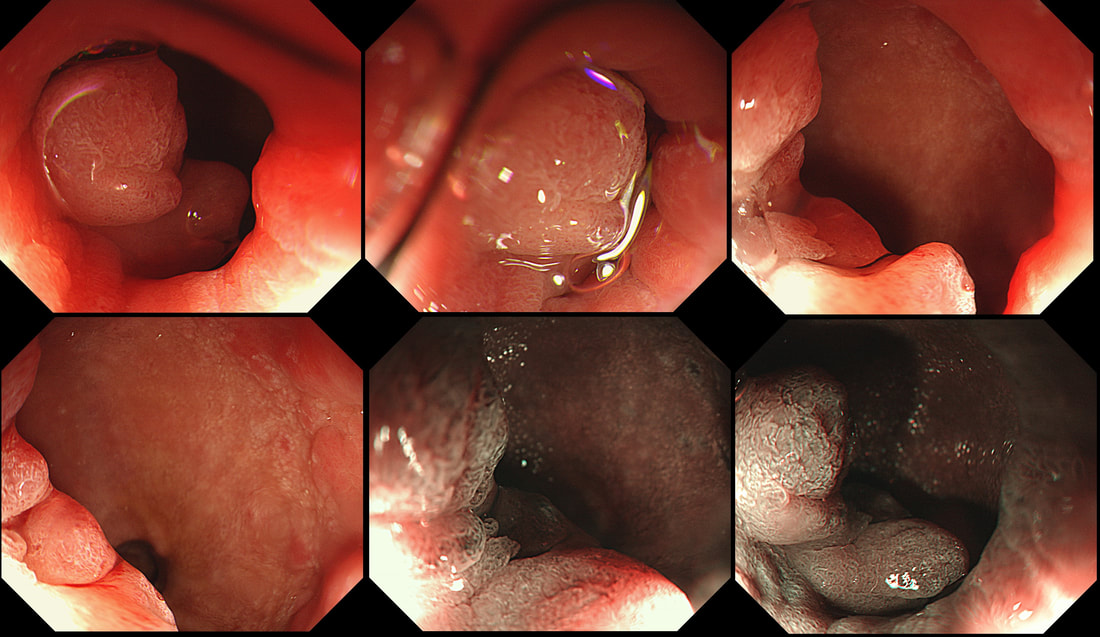
|
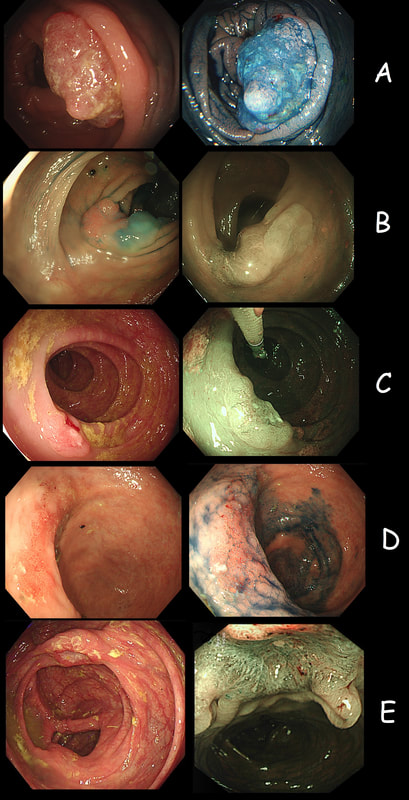
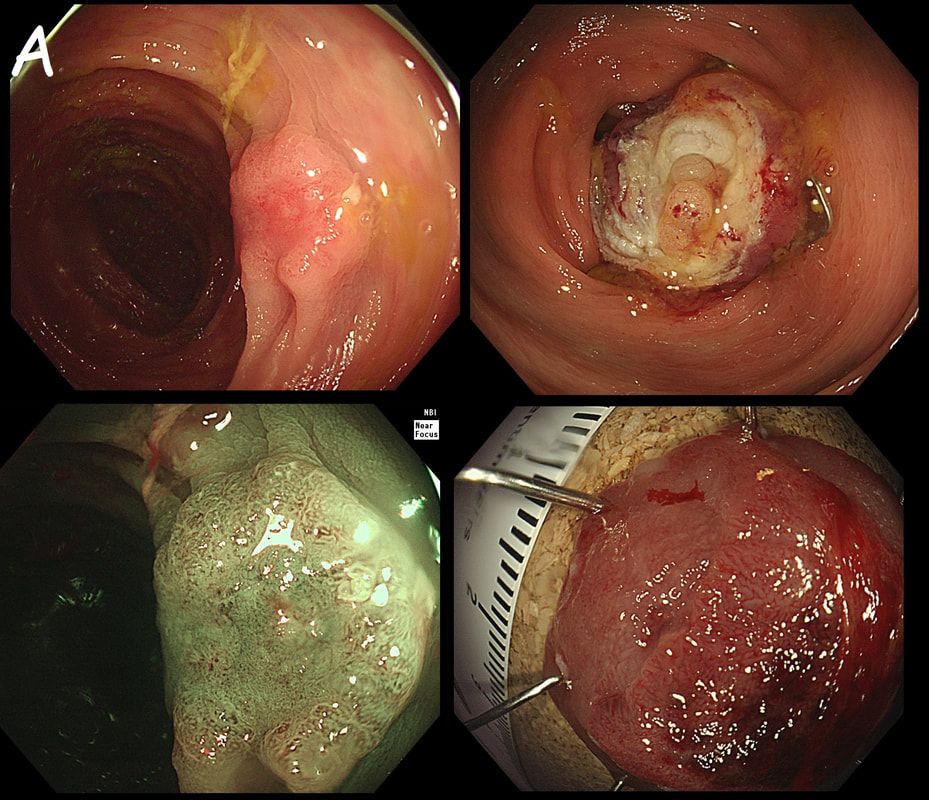
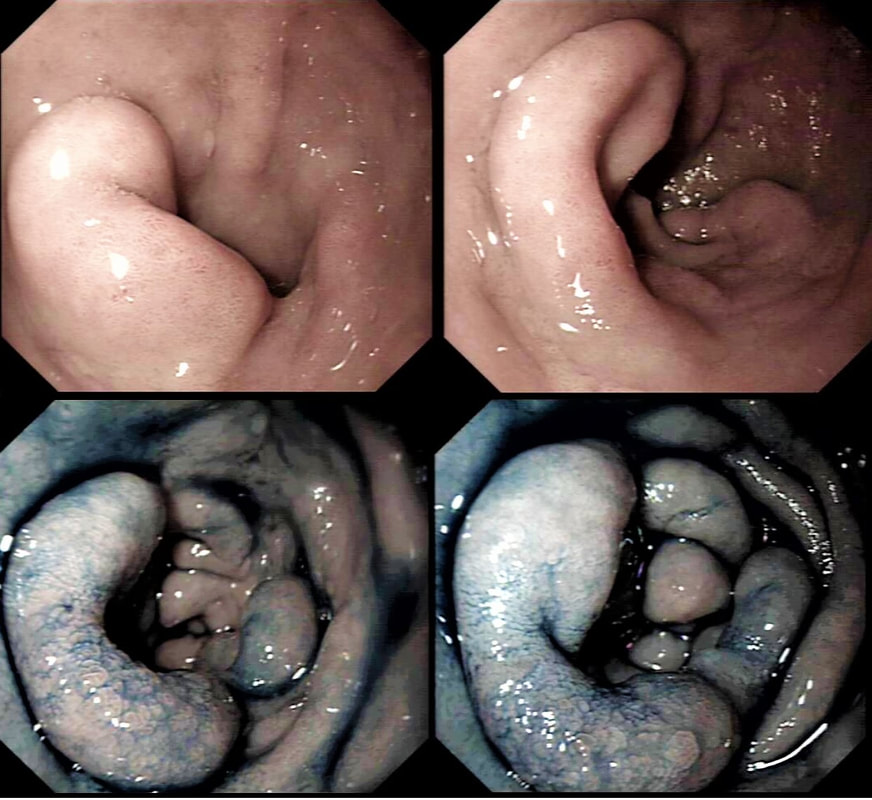
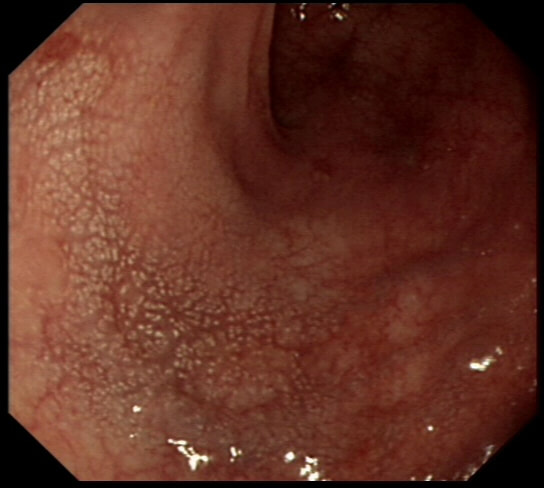
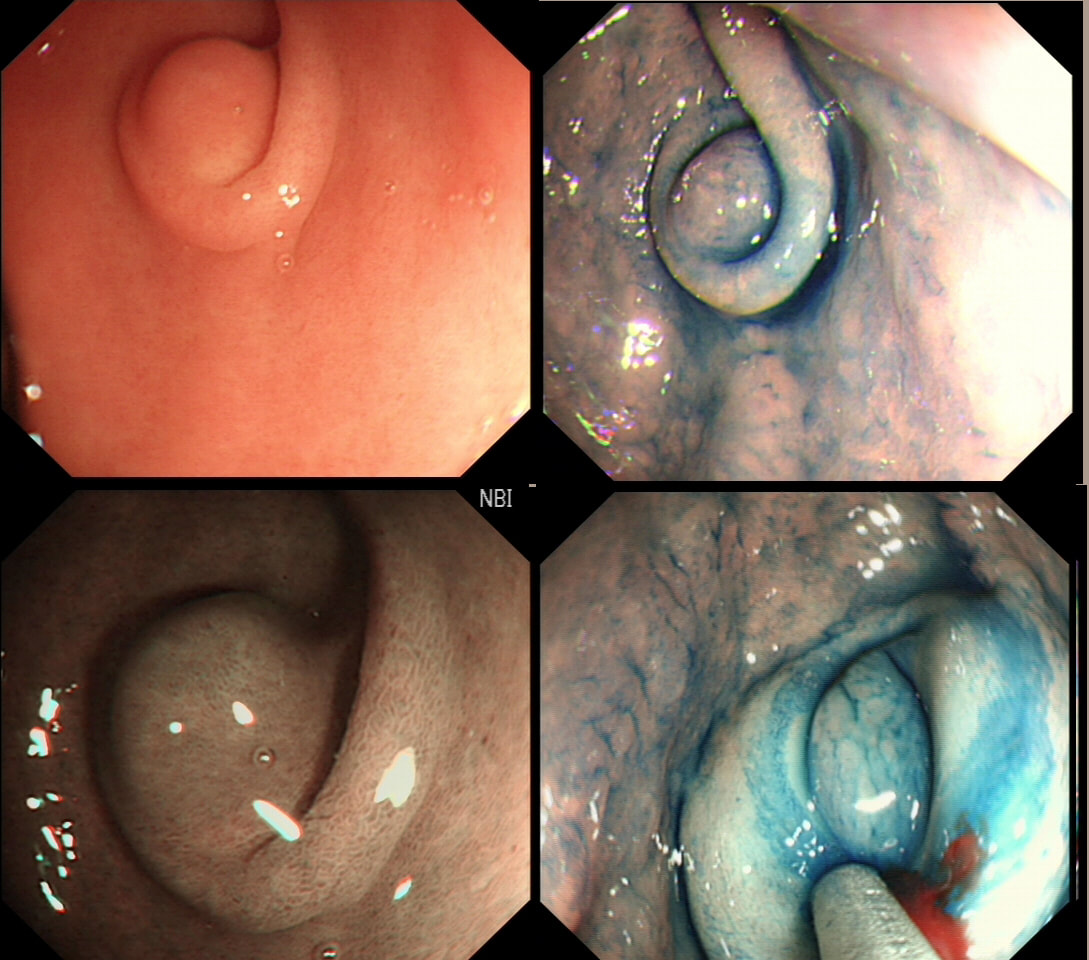
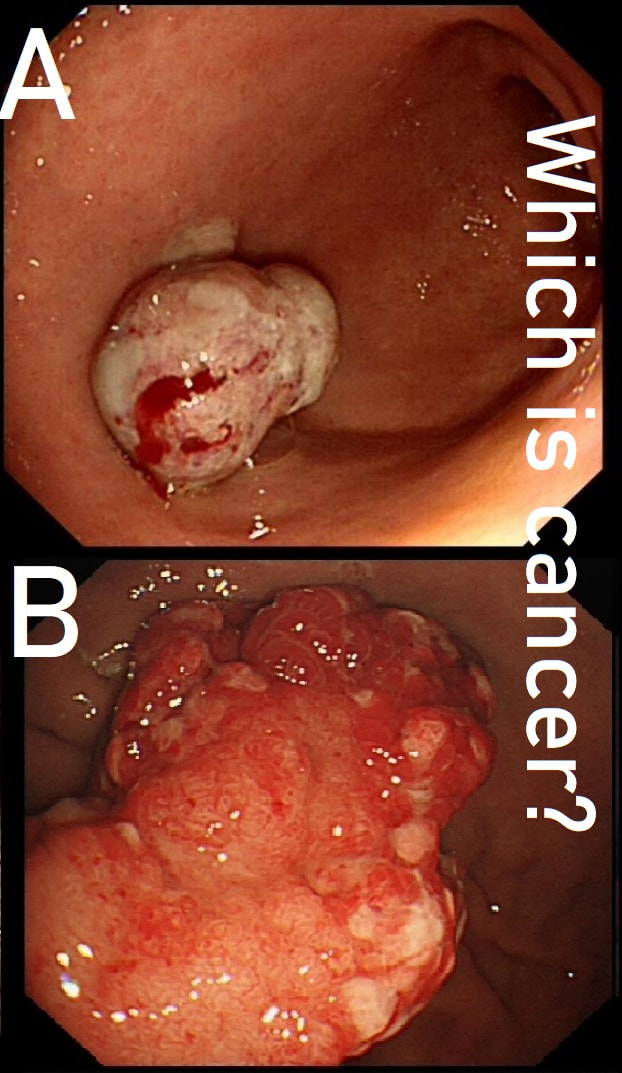
As you know, colonoscopy protects less well against future cancer in the right hemi-colon, than cancer elsewhere in the colorectum. Here are 5 lesions, all found in AC. Four are malignant and one isn't ...
