who has been a member of Friends of Endoscopy since 2014 !!! This patient has been referred for a dilatation WHAT IS THE AETIOLOGY OF THE STRICTURE?
■ Peptic stricture
Too membrane-like to be peptic
■ Schatzki ring
Of course it is - Well Done!
■ Eosinophilic oesophagitis
Strictures in EoE are long and tapered
■ Radiotherapy stricture
RXT strictures are at least a couple of cm in length
■ Malignant stricture
Of course it isn't !
explanation
This short almost membrane-like stricture is of course a Schatzki ring, named after the German-American physician Dr Richard Schatzki. It's usually easy to treat these with a balloon dilatation. This particular case is unusual as two dilatations were required. However, rather than going ahead with a second dilatation to 20mm (our largest balloon in Leeds), I decided to cut the membrane with the tip of a snare and the diathermy set on the normal sphincterotomy setting.
The uploaded video is a good example of this mishap when cutting across yourself. I don't think that I perforated the full thickness of the oesophagus but applied a clip just in case (I've never regretted placing too many clips).
A rather beautiful polyp found in the duodenum of a patient undergoing upper GI surveillance
WHAT IS THE LIKELY HISTOLOGY?
■ Inflammatory/reactive polyp
Looks too pretty to be inflammatory
■ Serrated polyp
Surprisingly, you don't get SSL's in the UGI tract
■ Hamartomatous polyp
Well done! These are usually pretty polyps!
■ Adenomatous polyp
If patient had FAP, there should be more small polyps about
■ Neuroendocrine tumour
Should have an indentation and no crypt pattern
explanation
This polyp doesn't look adenomatous. Remember that in the duodenum, the adenomatous polyp are usually flat, often with white white edges and with a slit-like or gyrate crypt pattern. Of course, in this case the crypt pattern is gyrate, almost exaggerated gyrate! The rather beautiful surface rules out an inflammatory polyp and an NET, both of which don't have this pretty surface structure. Even the histology is rather beautiful, see example below!
In addition, as you probably know, most duodenal NET's will have a central indentation and will not have a distinct crypt pattern. Cutting to the chase, this patient has Peutz-Jeghers syndrome ! This is a good example of the hamartomatous polyps these patients develop throughout their GI tract. Similar hamartomatous polyps also develop in the 'Cronkhite-Canada syndrome and Juvenile polyposis. Hamartomatous polyps are characterized by 'disorganized overgrowth' of the normal tissue but without visible cellular instability (dysplasia). Of course, these patient have an appalling lifetime risk of developing cancer which can be as high as 90%, including gastric, colorectal, breast, colorectal, and pancreatic malignancies. Unfortunately, there is no no evidence that removing hamartomatous polyp will reduce the subsequent risk of cancer. For this reason the only justification for attempting the somewhat hazardous endeavour to remove PJS polyps deep within the small bowel is to reduce the risk of subsequent intussusception. Rather surprisingly, a quick medline search has not revealed any RCT evidence that this actually is worthwhile !
These are the NBI images of a young woman complaining of chest pain, dysphagia and reflux symptoms. She also complains of feeling of a 'lump at the back of her throat'.
WHAT WAS THE ORIGINAL DIAGNOSIS?
■ Reflux oesophagitis
Would be an odd pattern surely?
■ Barrett's oesophagus
With squamous mucosa below?
■ Eosinophilic oesophagitis
EoE doesn't look brown on NBI !
■ Oesophageal inlet patch
Yes, but why is it getting smaller?
■ Squamous dysplasia
No dilated IPCL's !!!
explanation
This patient was found to have a large oesophageal inlet patch at the index endoscopy a year ago. Although his symptoms were rather mixed, we have spent 4 sessions on ablating her inlet patch with APC.
Of course, how often you find these will depend on whether you use NBI/FICE/BLE during extubation. Of course you should do because you are then less likely to miss squamous dysplasia! An interesting study reported that gastric inlet patches contained glucagon cells which would normally only be found in the embryo and subsequently disappear. To remind you, the hormone glucagon is normally produced by the alpha cells of the pancreas! Glucagon-like peptide-1 is also secreted by some cells in the ileum and colon. Anyway, the implication is that the gastric inlet mucosa is actually an embryonic left-over. Interestingly, there appears to be a lower prevalence reported in older populations, and some authors suggest that inlet patches may regress with age! Seems odd for a congenital condition? Endoscopic studies have found inlet patches in up to 10% of patients. Whether these give rise to any symptoms is contentious. It has been suggested that inlet patches may give rise to globus sensation due to irritation of the upper oesophageal sphincter by acid or mucus. Furthermore, others have suggested that H. pylori colonization of the heterotopic gastric mucosa causes symptoms. Others believe that it's not just the upper oesophageal sphincter which is abnormal but the entire oesophageal motility may be disorganised in these patients. However, prospective studies have usually not found that patients in whom an inlet patch is found, present with any different symptoms compared to patient without an inlet patch. Of course as anywhere in the body, cancer may develop. A study in Digestive Diseases & Sciences [2018;63(2):452-65] reported on 156,236 gastroscopies and may have found 3 cases of malignant conversion. Assuming that 10% of their patients had an inlet patch, that would suggest that you would have to regularly surveil 500 patients with an inlet patch over many years detect 1 cancer. If you grab 500 random people off a street and scope them annually for 5 years, wouldn't you pick up more than a single early oesophageal cancer? Seems like that lemon is not worth the squeeze? Finally, how about our patient? Over the period of one year, we spent about one hour ablating and completely getting rid of that large inlet patch (well two inlet patches really). The patient reported that symptoms were much better for a couple of months but then returned as before. Of course as she no longer has an inlet patch, we can now conclude that it never did contribute to her symptoms. Must admit that I was sceptical from the start! Show me a prospective RCT with evidence that ablation provides a clinically significant improvement in patients globus symptoms for more than a few months! Until I see that RCT, I will not be offering ablation for these patients. |
Categories
All
|