WHICH ONE IS NOT A CANCER?
■ A
SM3 invading CRC with very little adenomatous tissue present
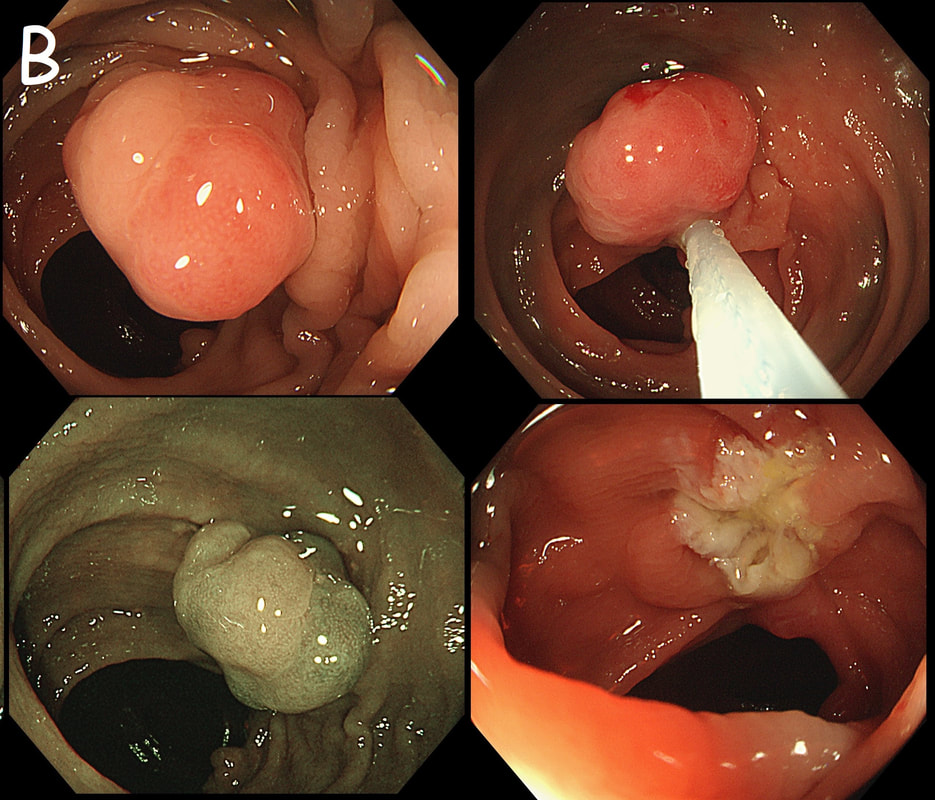
■ B
Cancer arising in an SSL !
■ C
A very superficial CRC removed by ESD
■ D
Yes! This is a 'caecal patch' in UC
■ E
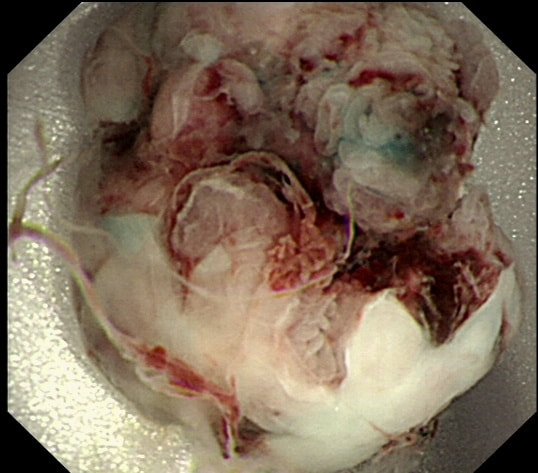
Nope, its a mucinous carcinoma!
explanation
You can probably tell that the first lesion is a cancer because of it's nobbly surface and lack of crypt pattern in places. It was a sm1 invasion T1,N0 cancer (i.e. invading up to the muscularis propria layer but didn't invade into it).
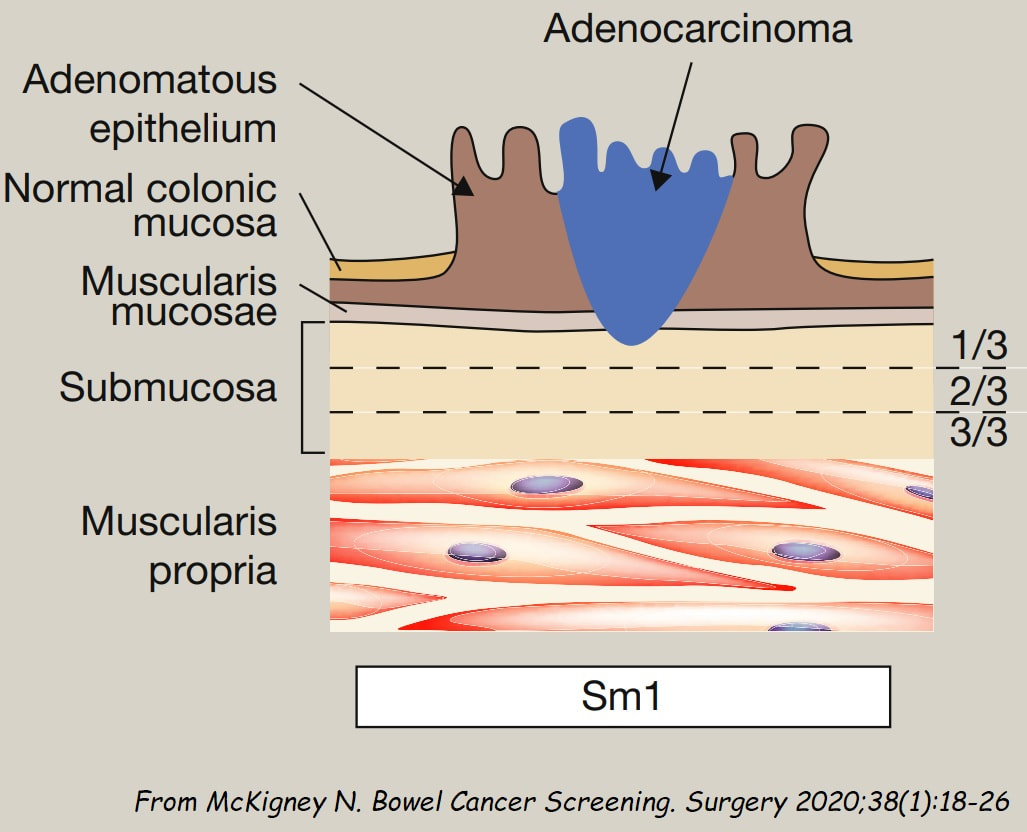
The second lesion is a sessile serrated lesion with a small cancer at it's edge. The staging was identical to the first lesion (sm1 invasion T1,N0 cancer). The third lesion was a superficial carcinoma (sm1 invasion only) and the only in the series actually removed endoscopically. The fourth lesion is the 'odd one out'. It's actually a 'caecal patch' in a patient with ulcerative colitis, only affecting the rectum. The final lesion was a 'mucinous adenocarcinoma'. Interestingly, analysis of the initial biopsies reported 'TA+LGD'. Don't believe everything those pathologists tell you! Your eyes don't lie! There is a further interesting twist here. After another set of samples, got the pathologists to agree with our endoscopic diagnosis, we were surprised that the patient was turned down for surgery due to severe comorbidities. Rather begs the question what was the point in putting the 85 year old gentleman through two colonoscopies !!! Anyway, that was 4 years ago and he has still not developed any symptoms from his cancer!? A recent CT confirmed that it was slowly growing and was now annular but without any evidence of spread or obstruction. Interesting and clearly turning someone down for surgery is not necessarily a 'bad thing' ! Still leaves me wondering why they keep scanning the poor bloke though? Below I've attached a reminder about the 'Kikuchi staging' which your pathologists should use to stage superficial flat cancers.
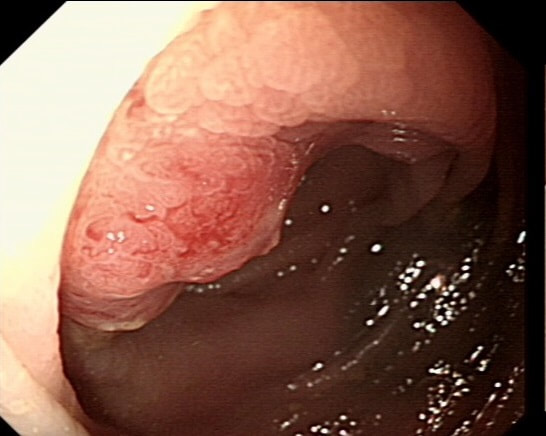
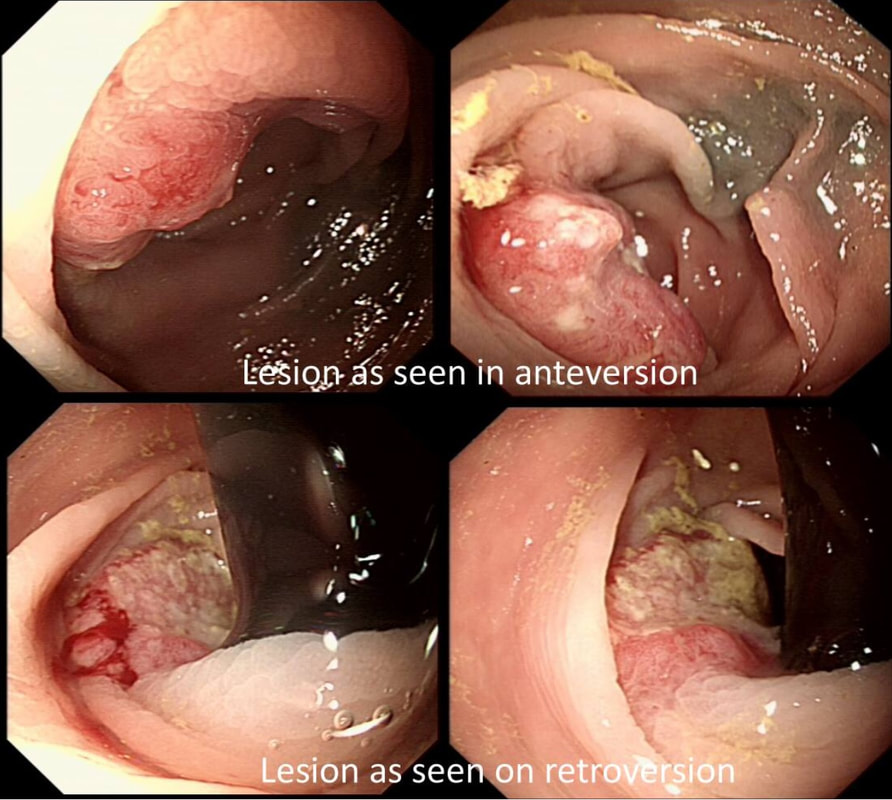
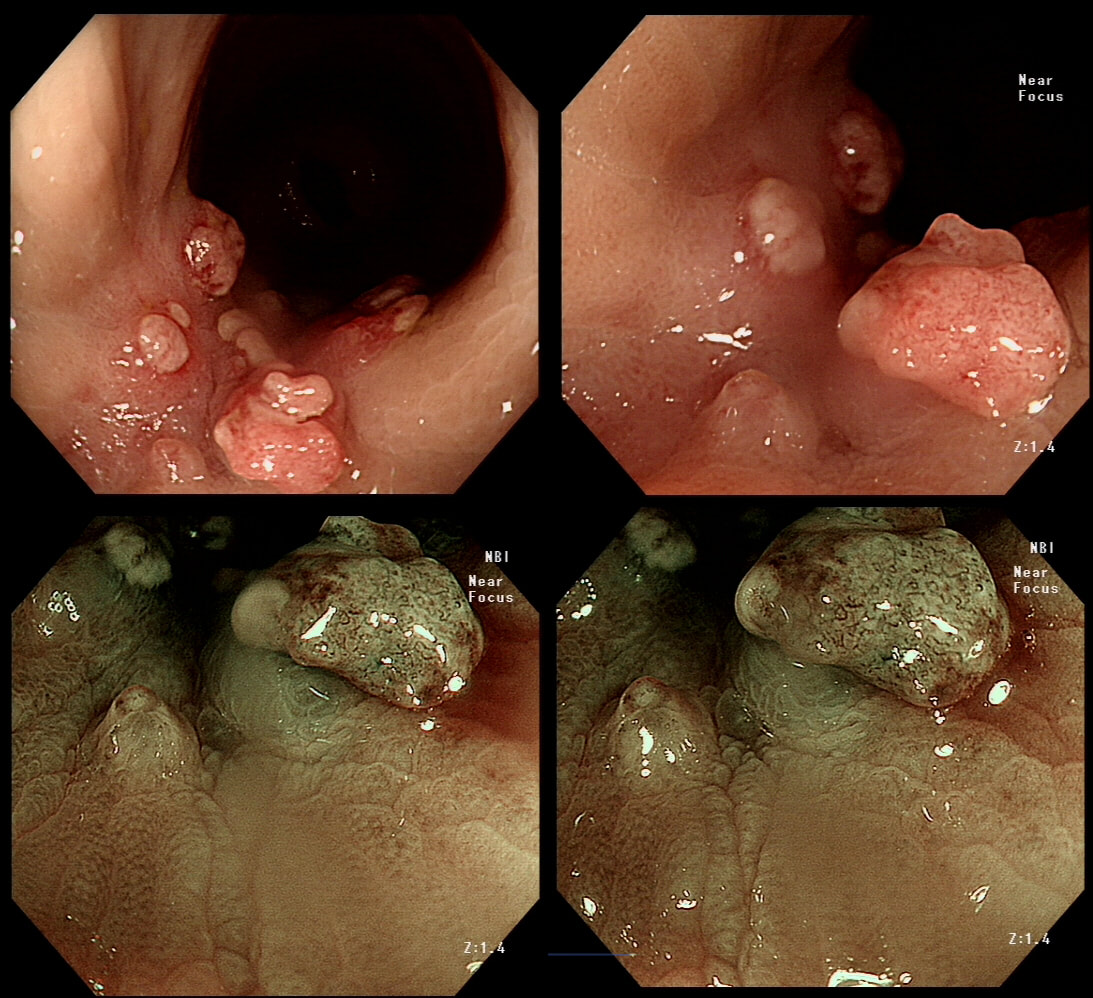
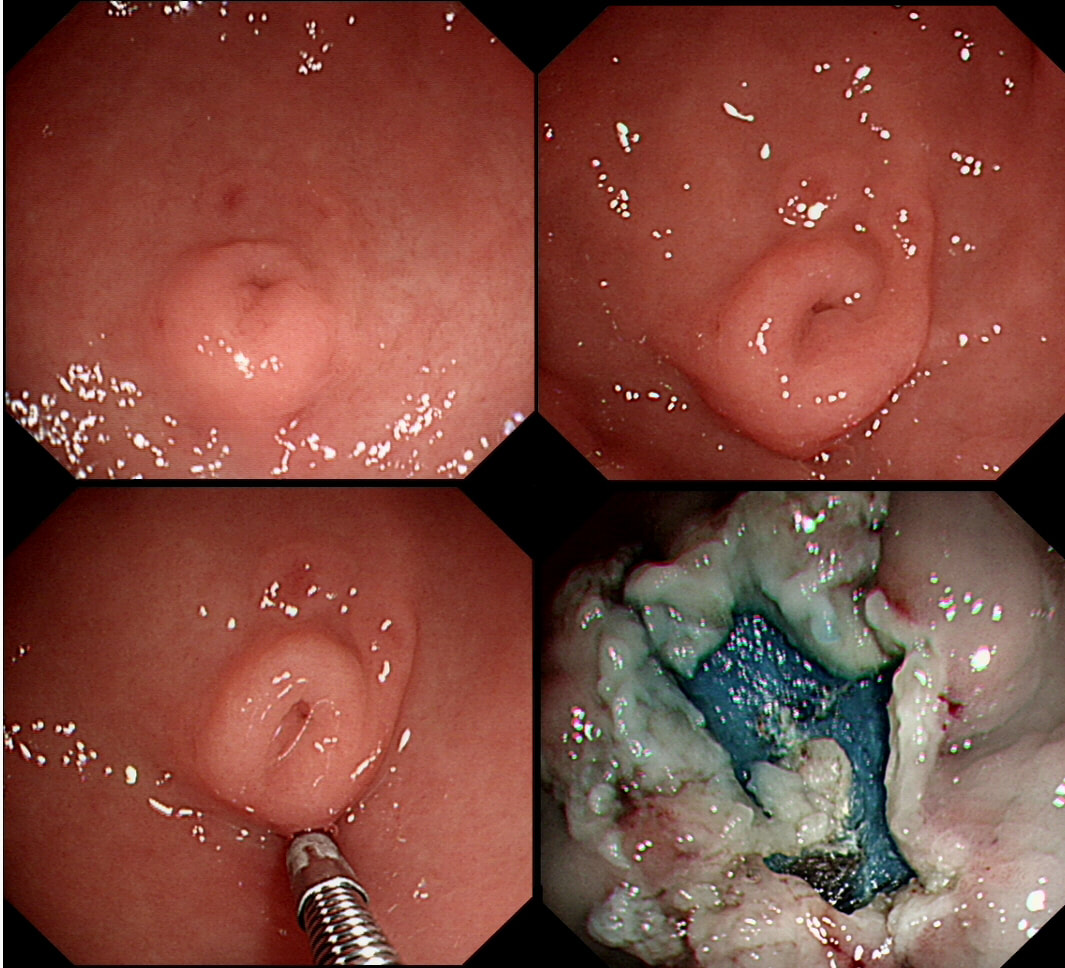
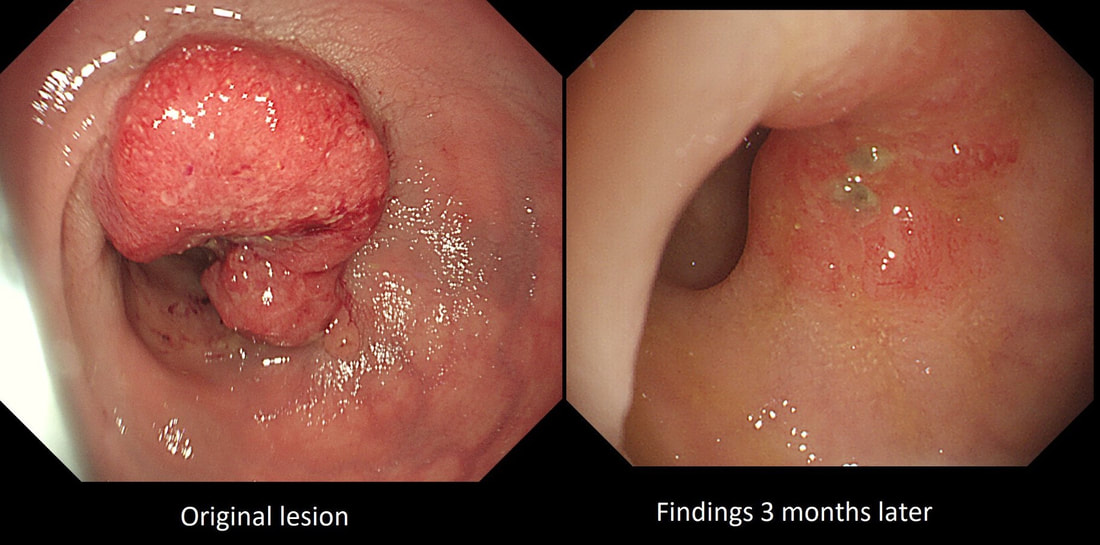
You are called into a neighbouring endoscopy room to advice on this lesion at the recto-sigmoid junction.
WHAT WOULD YOU DO NEXT?
■ Carefully assess and take samples
Yes but in what way would you 'more carefully' assess this?
■ Attempt to remove it now
Well, you would realise that it doesn't lift !
■ Attempt to remove it at a dedicated session
Noo! You missed it !
explanation
This lesion does look completely different when viewed in retroversion (image below). In retrovertion its obvious that it's malignant. Clearly the correct way ahead is to take some better targeted samples and refer to the next colorectal cancer meeting. If you hadn't assessed the lesion in retrovertion, samples would have revealed a TA+HGD only (this is actually what happened), delaying the correct management. The moral of the story is: always view all of the lesion before deciding on where samples should be taken from.
Aha! Are we not told that; "polyps which will be resected endoscopically shouldn't be sampled as it makes removal difficult"? The problem is that when patients have to wait long for their resection, it makes sense to take a few samples or at least do a 'test-lift'. Having to wait 3 month for a resection only to be told that the lesion is likely to be malignant is a disaster scenario. It's then better to exclude cancer as far as possible before asking the patient to wait months for the removal. Of course, its true that it can make a resection more difficult but it's hardly ever a showstopper !
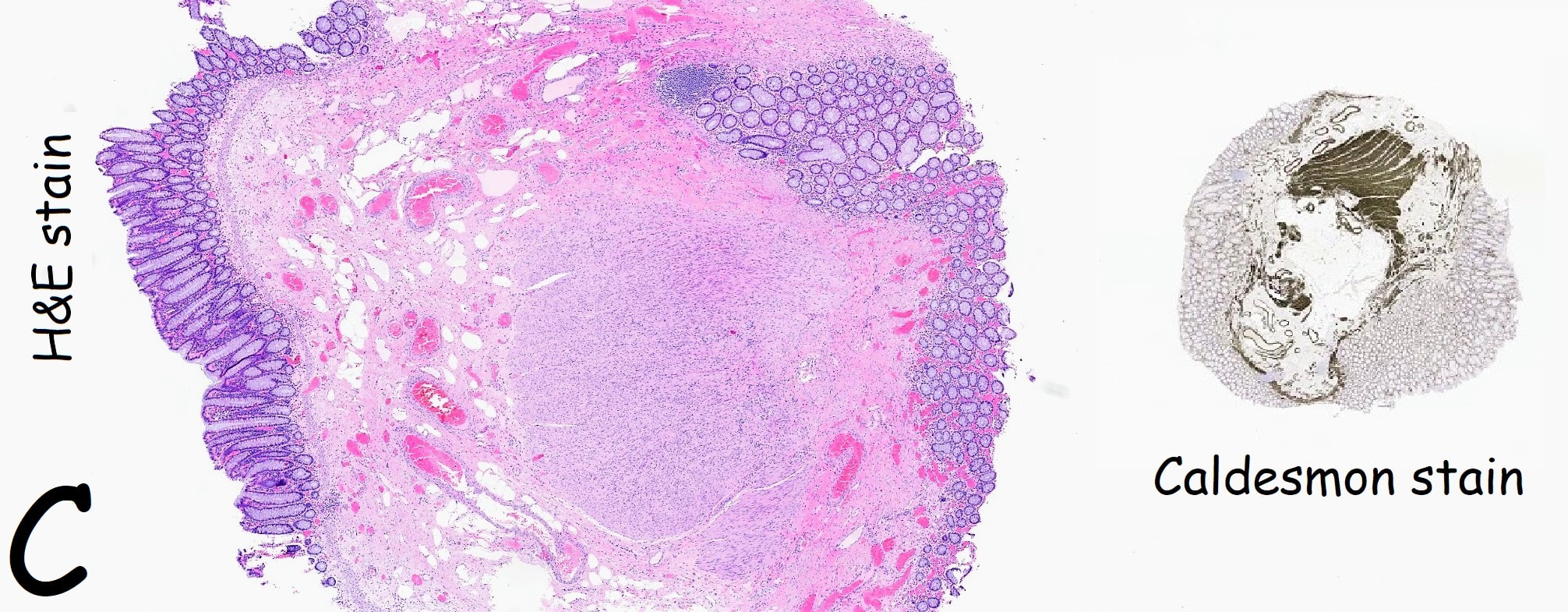
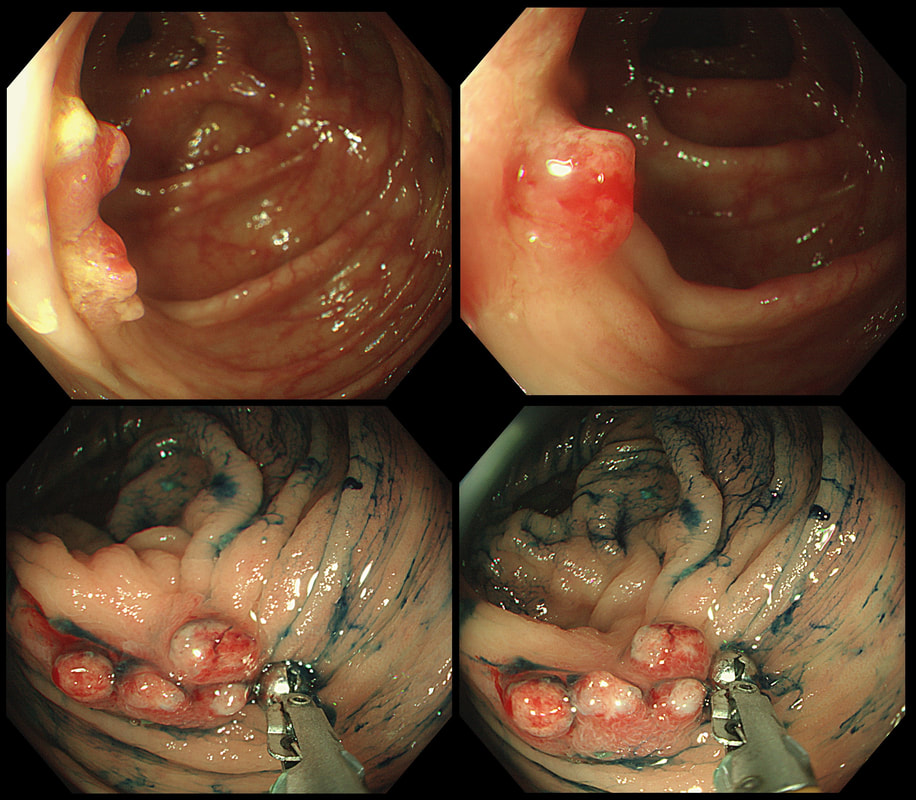
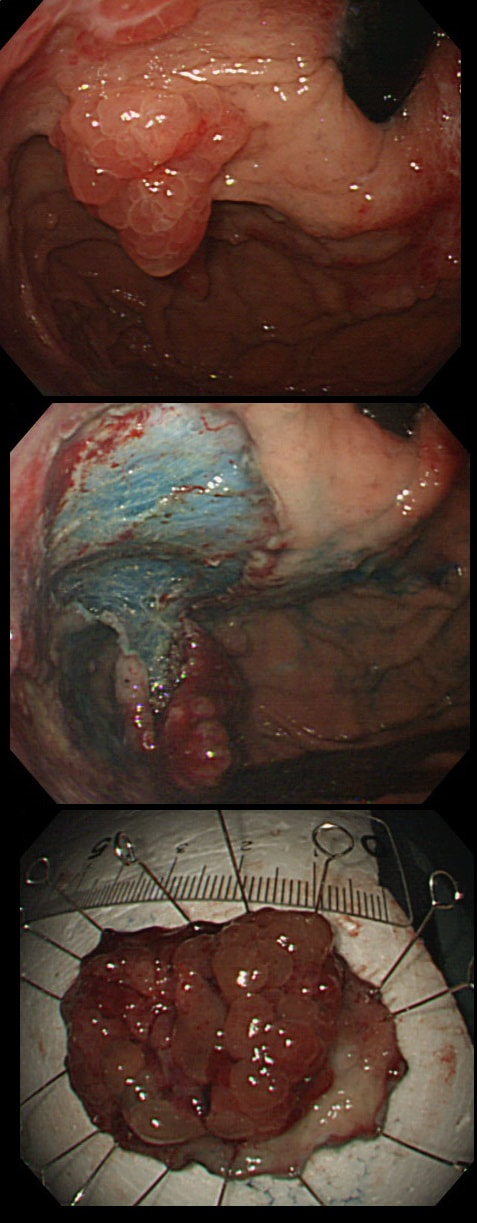
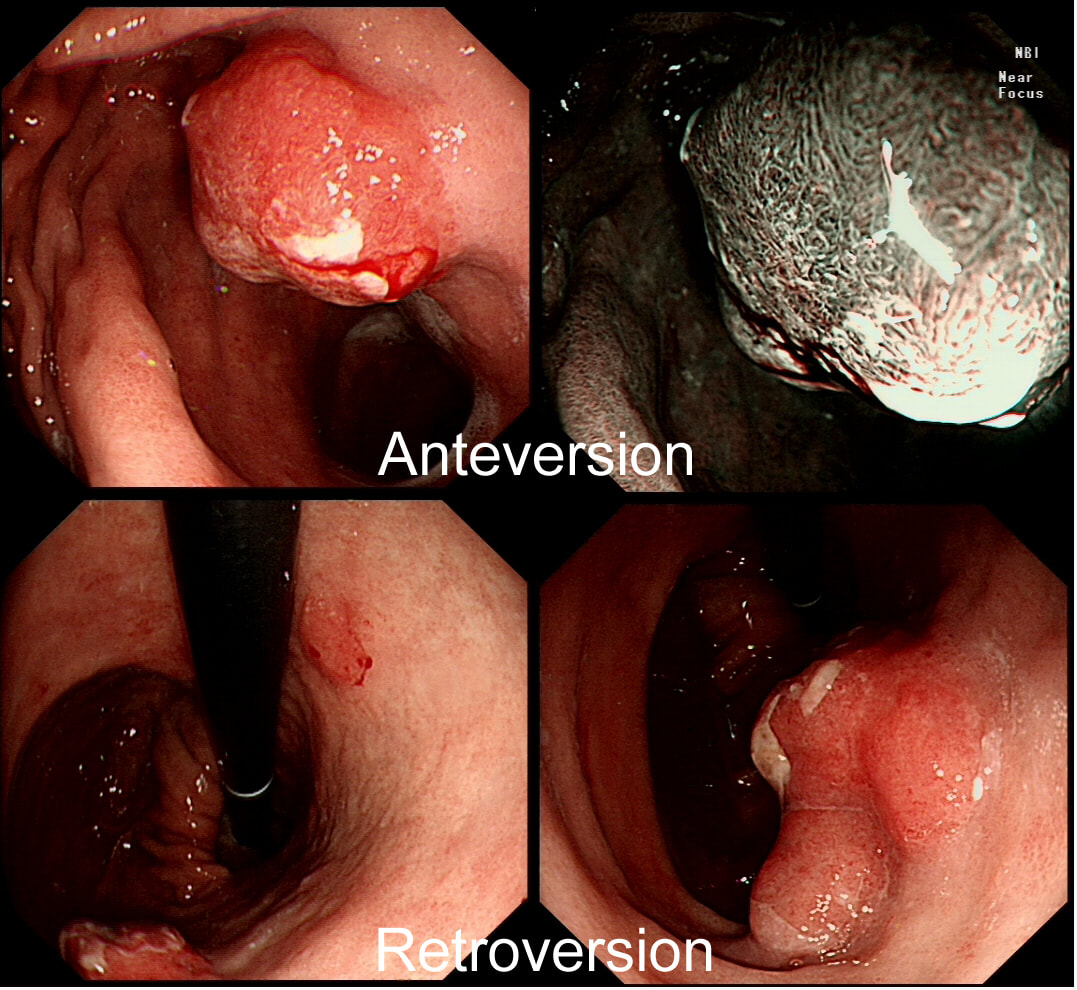
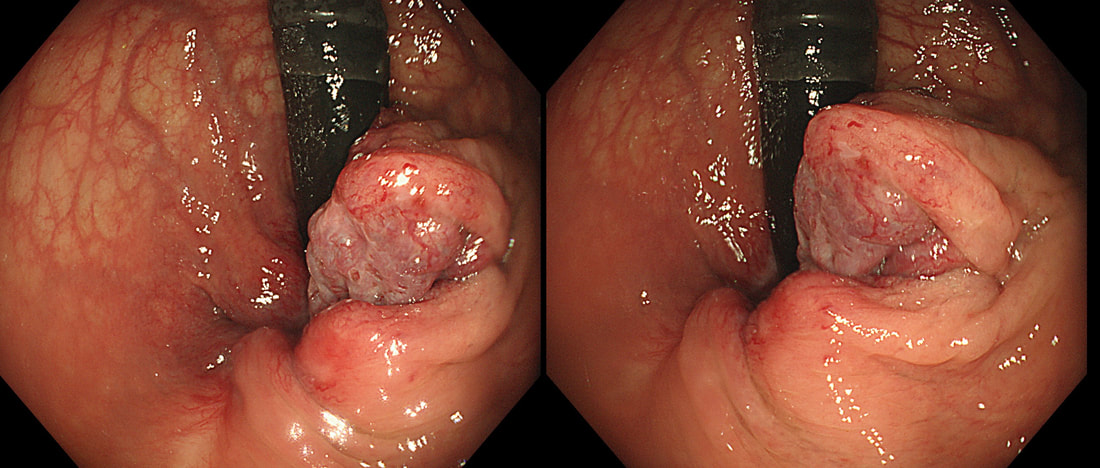
The lesion above is removed 'full thickness' and turns out to be an sigmoid carcinoma extending up to, but not involving the muscle propria layer (i.e. it's a T1 cancer with sm3 invasion). Histology reports clear margins but when the patient returns for a 'site-check' endoscopy, the nodule below in photograph B is found at the resection site ...
The mysterious nodule is removed and sent for analysis (Histology below as photograph C ) .
WHAT IS THE DIAGNOSIS?
■ Granulomatous/inflammatory reaction
Doesn't look like it!
■ Mucosal prolapse reaction
Yes, what else could it be!
■ Local recurrence of adenoma
Doesn't look adenomatous!
■ Local recurrence of carcinoma
Could be a concern, perhaps submucosal deposit but histology reported clear margins ...
Explanation
Endoscopically, this doesn't look like anything! It doesn't look either inflammatory or neoplastic (i.e. adenoma or cancer). Why then was it removed?! Endoscopically, this looked like normal mucosa but what then is it? To find out, the lesion was removed. I had my suspicions as to what was going on and took great care to close that tiny, unremarkable looking resection site with 4 clips.
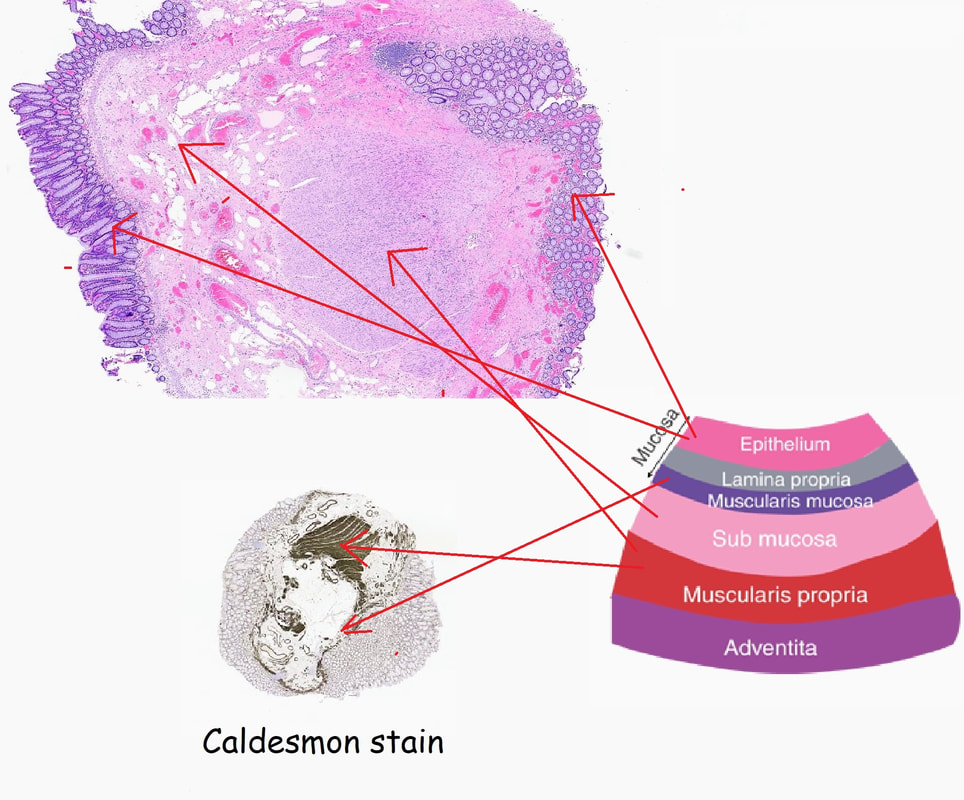
Histology did confirm that this is a full thickness mucosal prolapse at the FTR site. Of course, the point is that if you don't recognise this and resect it, you will end up with a delayed perforation !!! I've attached an annotated histology slide below. As a professional you should understand histology. By the way, the 'Caldesmon stain', shows up muscle. That large grey blob is of muscle propria in the centre of the 'polyp' and the thin grey layer around the periphery, just below the surface epithelium (the polyp is cut in a horizontal manner), is 'muscularis mucosa'.
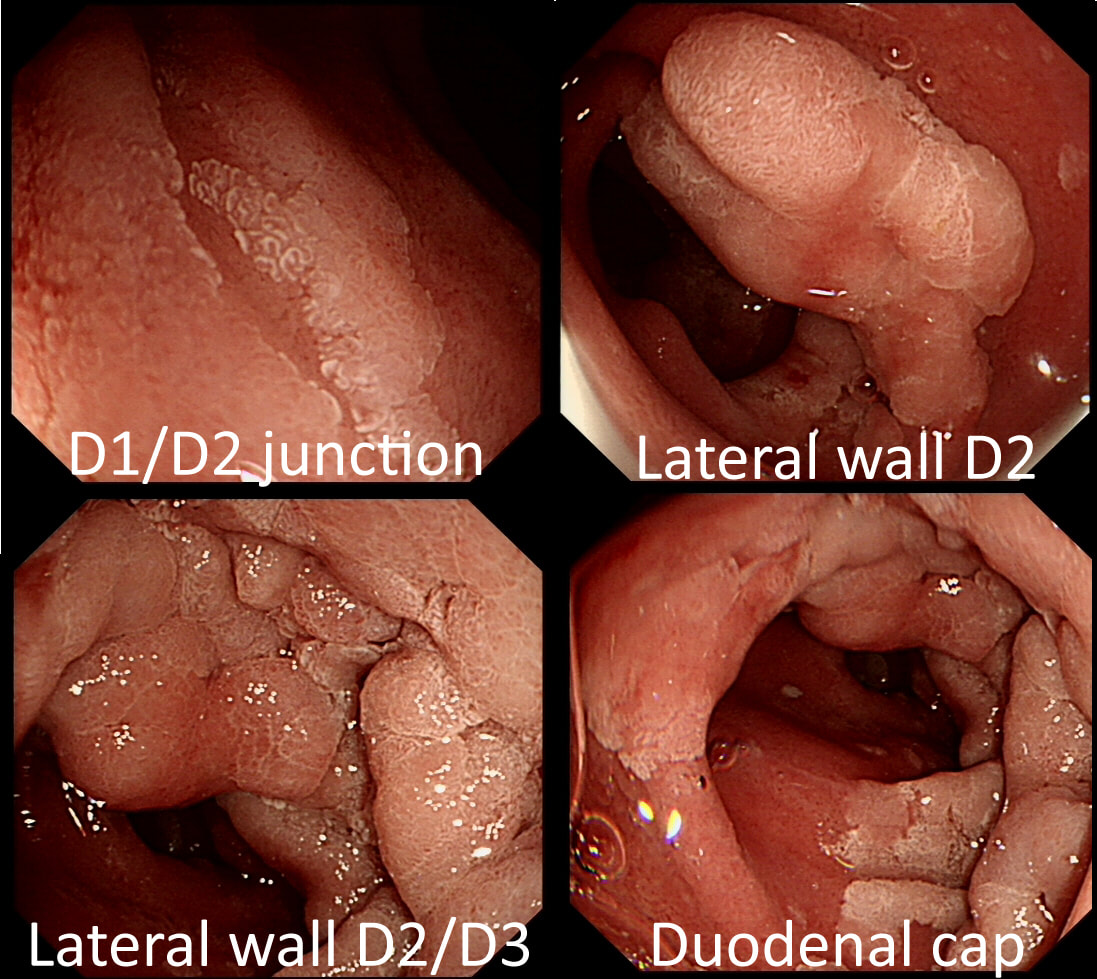
This was an incidental finding in the duodenal cap in a patient undergoing gastroscopy to investigate reflux symptoms. Although samples only showed a normal duodenal mucosa the patient was referred for an endoscopic resection. The lesion is soft to the touch.
WHAT WOULD YOU DO NEXT?
■ Abort this is nothing!
CORRECT!
■ Attack, this is something!
No it isn't !
explanation
This is exactly what happened ! The 'lesion' was soft to the touch and previous samples had not revealed any abnormality. Of course a Brunner's gland hyperplasia could be a possibility but the fold was too soft and in any event, these don't need to be removed! On the other hand, the patient was standing in the endoscopy room expecting his 'lesion' to be removed, to 'find out what it is! '
At times you may feel that you are 'put on the spot' in endoscopy, being asked to do things which you don't agree with or perhaps procedures you don't feel qualified to carry out unsupervised. Of course, you are more than a 'technician' and it's your duty to do what's best for the patient. That patient is 'your patient' in the endoscopy unit and should something go wrong, you'll have to take full responsibility. Don't let yourself be pushed around by either the patient or his physician. "I was just following orders" doesn't wash if there is a complaint ! Anyway, I diagnosed an 'odd looking but entirely innocent duodenal fold', took another couple of biopsies (to calm things down) and reassured the patient !
This patient has undergone a right hemicolectomy for cancer some 2 years earlier. He now attends for his first "post - surgery" colonic surveillance examination. This was found at the anastomosis.
WHAT IS YOUR DIAGNOSIS?
■ Granulation tissue
You ARE Right!!!
■ Adenomatous change
INCORRECT
■ Suspicious for tumour seeding
Looks odd but not because it's cancer!
explanation
There is no discernible crypt pattern, not because of cancer, but because these polyps are composed purely of granulation tissue! I also thought that it looked a little odd and took some samples (which I don't usually do when my endoscopic diagnosis is of "granulation tissue" at an anastomosis). Here is another example of granulation tissue, this time at a colonic EMR site, which looks worrying to the unwary ☺!
A fit middled aged patient with FAP is referred for consideration of a duodenal EMR for his duodenal polyposis. Earlier samples have indicated that this is a villous adenoma harbouring up to high-grade dysplasia (VA+HGD).
SHOULD WE ABORT OR ATTACK?
■ Abort and refer surgically
Yes, my inclincation as well
■ Attack!
Hm, what is that Spigelman score?
explanation
The Spigelman score is a little peculiar in that patients with scores of 0 to 3 have a very low risk of developing cancer. However, when patients accumulate a score of 4, surgery should be considered as there is a high risk of cancer.
Of course, many patients with FAP have desmoids which precludes surgery. In those cases I consider intervening endoscopically if: 1) the patient is fit enough to survive emergency surgery to sort out a perforation or cope with a 4+ unit blood loss AND/OR 2) there is a higher than average risk of cancer, for example in a patient with extensive HGD or confirmed IMca. Of course, the lesion also has to be 'resectable' within your level of expertise. Personally, I find lesions situated at 12 O'clock the most challenging to remove. Furthermore, a general anaesthetic is probably a prerequisite for resections taking longer than 20 min or so (which they always seem to do). Thus, I consider the "correct" answer to be 'Abort'! I decided that the lesion was likely to be too large for me to resect. Fortunately, the patient was a good surgical candidate. A 'pancreas sparing duodenectomy' was carried out and a TVA with mainly LGD but also areas with HGD was completely removed. Postoperatively the patient developed pancreatitis and was discharged 4 months later... Clearly this is 'Tiger Country' for surgeons as well !
This is the colonic mucosa close to a sigmoid polyp.
WHAT IS THE SIGNIFICANCE?
■ It's of NO significance!
ABSOLUTELY CORRECT!
■ It indicates a nearby cancer
INCORRECT!
explanation
I don't apply any significance to this finding! Below is an image from the low rectum of a patient with SPECTACULAR 'chicken skin appearance' of the low rectal mucosa. However, there was no nearby lesion and histology reported: "These biopsies are all normal. There are no features to suggest a diagnosis of active idiopathic inflammatory bowel disease or microscopic colitis and there is no evidence of dysplasia or neoplasia"
It was first suggested that this finding indicate that the polyp is malignant and that the chicken skin appearance is caused by lymphatic infiltration by cancer. However, with time it became clear that this was rarely the case. More recently it was suggested that the finding was more common in polyps harbouring HGD (Intest Res. 2015;13(4):318–25). However, I am convinced that it's actually mucosal 'trauma' which causes this 'chicken skin' appearance. In the case below, it was perhaps from straining on the toilet. When you see this close to a nearby polyp, the trauma is probably from the colonic peristalsis which intermittently pulls on the polyp. Of course, larger polyps are likely to be yanked more violently by the peristalsis and the link is therefore between the size and the location of the polyp rather than the histology. It was therefore entirely predictable that the authors found a link between 'chicken skin mucosa' and pedunculated or sessile polyps rather than flat polyps and 2) that the sign was most common in the sigmoid and rectum (because these generate the most propulsive force)! This patient is undergoing a gastroscopy because of abdominal pain. Some 20 years ago he underwent a distal gastrectomy for peptic ulcer disease. WHAT IS THE DIAGNOSIS?
■ a) Large hyperplastic polyp
No, there is more going on!
■ atrophic gastritis with malignant polyp
Closer to the truth but not the whole truth!
■ None of the above
What else could there be??!
explanation
You can tell that the mucosa must be atrophic by the mucosal xanthelasma at about 25seconds into the video. The polyp dangling at about 12 O'clock is covered with the same irregular surface as the base from which is arises. Finally, there is a large plaque-like area extending between 3-10 O'clock which does look concerning. It goes all the way up to the small bowel anastomosis. In general, large plaque-like cancers in the stomach do look intimidating but is usually intramucosal disease, provided that the appearance is uniform. The subpedunculated polyp indicate more 'heterogenous' disease. The obvious question is now, should we offer the patient an attempt at endoscopic resection or a 'completion gastrectomy'? The second part of that particular question is of course, 'is the patient actually a candidate for a completion gastrectomy?' At the time of my endoscopy, I didn't like the wide extend of the cancer and its heterogenous appearance. To also convince our MDT, I removed that subpedunculated polyp which was confirmed as intestinal-type intramucosal adenocarcinoma which was mainly 'moderately well differentiated' but with 'focal poor differentiation'. Predictably, the deep margin was positive, as the surface structure of both the polyp and the underlying gastric mucosa look the same. Although both an EUS and CT were reassuring, because the patient was a surgical candidate and also because of the large area of heterogenous disease, extending all the way up to the small bowel anastomosis, the patient was in the end offered a gastrectomy rather than an endoscopic resection. Ultimately, histology confirmed a very 'heterogenous disease' with areas within this 13cm lesion containing:
Is there any evidence that heterogenous gastric cancers (i.e. cancers with a mixed histology) are less likely to be cured by endoscopic resection? Actually there is! Ozeki et al looked at more than 3000 endoscopic resections for early gastric cancer and found that lesions with a mixed histology was significantly less likely to be cured by ESD (35-42% cure rate vs 76-92% cure rate). Ozeki Y. Mixed histology poses a greater risk for noncurative endoscopic resection in early gastric cancers regardless of the predominant histologic types. Eur J Gastro Hep 2021;32(2);186-193 Fortunately, there was no nodal disease. Almost everyone got this right when first published on our FoE facebook group.
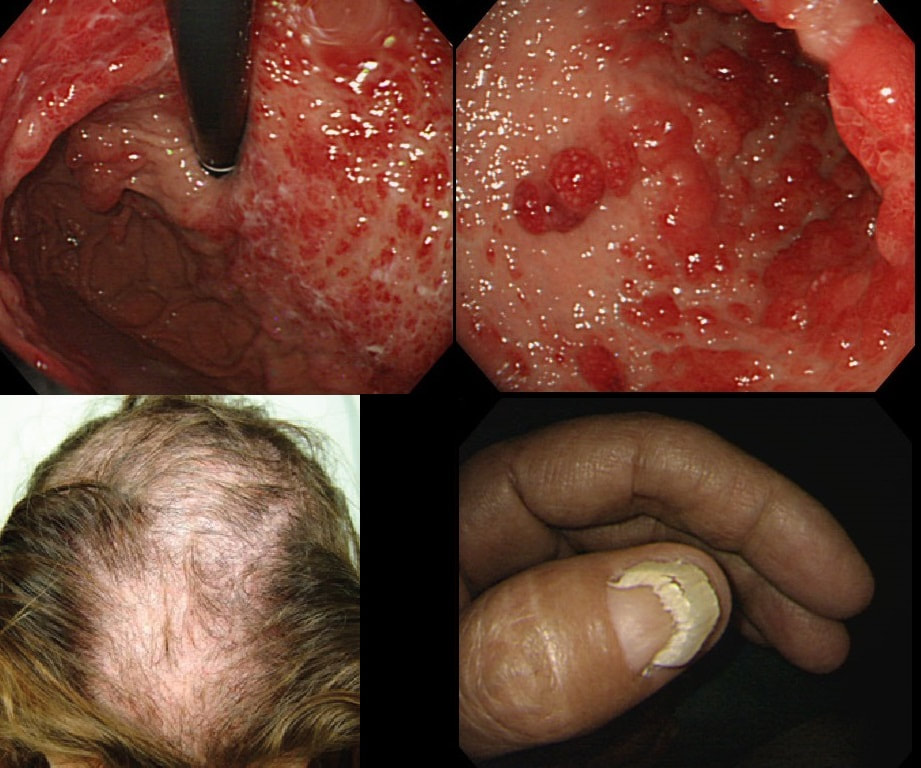
You are called into another endoscopy room to give an opinion on this gastric findings. It's a 65 year old man undergoing investigations for severe weight loss and anaemia. The anaemia was discovered by the dermatologists where he had been referred for investigations of suspected onychogryphosis and thinning of the hair.
WHAT IS THE EXPLANATION FOR THE GASTRIC POLYPS?
■ Likely hyperplastic polyps
INCORRECT!
■ Likely hamartomatous polyps
CORRECT! Clearly you know what the underlying diagnosis is?
■ Likely florid GAVE
INCORRECT!
■ Likely lymphoma
Gastric Lymphomas are usually ulcerated
■ Likely linitis plastica
INCORRECT! (although I guess it could be)
explanation
The nail dystrophy is not due fungal infection (onychogryphosis). In fact, this patient has Cronkhite-Canada syndrome. This enigmatic acquired syndrome is characterised by malabsorption, gastrointestinal polyposis, nail dystrophy, alopecia, cutaneous pigmentation, diarrhoea and weight loss. The nail dystrophy and skin pigmentation is clearly seen in the photograph. Other common symptoms include abdominal discomfort and a protein loosing enteropathy. The underlying cause is unknown. The cutaneous manifestations are probably all secondary to severe malnutrition due to diffuse small bowel mucosal involvement.
Histologically the mucosa is oedematous with dilated glands and an inflammatory cell infiltrate of the lamina propria. Gastrointestinal polyps are hamartomatous but may contain foci of adenomatous epithelium. The histopathologists will struggle in interpreting the biopsy findings and will mention things like 'marked foveolar hyperplasia', 'cystically dilated irregular glands', 'oedematous stroma' and 'scattered mixed inflammatory cells'. The differential diagnosis of such hamartomatous findings includes juvenile polyposis syndrome, Cronkhite-Canada syndrome, hyperplastic polyposis and Menetrier's disease. Of course, it will help them to know about the nails and hairloss etc!!! Malignant transformation has been reported and may not be as rare as initially thought . The prognosis is said to be poor but I'm not sure that this is still the case with 'modern management'. Interestingly, in this case a suspicious nodule was found (image below). It was removed endoscopically and found to be an early gastric cancer!
This antral lesion was found in a 25 yr old man undergoing gastroscopy for dyspepsia. Surface biopsies have only revealed a normal gastric mucosa.
WHAT WOULD YOU ADVICE?
■ This lesion does not require removal
Damn right it doesn't!
■ Another set of samples is required first
INCORRECT as these will only show normal mucosa again
■ an EUS is first required
INCORRECT! Because you will not be removing it anyway!
■ a CT should be done first
Don't be stupid!
■ I'll go ahead with the EMR/ESD
INCORRECT! Doing what you are told to do is not always the right thing do to !
explanation
This is a typical appearance of an 'ectopic pancreas' in the classical location along the greater curve of the antrum. The lesion even has a central dimple where the pancreatic duct finishes and a beautiful fold draped around it (which may not always be present in these lesions of course).
Sometimes I am asked to remove these for the 'reassurance' or to obtain a 'definite diagnosis' or because it's causing pain. Pain due to pancreatitis arising in these has been reported but is probably less common than finding that the rocking horse has shat on the floor. When the endoscopic appearances are typical, I don't believe that any further 'confirmation' is necessary. At Gastroscopy, I report these as: "small pancreatic rest was noted in the antrum. This is a normal, incidental finding of no significance" If you foolishly try to remove these, you will find that the endoscopic resection is actually quite difficult. The central pancreatic duct tethers the lesion down and therefore these never elevate well. You can see that in the bottom row image on the right. After struggling, you will end up with a mucosal defect and a central pink area (see image below). The resected fragments will only contain normal gastric mucosa and you will have to either dig deeper (have you heard of the expression: "to dig an even deeper hole for yourself?") or take some samples from the central pink area to capture some pancreatic duct tissue. If the referring team are steadfast that "proof is needed that the lesion is innocent", an EUS is safer and easier than an attempt at endoscopic resection.
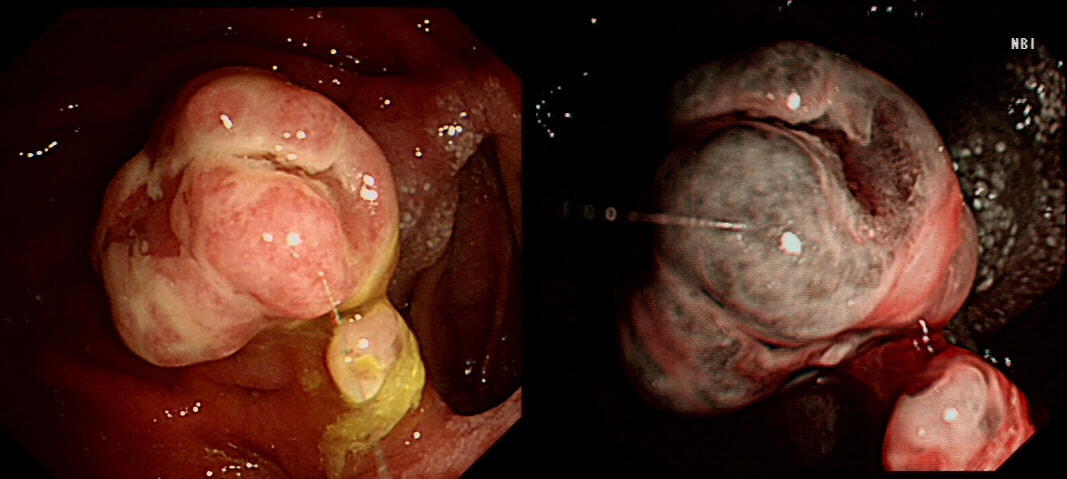
Previous sampling of this gastric nodule has confirmed HGD and an EMR has been organised.
WHAT IS THE MOST APPROPRIATE MANAGEMENT?
■ Take further set of Samples!
No, because it's unlikely to change anything!
■ Organise staging CT and EUS first!
YES! This may well be invasive cancer!
■ Go ahead and resect it!
INCORRECT!
explanation
The correct approach would be to slow down a little! Pathologists would always like more samples but actually, surface samples may not be a true reflection of what is nestling deep within this lesion! This lesion is a little over 3cm in diameter and it would be very surprising if it wasn't an invasive cancer after all. Furthermore, there is a suspicious looking red, flat patch close to the shaft of the endoscope in the bottom row image on the left.
Ultimately, this patient underwent a gastrectomy which confirmed that the large nodule was indeed an invasive adenocarcinoma invading as deep as the muscle propria layer (T1b). In addition, there was multifocal LVI and a nearby involved node (N1). That red patch turned out to be a spot of HGD. Because EUS found a suspicious node, a gastrectomy was the most appropriate therapy. Of course, we don't normally organise a CT and EUS for endoscopically resectable lesions which are not thought to be malignant. Of course biopsies have indicated that this is HGD only. However, the lesion is chunky and its unthinkable that it's HGD only. More surface biopsies may or may not reveal the true nature of the lesion. And frankly it doesn't really matter because endoscopically this is likely to be an invasive cancer. It's a dysplastic nodule about 3cm in diameter and probably close to 2cm in height !!! Of course, the pathologists always want more samples but with a lesion of this size, of borderline resectability, a CT and EUS is more reassuring than further samples. The EUS revealed a chunky node and the patient had an appropriate gastrectomy. How about a a test-lift? Unfortunately, test-lifts are difficult to interpret in the stomach and is likely to be 'borderline' committing you to 'having a go' trying to resect the lesion endoscopically. Of course, there is nothing wrong with an 'attempt at EMR/ESD' but the point is that first you would like to have the reassurance of a normal CT and EUS. Of course, you would probably take another set of samples rather than just looking at the lesion but whatever the pathologists say, it will not trump the endoscopic diagnosis of; ' invasive cancer'. But why not just 'lob it off' for a definite diagnosis as well as staging? The reason is that a ESD resection of a cancer is very difficult. This is because the semi-translucent lifting plane disappears due to the desmoplastic reaction and you end up cutting blindly through opaque beige coloured tissue. Perforating during the resection of a cancer is potentially devastating for the patient as it can upstage the lesion to 'disseminated disease'. As the stakes are very high in cancer, it would be inappropriate to subject the patient to a significant risk with only a slim chance of benefitting.
This nodule has developed at the gastric anastomosis in an elderly patient who had undergone a Billroth II procedure some 30 years ago for peptic ulcer disease. Initial biopsies (4 samples) have indicated that it is a hyperplastic polyp.
HOW WOULD YOU MANAGE THIS CASE?
■ Reassure and discharge the patient
INCORRECT!
■ Keep lesion under surveillance
INCORRECT
■ Take another set of biopsies
INCORRECT!
■ Remove the lesion endoscopically
ABSOLUTELY!
■ Refer patient for surgery
INCORRECT!
explanation
For some reason, histopathologists often find it difficult to report on nodules sampled at a gastric stoma. In this case, I actually removed the polyp and took a full set of biopsies from around the stoma. The histology wasn't entirely clear cut. A couple of our histopathologists thought that the lesion was benign whilst three thought that it contained a small spot of intramucosal cancer.
My endoscopic diagnosis was of a malignant polyp as it seemed a little too irregular with a funny deep cleft for being a simple inflammatory polyp.
This was found at Gastroscopy in a middle aged man without any family history undergoing gastroscopy because of an iron deficiency anaemia.
WHAT WOULD YOU DO NEXT?
■ Take a full set of biopsies
INCORRECT!
■ Organise and EMR
INCORRECT
■ Request a capsule study
INCORRECT!
■ Organise a colonoscopy
ABSOLUTELY!
■ Request a CT abdomen
INCORRECT!
explanation
There lots of "cystic fundic polyps" and there is also at least one duodenal adenoma. Such findings in a 55-year-old man is suggestive of the "attenuated" form of familial adenomatous polyposis (FAP). In the absence of a family history, presumably this is a new mutation. Of course this means that the most likely reason for the iron deficiency anaemia is a colorectal cancer. This patient needs an urgent colonoscopy!
To remind you, the APC gene is located on the long arm of chromosome 5 and encodes a tumour suppressor protein. APC is a huge protein that no doubt acts in many different ways to help control cell division and cell attachment and preserve the chromosome number through cell division. Normally, APC mops up intracellular ß-catenin. The newly formed “APC– ß-catenin complex” is quickly destroyed. The removal of intracellular ß-catenin is a good thing! Because when a mutated APC gene is less capable of mopping up intracellular ß-catenin, cell-to-cell adhesion is reduced and cells are allowed to stay non-differentiated and immature. That's not good! Of course there is a second possibility. This could also be a new MYH gene mutation. Briefly, the MUTYH gene encodes 'MUTYH glycosylase', which is a DNA repair enzyme. Similarly to attenuated FAP, patients with MUTYH gene mutations develop multiple colonic polyps in adulthood. However, in contrast to attenuated FAP, patients with MUTYH gene mutations have both an markedly increased risk of both colonic and gastric cancer. Characteristics of 'attenuated FAP' compared with 'classic FAP' are:
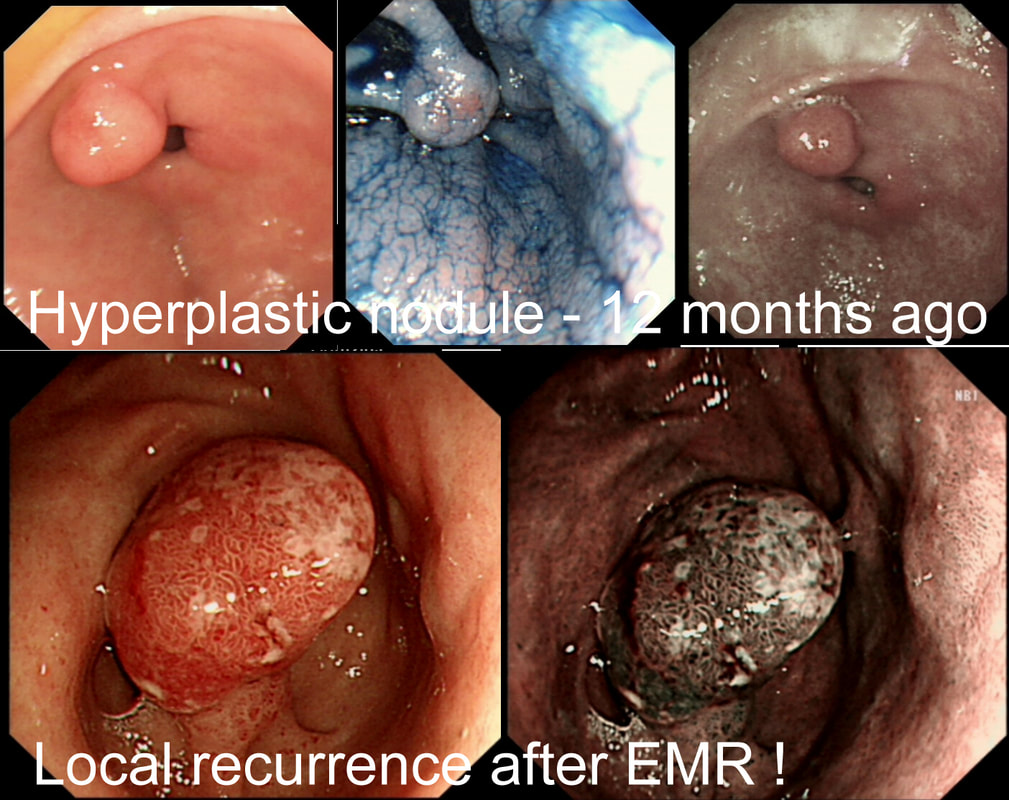
I removed this pre-pyloric nodule about a year ago. Histology confirmed an inflammatory/hyperplastic polyp without dysplasia. Now the thing has grown back!?
WHAT IS LIKELY TO BE THE APPROPRIATE MANAGEMENT?
■ Take a full set of biopsies
INCORRECT!
■ Remove the local recurrence by snare
INCORRECT
■ Eradicate the Helicobacters
CORRECT!
EXPLANATION
In most cases, gastric hyperplastic polyps are caused by Helicobacter pylori. However, because of the 0.6% - 2.1% risk of cancer arising within a hyperplastic polyp in the stomach, I do remove the larger ones. However, a year ago, this proved to be a hyperplastic polyp caused by Hp. The polyp recurred because the underlying cause had not been dealt with. This patient needs to have his Hp eradicated!!
A study published in Digestive Diseases & Sciences 2020;65(12):3652-59 confirmed that all 14 patients had complete regression of their hyperplastic gastric polyps 10 months after eradication of their Helicobacter’s compared with none of the 13 patients in the non-eradication group.
This is a 55 year old lady undergoing flexible sigmoidoscopy to investigate her PR bleeding. This lesion was found on retroverting the endoscope in the rectum.
WHAT IS THE MOST LIKELY DIAGNOSIS?
■ Fibro-epithelial polyp
INCORRECT
■ Inflammatory polyp
INCORRECT
■ Haemorrhoid
Yes!
■ Adenocarcinoma
INCORRECT!
■ Squamous cell carcinoma
INCORRECT
explanation
The three anal vascular cushions (at 3, 7 and 11 O’clock) help with maintaining continence.
When these cushions become abnormally enlarged, due to for example excessive straining (chronic constipation), age, or raised intra-abdominal pressure (such as pregnancy, chronic cough, or ascites), they become pathological. Haemorrhoids are classified as follows:
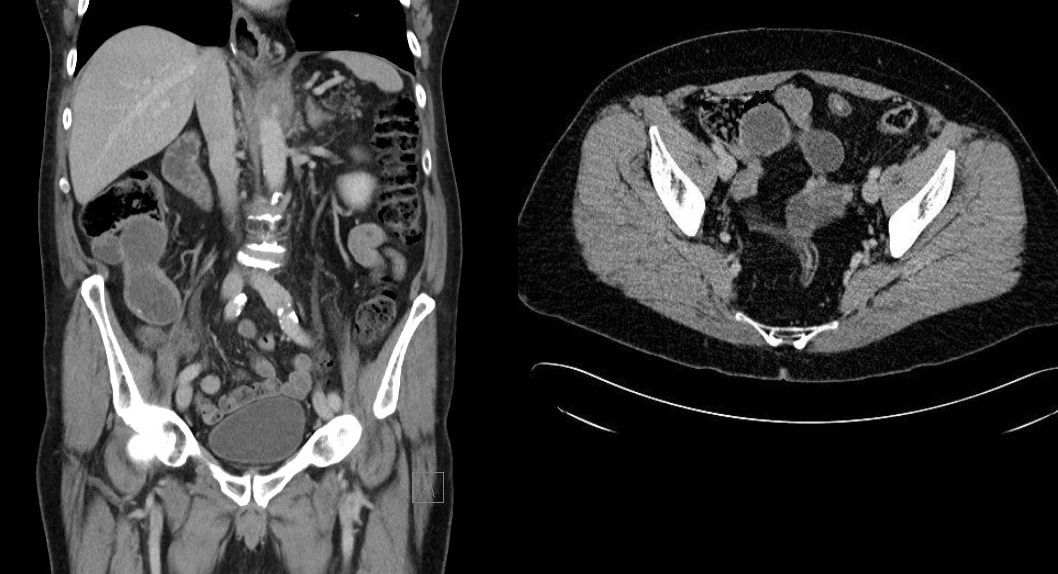
After a flexible sigmoidoscopy has excluded a low colonic lesion, most cases can be managed conservatively by reassurance, preventing constipation and topical analgesia. Alternatively, 1st and 2nd degree haemorrhoids can be treated with rubber-band ligation (RBL). A surgical haemorrhoidectomy may be indicated in the case of 3rd degree and 4th degree haemorrhoids. The serious complication of hemorrhoidectomy is anal stenosis which occurs after 2-4% of operations. However, I have the impression that this procedure is performed far less now than in the past but have not seen any data to actually confirm this impression. A 50 yr old patient complains of some low abdominal discomfort and undergoes a CT (image 1) following which a colonoscopy is organised to examine the caecum (video). WHAT IS THE MOST APPROPRIATE MANAGEMENT?
■ Biopsies only
THERE'S NO NEED FOR BIOPSIES
■ Snare Polypectomy
There is no need to remove this
■ EMR
There is no need for a polypectomy
■ ESD
INCORRECT!
■ Surgery
CORRECT, THE CT TELLS YOU THIS!
explanation
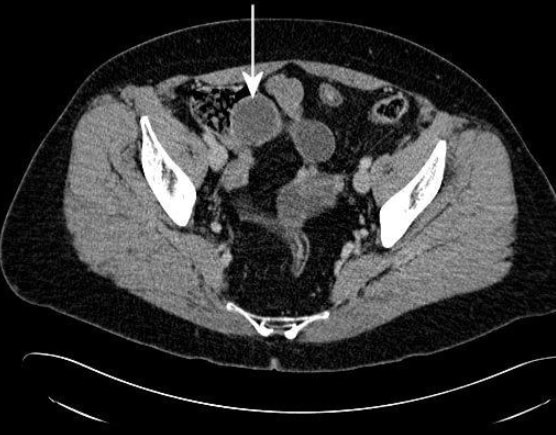
The CT shows a large appendiceal mucocoele (see arrow below) and of course therefore, this is simply a prolapsed appendix. More than 90% of our FoE group got this right.
Appendiceal mucocoeles are rare, incidence of 0.07 % - 0.3 % with a female predominance (M:F 1:4), and mean age at presentation of around 50 years. It most cases it is an incidental finding at for example ultrasound, laparoscopy or colonoscopy. The most likely aetiology is proximal obstruction of appendiceal lumen due to chronic inflammation and subsequent cicatricial contraction. Continued production of mucous from the distal appendiceal cells lead to large collection of mucus within the lumen. Noriko Suzuki told us the story of her as a junior endoscopist taking a biopsy from a mucocele, following which pus started coming out from the biopsy site! Following which the patients abdominal pain improved. She thought that she had treated the patients appendicitis but of course the abdominal pain recurred a few days later. Of course, an open appendicectomy is the treatment of choice. It has been proposed that a laparoscopic resection may be linked with a greater risk of iatrogenic rupture of the cyst. Leakage of the gelatinous material into the peritoneal cavity may lead to peritoneal dissemination of thick mucinous blobs causing recurrent intestinal obstructions, a condition called “Pseudomyxoma peritonei”. Pseudomyxoma peritonei carries a significant morbidity due to recurrent hospitalizations and a reported mortality of 50% in 5 years (Hinson 1998). There is a second reason to recommend surgery and this is the potential for malignant progression to “mucinous cystadeno-carcinoma”.
These two antral polyps look very different. That's because one is a polypoid cancer and the other is an innocent hyperplastic polyp.
WHICH IS WHICH?
■ A is malignant
Yes! A was poorly differentiated gastric carcinoma!
■ B is malignant
Surprisingly, B is actually the innocent hyperplastic polyp
explanation
Most (73%) thought that the hyperplastic polyp (polyp B) was the malignant one. Although large, the angry red colour and white spots are both strongly suggestive of a hyperplastic polyp.
Interestingly, in both cases, the patient presented with anaemia. Of course, when the patient undergoes the endoscopy because of anaemia, any gastric polyp should be viewed with suspicion. Although the lack of surface crypts was disconcerting, as polyp A did not seem to be firmly attached, it was removed as a single fragment (of course). Histology confirmed that the lateral and deep margins were clear and there was no lymphovascular invasion (LVI). In the subsequent 'cancer meeting', it was pointed out that 'poor differentiation' may be considered an indication for surgery. This would indeed be the case elsewhere in the GI tract. However, in the stomach, poor differentiation 'on its own' (clear margins and no LVI) is 'allowed' provided that margins are clear, there is no invasion into the deepest layer of the submucosa and, most importantly, there is no LVI. Emerging data highlights LVI as THE MOST IMPORTANT predictive factor anywhere in the GI tract. The original ‘Japanese Standard Gastric EMR criteria’ gave the go-ahead for resection of well differentiated cancers up to 2cm provided that there was no surface ulceration. Nowadays, most of us would apply the 'Japanese Extended criteria’ which allows us to attack well differentiated EGC's up to 3cm in size even if there is some superficial ulceration of the surface. Pertinent to this case, the extended criteria also allows us to attempt to resect poorly differentiated cancers up to 2cm provided that there is no surface ulceration.
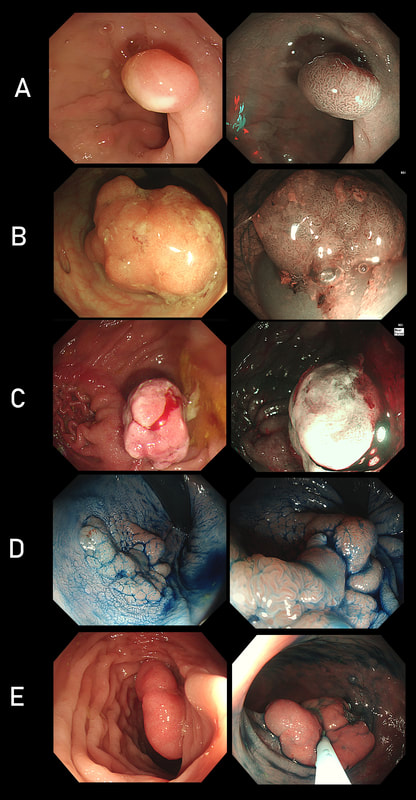
Here are five polyps in five patients.
CAN YOU TELL WHICH IMAGE IS SHOWING;
■ TA + LGD
Polyp A shows a small TA with LGD (most small polyps are TA's)
■ TA + HGD
Polyp E is a TA with HGD. All LST-NG's are TA's and many harbour HGD or even superficial invasive cancer. Recognise them and try to remove them single fragment or in as few fragments as possible.
■ TVA + LGD
Polyp D is a LST-G type of polyp. LST-G's are almost always TVA's with no more than LGD. When they 'turn' they usually develop a nodule at the site of cancer.
■ TVA + HGD
Polyp B is a sessile TVA with HGD. Of course the Devil has Horns! Furthermore, in large sessile polyps surface biopsies may not reveal what hides deeper inside ...
■ NON-NEOPLASTIC
Polyp C is an ugly little polyp without any crypts composed entirely of inflamed granulation tissue.
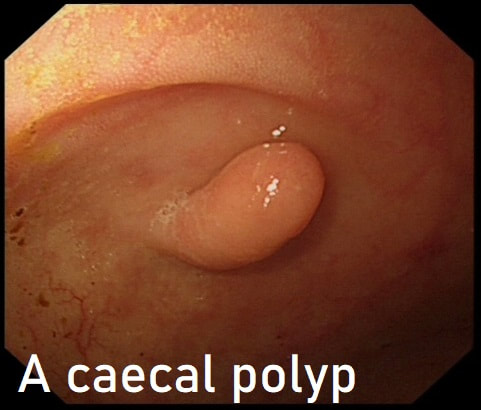
This polyp was seen in the caecum.
WHAT WOULD YOU DO NEXT?
■ Nothing!
CORRECT!
■ Take a biopsy
INCORRECT!
■ Snare polypectomy
INCORRECT!
■ EMR
INCORRECT!
■ Full thickness resection
CORRECT!
Explanation
Of course this is a normal inverted appendix and there is no need to do anything. However, occasionally there is a mass in the appendix pushing the root back into the caecum. Therefore it may be prudent to have a look at the 'apex' of the caecum to make sure that this is not indented in any way. If it was, or if there is another reason to be suspicious, a CT or an ultrasound would be the next step
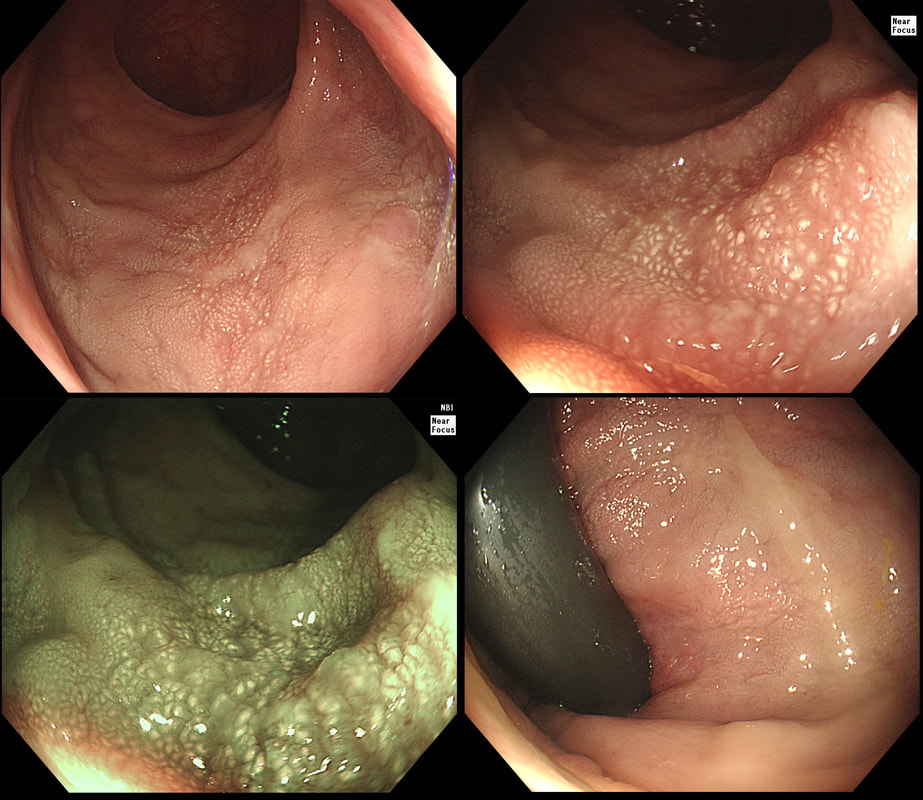
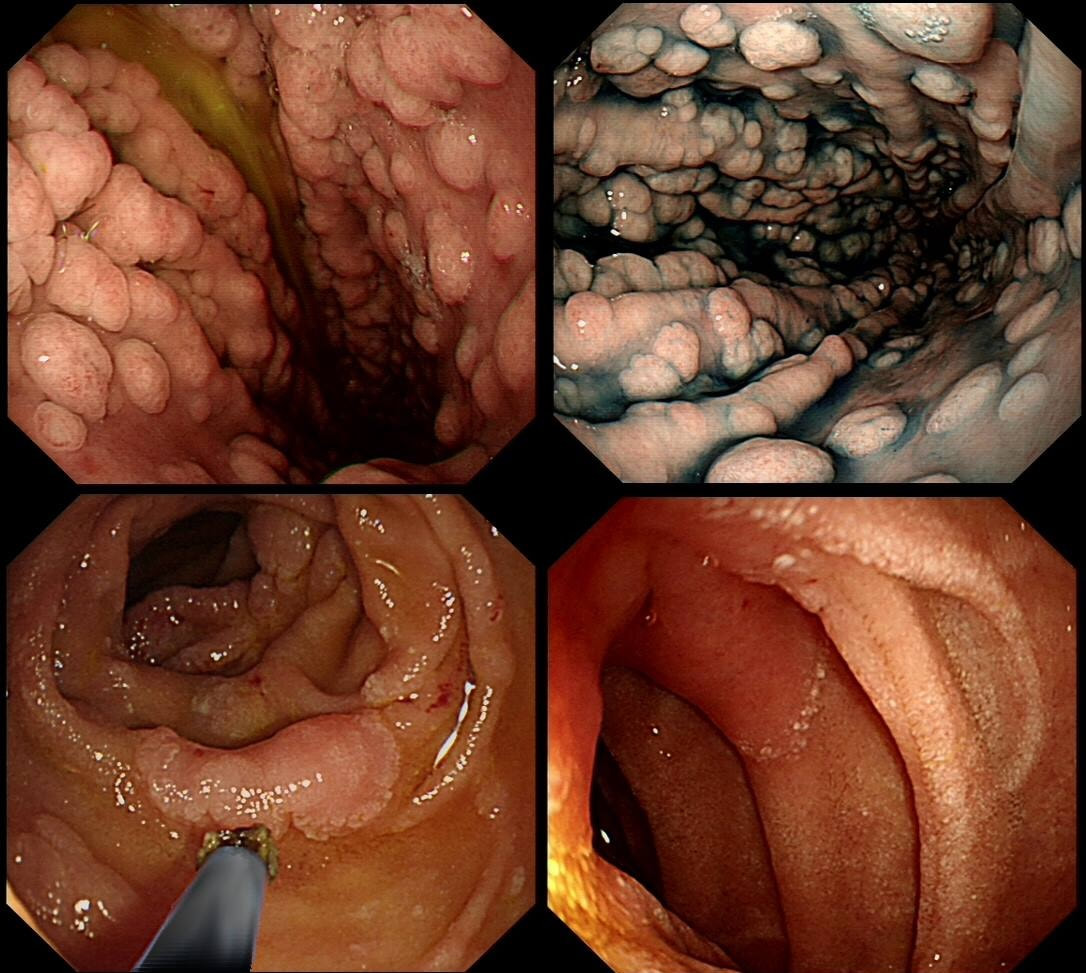
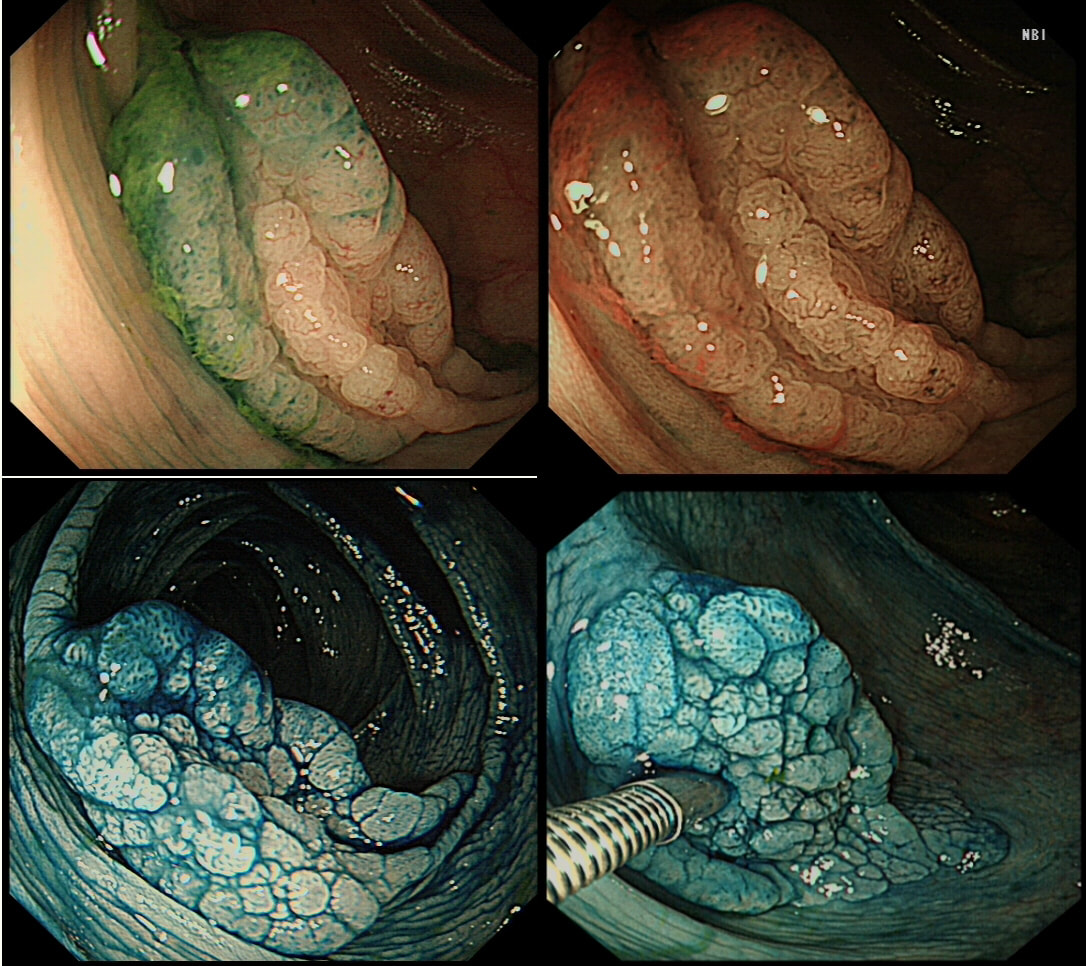
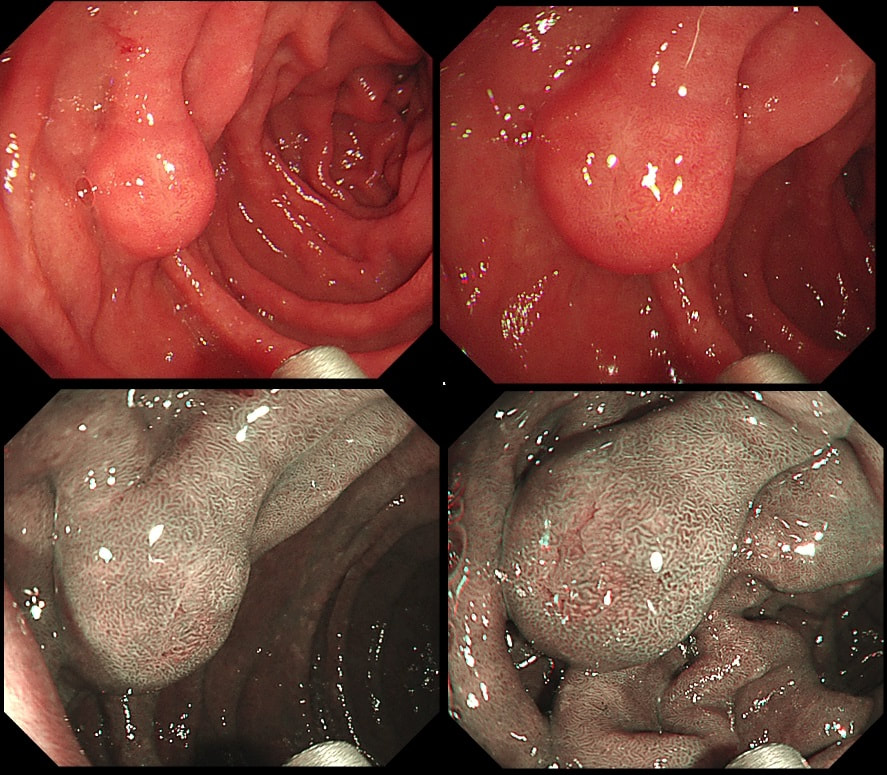
This lesion was found in the ascending colon and was subsequently referred for endoscopic resection.
WHAT IS YOUR ENDOSCOPIC DIAGNOSIS?
SSL
CORRECT!
TUBULAR ADENOMA
INCORRECT!
TUBULO-VILLOUS ADENOMA
INCORRECT!
VILLOUS ADENOMA
INCORRECT!
EXPLANATION
In the indigo carmine dye images, you can see some large, round/oval crypt openings. This was a Sessile Serrated Lesion which actually harboured HGD. 75% of our Facebook group members got the diagnosis right.
The confusing thing is that the growth pattern is that of a LST-G, which of course means that it should be a TVA! I wonder if there is any way I could have predicted the dysplasia? The fact that the lesion is rather "chunky" (but still soft) and not thin and flat, is the sign dysplasia within an SSL . The image caused some controversy back in 2014. Several endoscopists didn't believe that an endoscopic diagnosis could be made. In fact, some hinted that it would be inappropriate to second guess histology. After all, these are the guys with all the answers right? You should stand 'cap in hand' and wait patiently until they tell us what we have seen. It makes my blood boil ! Fortunately, endoscopy has moved on from those dark times. Now we recognise that we have a duty as endoscopists to contribute to the diagnosis. The diagnosis is based on an assessment of the entire lesion and it's degree of lifting. In contrast, our poor pathologists can only give an opinion on the square mm of tissue which we provide them with. That is why they need microscopes! By recognising our responsibility, we accept a duty to develop expertise. Of course, this directly benefits patients as we are more likely than our pathologist to recognise an early malignant polyp and thus target samples to the area which is the most suspicious in appearance. Our pathologists also benefit from our diagnosis, although some endoscopists believe that we shouldn't tell them anything as it could bias their assessment ☺ . To conclude, the combined endoscopic and histological assessment is what is most likely to reach the correct diagnosis - a Team Effort !!!
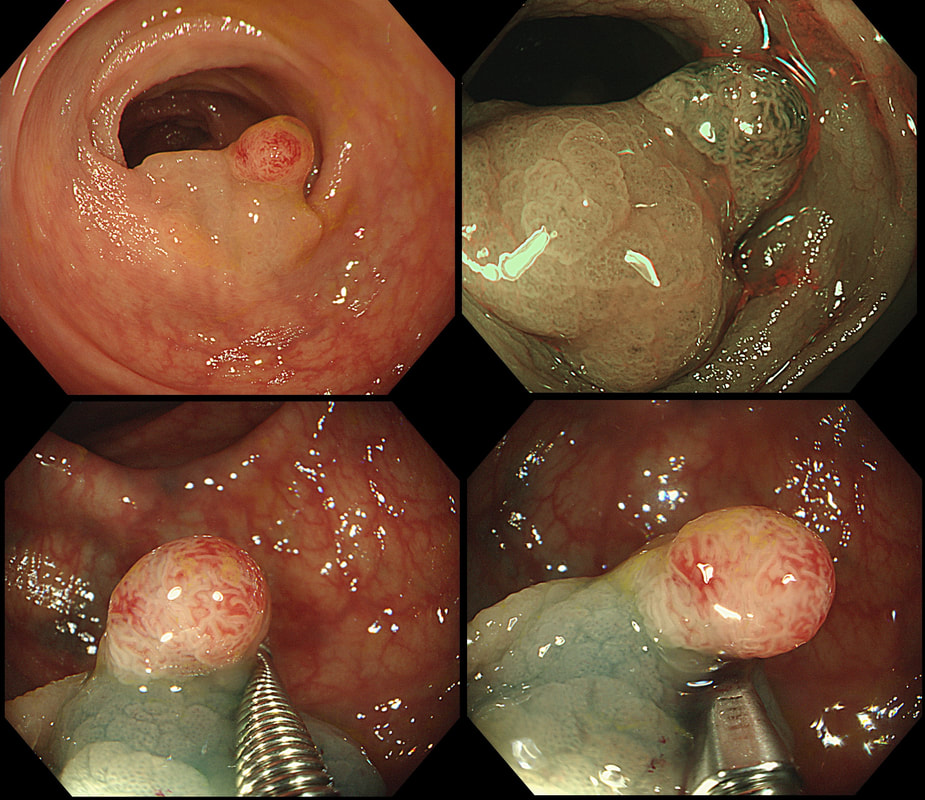
This polyp was found in the duodenum of a 65 yr old man undergoing OGD for dyspepsia.
WHAT IS YOUR DIAGNOSIS?
a) Hyperplastic Polyp
INCORRECT!
b) Lipoma
INCORRECT!
c) Neuro-Endocrine Tumour (NET)
INCORRECT!
d) Adenoma
CORRECT!
e) Carcinoma
INCORRECT!
explanation
>95% of our facebook group correctly recognised that this lesion is a sporadic duodenal adenoma. Of course, you should now consider offering your patient a colonoscopy as there is a link with colorectal adenomas.
Actually, it turned out to be tubular adenoma with low-grade dysplasia (TA+LGD). The risk of malignant conversion in a 'sporadic duodenal polyp', is probably very low. Nevertheless, I do remove these lesions in patients who are at least "reasonable fit" with a life expectancy of at least 10 yrs. Because there is a 1:20-30 risk of late bleeding even after removing a small polyp, I always apply clips to the mucosal defect. In addition, I tend to reject patients who I think may not 'cope well' with a 2 unit blood loss or an emergency laparotomy. By the way, I think that using a banding device such as the Cook 'Duette' or the Boston Sci 'captivator', is probably dangerous in the thin walled duodenum. You should also be careful with the use of the haemostatic forceps. There is a real risk of a late perforation if too much heat is applied to the thin mucosa. Finally, immediate perforations are often difficult to recognise in the duodenum. Not sure why this is but my last perforation was pinhole and entirely invisible at the time of the procedure. I know this because I watched the video clip several times afterwards. After a few hours exposure to pancreatic juice, the hole was an a centimetre in diameter (measured at the time of laparotomy) and the poor patient spent another 2 months in hospital recovering. Fortunately, 12 months later the external drains could be removed. A recent study published in GIE [Fukuhara S et al. Management of perforation related to endoscopic submucosal dissection for superficial duodenal epithelial tumors. Gastrointestinal Endoscopy 2020;91(5):1129-11370] reinforced my view. This Japanese multicentre study reported on the management of perforations following ESD resections of duodenal polyps. A perforation complicated 14% of ESD resections and only 1/3 of perforations could be closed completely endoscopically. Fortunately, even perforations which were not completely closed, only 2/36 perforations required surgery. The remainder could be managed with the placement of an 'endoscopic naso-biliary and pancreatic duct drainage tube. The duodenum is "Tiger Country" and I recommend that you avoid polypectomy here unless you have full 24 hour/364 days/year backup by expert upper GI radiology and specialist surgical backup. You need the reassurance that it will be a friendly and support specialist upper GI surgeon who is sorting out your patients biliary peritonitis at 3 O'clock in the morning!
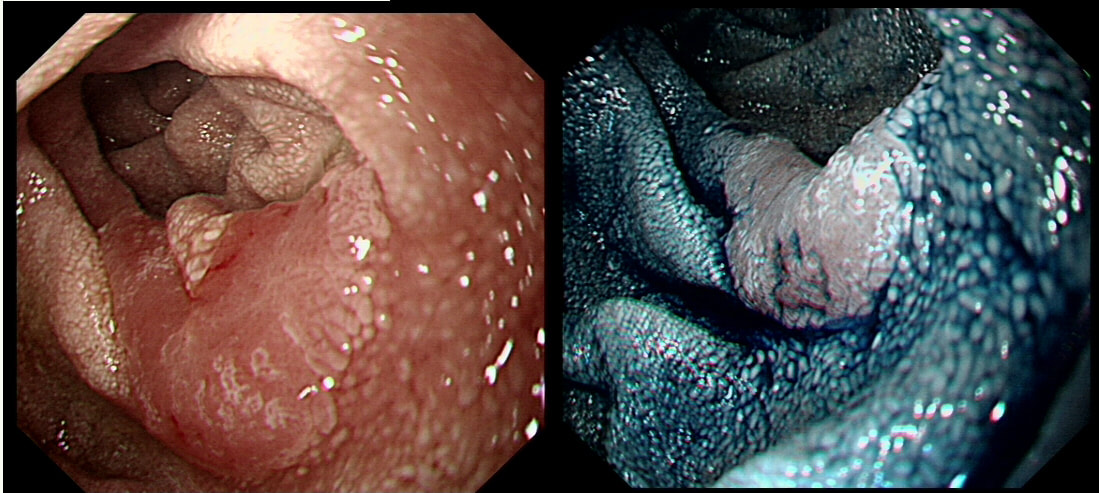
This polyp was found in a 30 year old patient with a family history of bowel cancer undergoing colonic surveillance because of a 'polyp syndrome'.
WHAT IS THE LIKELY POLYP SYNDROME?
a) Familial Adenomatous Polyposis (FAP)
INCORRECT!
b) Peutz-Jeghers syndrome
INCORRECT!
c) MUTYH associated polyposis (MAP)
INCORRECT!
d) Serrated Polyposis Syndrome)
CORRECT!
e) Lynch Syndrome (formerly HNPCC)
INCORRECT!
explanation
This proved to be a difficult question and only 15% of our facebook group got the correct answer of: Serrated Polyposis Syndrome !!!
The odd thing about this polyp is that it appears to be a tubular adenoma (slit-like crypts) arising from a broad somewhat odd looking fold. The 'fold' is actually a larger hyperplastic polyp. Of course large hyperplastic polyps are now referred to as 'sessile serrated lesions' (SSL's). You are looking at an SSL, sprouting a TA !? A 'purist' histopathologist may refer to this entity as a 'mixed serrated, adenomatous polyp'. Do you know of a polyp syndrome where patients have a mixture of SSL's and adenomatous polyps? Of course, the serrated polyposis syndrome 'SPS' (previously called the sessile serrated polyposis syndrome). It's now realised that many patients with serrated polyposis syndrome actually have a mixture of serrated and adenomatous polyps. It's possible that these patients have a greater risk of developing cancer. The correct answer is therefore 'A' ! The BSG has published a 'position statement' on the topic of SSL's which is well worth reading. The BSG sensibly recommend annual surveillance until all serrated polyps above 5mm have been cleared, following which the surveillance interval may be reduced. This is because patients with SPS do not seem to have a higher risk of developing cancer than patients with Lynch syndrome. This odd little polyp glimpsed just beyond the pyloric ring. It was a bugger to remove as it proved impossible to retrovert in the duodenal cap. In the end it was possible by pulling it into my snare, using a double channelled gastroscope. The histology is attached. WHAT IS THE DIAGNOSIS ? a) gastric heterotopia b) pyloric gland adenoma c) duodenal adenoma d) Brunner's gland hyperplasia e) Neuroendocrine tumour Explanation
OK, there is no way to beat around the bush. This turned out to be a 'pyloric gland adenoma'. I don't think that there is any way that I could have distinguished this from a 'normal' duodenal adenoma endoscopically (you also have the histology to help you though). A Brunner's gland polyp is unlikely as the lesion isn't a beautiful, symmetrical submucosal ball, covered with normal duodenal mucosa. Gastric heterotopia is never this 'polypoid' or 'flamboyant'. Finally, a NET is unlikely as these should also be 'balls', classically with a central depression in the duodenum. The first time I came across a 'Pyloric gland adenoma', I objected to the histology because the polyp was situated in the stomach. Our lovely pathologist patiently educated me and explained that in spite of the name, they don't necessarily arise from the pylorus. Instead, they got their name from the fact that they arise from 'pyloric glands' which are found in the oesophagus, stomach, pylorus and the duodenal cap. Furthermore, I didn't know that gastric adenomas are broadly classified into two types: 1) Gastric type and 2) the Intestinal type of gastric adenoma. Gastric-type adenomas are further subclassified into 'pyloric gland adenomas' and 'foveolar-type adenoma'. However, it's the intestinal-type adenomas which we endoscopists usually refer to as 'adenomas' in the upper GI tract. Pyloric gland adenomas are found in patients with Helicobacter pylori associated gastritis and 'chemical gastritis'. However, they are indeed 'neoplastic' (the cells are unstable) and may harbour low-grade dysplasia, high-grade dysplasia or even cancer. If you would like to learn more about 'Pyloric gland adenomas', this is a good link: http://www.archivesofpathology.org/doi/pdf/10.5858/arpa.2013-0613-RS This high rectal lesion was treated with chemoradiotherapy 3 months ago (left image). On re-assessment of the site, there is some erythema remaining. WHAT IS THE LIKELY NEXT STEP? a) APC ablation b) EMR c) ESD d) Further radiotherapy e) Surgery explanation
The initial staging of this rectal cancer was T2, N0. It took many years for Prof Angelita Habr-Gama (a famous colorectal surgeon from Sao Paulo, Brazil) to convince her fellow surgeons that neo-adjuvant chemoradiation for stage 0 rectal cancer could be as curative as surgery but without the need for a subsequent AP resection and a stoma. She produced a landmark paper; “Operative versus nonoperative treatment for stage 0 distal rectal cancer following chemoradiation therapy: long-term results,” [Ann Surg 2004;240(4):711-7] which proved the case. This particular patient had received chemoradiotherapy and the result has been good but NOT complete. Samples taken from that red area confirmed residual cancer. As it happens, the patient was not a surgical candidate and a brachytherapy boost was instead given. This was of course something of a trick question as both D and E are 'correct' next steps. This polyp was found in a middle aged lady undergoing gastroscopy because of indigestion. WHAT TYPE OF POLYP IS THIS? a) inflammatory polyp b) Brunner's gland hyperplasia c) lipoma d) adenomatous polyp e) neuroendocrine tumour Explanation
Of course the endoscopic appearances are that of a submucosal lesion, either a lipoma, a lymphangioectatic cyst (if soft when prodded with a biopsy forceps), or firms as you would expect with a GIST, a leiomyoma, an ectopic pancreas (although have never seen one in the duodenum, a NET or a 'Brunner's gland hyperplasia'. Of course, if a submucosal lesion is firm, the only way to make a diagnosis is to slice the mucosa open with a needle knife (or if like me you don't want to spend hundreds of pounds, use the tip of a snare with the diathermy set on 'sphincterotomy' or EndoCut I ) and then sample from deep within the lesion (don't forget to place a couple of clips afterwards). Although on balance of probabilities, a Brunner's gland hyperplasia would be the most likely diagnosis, it could make sense to obtain some samples to rule out a GIST . Seeing >10 mitotic figures/HPF is a sign of a 'bad GIST' requiring surgery. Of course, if the patient is too old for surgery, it would make little sense in obtaining any samples. I leave the decision about sampling to the 'upper GI cancer team'. Anyway, Brunner's gland hyperplasia (sometimes called a 'hamartoma' or 'adenoma' even though they are not neoplastic) are supposedly rare, only accounting for some 5% of all duodenal masses seen on radiology. In my experience, excluding lipomas and lymphoangioectatic cysts (which are both soft of course) Brunner's gland hamartomas are the most common 'firm' submucosal lesion. I usually find these in the duodenal bulb and less commonly just beyond the D1/D2 junction. |
Categories
All
|